RESEARCH ARTICLE
Occurrence and Abundance of Vibrio spp. in Traditional Shrimp Ponds of Tarakan, Indonesia: Implications for Aquaculture Biosecurity
Aquatic Life Sciences|Vol. 3, Issue 1, pp. 20-27 (2026)
Received
Sep 29, 2025Revised
Jan 7, 2026Accepted
Mar 26, 2026Published
Apr 9, 2026
Abstract
Introduction
Shrimp aquaculture represents one of the most important sectors in global fisheries, contributing significantly to food security and economic development (1). In Indonesia, Penaeus monodon (tiger shrimp) remains a key aquaculture commodity, particularly within traditional farming systems such as those found in Tarakan City, North Kalimantan. These systems rely heavily on extensive coastal and estuarine environments and are characterized by minimal technological input and limited biosecurity management (2). Despite their economic importance, traditional shrimp ponds are highly vulnerable to disease outbreaks, which pose serious threats to production sustainability.
Among the major constraints in shrimp aquaculture, infectious diseases caused by bacterial pathogens are considered one of the most serious threats to production sustainability, particularly those caused by Vibrio species (3, 4). Vibrio spp. are ubiquitous in aquatic ecosystems and are widely recognized as important opportunistic pathogens in marine environments. Their abundance is strongly influenced by environmental conditions, particularly temperature and salinity, which regulate their distribution and proliferation in coastal waters (5, 6). Clinical symptoms of vibriosis include reddish discoloration of the body, hepatopancreatic necrosis, reduced motility, and, in acute cases, mass mortality occurring within 24-48 h (7). Notably, outbreaks of Vibrio harveyi have been reported to cause mortality rates of up to 100% in shrimp larvae, resulting in severe economic losses in hatchery and grow-out systems across Asia (8).
Previous studies have established that shrimp mortality is commonly associated with Vibrio densities exceeding 10⁴ CFU/mL, while concentrations above 10⁶ CFU/mL may lead to mortality rates exceeding 90% (9). Consequently, Vibrio levels below this threshold are generally regarded as sub-pathogenic. However, traditional ponds located near agricultural land or human settlements frequently experience fluctuating environmental conditions and limited biosecurity implementation, conditions that may allow Vibrio populations to persist and fluctuate even when remaining below pathogenic thresholds (10). Conventional management practices, such as water exchange and lime application, are commonly employed but often insufficient to suppress bacterial proliferation effectively (11). Moreover, the use of antibiotics is increasingly discouraged due to the risk of antimicrobial resistance and disruption of beneficial microbial communities (12).
Although it is generally assumed that Vibrio concentrations in traditional ponds remain below pathogenic levels, this assumption may underestimate the operational risks faced by farmers. Sub-pathogenic concentrations of Vibrio spp. can persist throughout the culture cycle and may rapidly increase when environmental conditions become favorable for bacterial proliferation (13). Therefore, continuous monitoring of Vibrio abundance, even at non-lethal levels, is critical for early warning, risk assessment, and biosecurity planning in traditional aquaculture systems.
Despite the recognized importance of Vibrio monitoring, empirical data on the temporal persistence, spatial distribution, and environmental associations of Vibrio spp. in traditional P. monodon ponds in Tarakan City remain limited. Most existing studies focus on outbreak conditions or intensive farming systems, leaving a knowledge gap regarding baseline Vibrio dynamics in low-input traditional ponds. Addressing this gap is essential to support evidence-based disease prevention strategies tailored to local farming conditions.
Accordingly, this study aimed to isolate and enumerate Vibrio spp. colonies from pond water samples collected from two traditional shrimp farming locations in Tarakan City using selective Thiosulfate Citrate Bile Salt Sucrose Agar (TCBSA) and the Total Plate Count method. By examining Vibrio abundance in relation to key water quality parameters, this research establishes a local baseline for Vibrio populations, identifies sub-pathogenic fluctuation patterns, and highlights their relevance as early warning indicators for disease emergence. The findings are expected to contribute to improved pathogen monitoring and biosecurity management in traditional shrimp aquaculture systems, thereby supporting production sustainability and reducing crop losses.
Methodology
Study Design and Rationale
An exploratory survey design was applied to quantify the abundance of Vibrio spp. colonies in traditional tiger shrimp (P. monodon) ponds in Tarakan City, North Kalimantan. This design was selected to generate baseline microbiological and water quality data in extensive pond systems characterized by minimal technological input, reliance on natural tidal water exchange, and limited biosecurity implementation. Such baseline information is essential for understanding microbial dynamics in traditional aquaculture systems and for supporting early disease risk assessment under low-input management conditions.
Study Area and Sampling Strategy
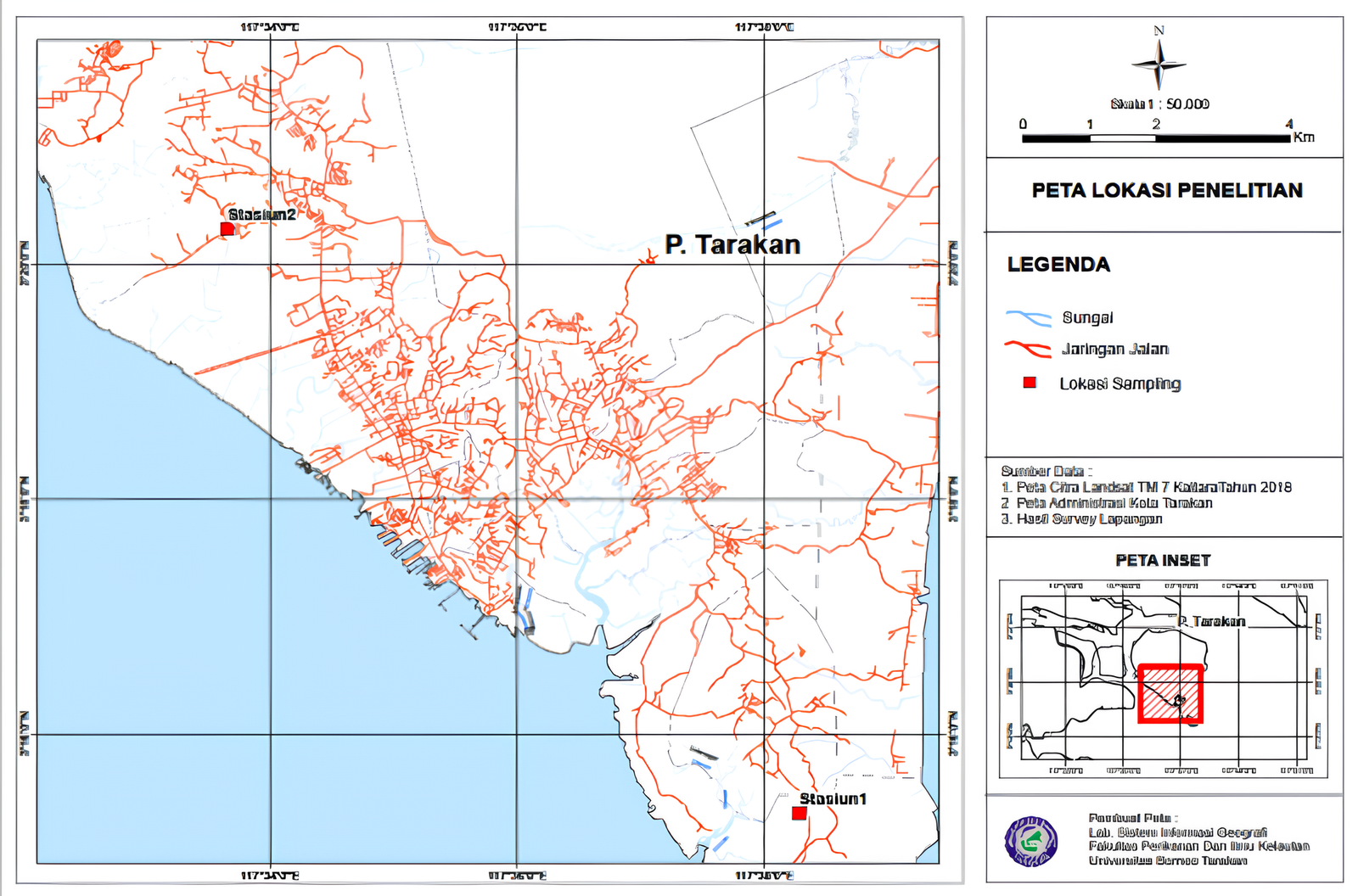
Sampling was conducted from April to July 2022 at two traditional tiger shrimp (P. monodon) ponds located in West Tarakan (Karang Harapan) and East Tarakan (Mamburungan), North Kalimantan, Indonesia, as shown in Figure 1. Each pond covered approximately 7 ha and was managed using an extensive culture system without artificial aeration or formulated feed, relying primarily on natural tidal water exchange to maintain basic water quality conditions and support natural food availability within the culture environment. Shrimp post-larvae (PL20) were stocked at an approximate density of 3, 000 post-larvae per hectare, consistent with local traditional farming practices.
Water sampling was conducted biweekly over a three-month culture period from stocking until harvest. Sampling was performed at three predetermined stations within each pond representing spatial variability in pond hydrology: (i) inlet channel, (ii) left pond margin, and (iii) right pond margin. At each station, water samples were collected at approximately 20 cm below the surface using sterile 250 mL sampling bottles that had been previously sterilized by autoclaving to ensure the elimination of potential microbial contaminants prior to use.
The sampling procedure followed sterile collection techniques in which the bottle was submerged with the opening facing downward and sealed underwater to minimize contamination from airborne microorganisms. All collected samples were placed in sterile containers, stored in an ice box, and transported to the Aquaculture Laboratory of the Faculty of Fisheries and Marine Science, Universitas Borneo Tarakan for analysis following standard laboratory sample handling protocols.
In total, the sampling design consisted of two ponds × three sampling stations × six sampling events, resulting in 36 water samples. This sampling scheme was designed to capture both spatial variability within ponds and temporal dynamics throughout the culture period, thereby providing representative baseline data on Vibrio abundance in large traditional shrimp ponds under extensive aquaculture management conditions.
Materials and Culture Techniques
All water samples were transported on ice to the Aquaculture Laboratory, Faculty of Fisheries and Marine Science, Universitas Borneo Tarakan, and processed within 6 h after collection. Vibrio spp. were isolated using Thiosulfate Citrate Bile Salt Sucrose Agar (TCBSA; Merck, Germany), a selective and differential medium commonly used for enumerating Vibrio colonies in aquatic samples.
Serial dilutions of each sample were prepared in sterile 1% NaCl solution up to 10⁻⁴. The use of 1% NaCl was intended to maintain osmotic conditions favorable for halophilic marine Vibrio spp. and to minimize osmotic stress during dilution. From each dilution, 1 mL aliquots were plated in triplicate onto TCBSA plates.
Plates were incubated at 34 ± 1 °C for 24 h, a temperature selected to promote optimal growth of Vibrio spp. commonly associated with tropical shrimp aquaculture environments. After incubation, colonies displaying typical Vibrio morphology (yellow pigmentation, circular shape, and convex elevation) were enumerated. Vibrio abundance was expressed as colony-forming units per milliliter (CFU/mL) using the standard Total Plate Count calculation, as shown in Eq. 1:
It is acknowledged that this study relied on culture-based identification using colony morphology on TCBSA agar and did not include biochemical or molecular confirmation. Molecular approaches such as 16S rRNA sequencing are increasingly used to improve taxonomic resolution of Vibrio populations in aquatic environments (14).
Water Quality Assessment
In situ water quality measurements were conducted concurrently with microbiological sampling. Parameters included temperature (°C; mercury thermometer), salinity (ppt; refractometer), dissolved oxygen (mg/L; portable DO meter, YSI), and pH (digital pH meter, Hanna Instruments). Measurements were taken in triplicate at each sampling station to capture local variability.
Data Analysis
Vibrio abundance data were summarized as mean ± standard deviation (SD). Temporal and spatial variations in bacterial counts were evaluated descriptively, with reference to the commonly accepted pathogenic threshold of 10⁴ CFU/mL. Water quality parameters were compared with standard tolerance ranges for P. monodon culture (28-32 °C, 10-35 ppt, pH 7.5-8.5, DO 4-8 mg/L). Pearson’s correlation coefficients were calculated to explore associations between Vibrio abundance and environmental parameters. Statistical analyses were performed using SPSS v.25 (IBM Corp. , USA), with significance defined at p < 0.05.
Ethical Considerations and Methodological Limitations
This study involved no human or vertebrate animal subjects. All laboratory procedures followed institutional biosafety guidelines for handling aquatic microbial samples. It is acknowledged that the absence of sediment sampling, where Vibrio populations may be higher, and the limited sample size constrain the interpretation of absolute bacterial abundance. Nevertheless, the methodology remains appropriate for establishing an initial baseline of Vibrio occurrence in traditional shrimp ponds.
Results and Discussion
Isolation of Vibrio spp.
Bacterial isolation on TCBS agar consistently revealed colonies with distinct yellow pigmentation, a characteristic feature of sucrose-fermenting Vibrio spp. As reported by Handayani et al. (2020), yellow coloration on TCBS agar results from sucrose fermentation that produces acidic metabolites, leading to a detectable color change in the medium (15). TCBS agar is widely recognized as a selective and differential medium for Vibrio spp, including V. cholerae, V. parahaemolyticus, and V. vulnificus, due to its composition of bile salts, sodium chloride, sucrose, bromothymol blue, thiosulfate, and yeast extract (16). The presence of bile salts suppresses the growth of non-Vibrio microorganisms, thereby enhancing isolation specificity (17).
Across all sampling weeks (0-10) and both pond locations, isolated colonies exhibited uniform macroscopic morphology, characterized by yellow pigmentation, circular shape, flat elevation, entire margins, and small colony size. The absence of noticeable morphological variation across time and sampling stations indicates a stable dominance of sucrose-fermenting Vibrio spp. throughout the culture period. Colony diameters ranged from 1.0 to 3.5 mm, with a mean diameter of approximately 2.0 mm, which is consistent with reported characteristics of V. alginolyticus on TCBS agar (15). As gram-negative bacteria, Vibrio spp. possess relatively thin peptidoglycan layers and a complex outer membrane structure (18). It should be noted that species-level identification in this study is presumptive and based solely on colony morphology.
Abundance of Vibrio spp.
The abundance of Vibrio spp. in traditional shrimp ponds in West and East Tarakan is summarized in Table 1 and Table 2. Overall, Vibrio populations fluctuated throughout the culture period, exhibiting clear temporal variation across sampling weeks and stations, yet consistently remaining below the pathogenic threshold of 1 × 10⁴ CFU/mL. The highest Vibrio counts were recorded at week 4 in Pond 1 (2.6 × 10³ CFU/mL) and week 4 in Pond 2 (1.2 × 10³ CFU/mL), indicating a mid-culture peak common to both pond systems.
| Week | TS1 (CFU/mL) | TS2 (CFU/mL) | TS3 (CFU/mL) | Mean (CFU/mL) |
|---|---|---|---|---|
| 0 | 1.0 × 10¹ | 1.5 × 10¹ | 2.0 × 10¹ | 1.5 × 10¹ |
| 2 | 3.5 × 10¹ | 1.5 × 10¹ | 2.6 × 10¹ | 2.5 × 10¹ |
| 4 | 1.6 × 10² | 2.6 × 10³ | 1.0 × 10² | 9.1 × 10² |
| 6 | 1.1 × 10² | 1.1 × 10³ | 1.6 × 10² | 4.5 × 10² |
| 8 | 3.5 × 10² | 3.0 × 10¹ | 8.3 × 10² | 4.0 × 10² |
| 10 | 0 | 0 | 0 | 0 |
| Notes: TS1 = inlet; TS2 = left margin; TS3 = right margin. | ||||
In Pond 1, total Vibrio counts (TVC) ranged from 1.5 × 10¹ to 2.6 × 10³ CFU/mL between weeks 0 and 8, remaining well below the critical threshold established as a risk level for disease outbreaks (Ministry of Marine Affairs and Fisheries, Regulation No. 75/2016). No Vibrio colonies were detected at week 10, coinciding with the harvest period (Table 1). Initial bacterial abundance during weeks 0-2 was low (< 3.5 × 10¹ CFU/mL), suggesting favorable conditions for shrimp culture during the early stocking phase. A transient increase occurred at week 4, particularly at the left margin station (TS2), where counts reached 2.6 × 10³ CFU/mL. This increase is likely associated with enhanced availability of organic substrates derived from phytoplankton and zooplankton proliferation, which is known to promote Vibrio growth (19). Additional environmental factors, including rainfall-induced temperature fluctuations and seawater intrusion during high tide, may have further contributed to this mid-cycle peak (20). Subsequently, Vibrio abundance declined at weeks 6 and 8 (4.0-4.6 × 10² CFU/mL), coinciding with more stable pond conditions and shrimp reaching later culture stages characterized by higher resilience.
Despite these fluctuations, Vibrio abundance in Pond 1 remained sub-pathogenic throughout the culture cycle. The pond yielded 90 kg of shrimp with a market size of 35 individuals per kilogram and an estimated survival rate of approximately 90% from an initial stocking of 3, 000 post-larvae. No clinical signs of bacterial, viral, or parasitic disease were observed during the production cycle, supporting the interpretation that Vibrio levels detected did not pose a direct disease risk.
In Pond 2, total Vibrio counts ranged from 2.4 × 10¹ to 1.2 × 10³ CFU/mL between weeks 0 and 10 (Table 2), similarly remaining below the pathogenic threshold. Bacterial abundance was moderate at week 0 (9.7 × 10¹ CFU/mL), declined at week 2, and increased markedly at week 4, followed by slightly lower yet still elevated values at week 6. Thereafter, Vibrio abundance decreased toward the end of the culture period, reaching 1.9 × 10² CFU/mL at week 10. Such fluctuations are typical of pond-based aquaculture systems and are strongly influenced by environmental dynamics, particularly phytoplankton and zooplankton proliferation, which provide organic substrates supporting Vibrio growth (19), as well as variations in temperature, dissolved oxygen, and salinity (21).
| Week | TS1 (CFU/mL) | TS2 (CFU/mL) | TS3 (CFU/mL) | Mean (CFU/mL) |
|---|---|---|---|---|
| 0 | 2.6 × 10² | 1.5 × 10¹ | 1.7 × 10¹ | 9.7 × 10¹ |
| 2 | 0 | 7.3 × 10² | 0 | 2.4 × 10¹ |
| 4 | 1.35 × 10³ | 1.1 × 10³ | 1.2 × 10³ | 1.2 × 10³ |
| 6 | 2.2 × 10² | 1.5 × 10² | 3.0 × 10³ | 1.1 × 10³ |
| 8 | 8.5 × 10¹ | 1.1 × 10³ | 2.4 × 10² | 4.6 × 10² |
| 10 | 1.1 × 10² | 3.0 × 10¹ | 4.4 × 10² | 1.9 × 10² |
| Notes: TS1 = inlet; TS2 = left margin; TS3 = right margin. | ||||
Unlike Pond 1, no significant shrimp harvest was reported from Pond 2. According to farmer observations, this outcome was primarily attributed to predation during the early stocking phase rather than disease-related mortality, as no mass mortalities or clinical signs of infection were observed. Instead, milkfish (Chanos chanos) were co-cultured, yielding approximately 3, 500 kg from an initial stocking of 30, 000 fish, with an average body weight of 400-600 g and a survival rate of 23%.
Comparatively, Vibrio abundance in Pond 2 was generally higher than that observed in Pond 1, although still within non-pathogenic ranges. This difference may be explained by variations in pond management practices. Pond 1 benefited from routine water exchange and was located away from human settlements, whereas Pond 2 lacked regular monitoring and water exchange, potentially leading to increased organic loading and higher bacterial proliferation. It is also important to note that this study focused exclusively on water samples. Previous studies have shown that Vibrio spp. are often more concentrated in pond sediments, where organic matter accumulation supports microbial growth (22). Consequently, the high organic content typically found in sediments (Avnimelech & Ritvo, 2003) suggests that Vibrio abundance may be underestimated when analyses are limited to the water column alone (23).
Overall, the observed temporal fluctuations demonstrate that Vibrio spp. persist in traditional shrimp ponds throughout the culture cycle, even under sub-pathogenic concentrations. These findings highlight that Vibrio dynamics are influenced not only by water quality parameters but also by organic matter availability, microbial interactions, and environmental fluctuations that shape Vibrio population structure in aquatic ecosystems (24). Continuous monitoring of Vibrio abundance, therefore, remains important as an early warning tool for disease risk management in traditional aquaculture.
Water Quality Parameters
Water quality parameters were monitored during the sampling period to characterize the environmental conditions of the ponds. Although microbial sampling was conducted biweekly, physicochemical parameters were recorded periodically to represent the overall environmental conditions of the pond systems throughout the culture cycle. The measured values for Ponds 1 and 2 are presented in Table 3.
| Parameter | Pond 1 - TS1 | Pond 1 - TS2 | Pond 1 - TS3 | Pond 2 - TS1 | Pond 2 - TS2 | Pond 2 - TS3 | Standard Range* |
|---|---|---|---|---|---|---|---|
| Temperature (°C) | 31.8 | 31.4 | 31.4 | 31.7 | 32.0 | 31.7 | 28-32 |
| pH | 7.80 | 7.29 | 7.44 | 7.62 | 7.48 | 7.56 | 6.8-8.7 |
| Salinity (ppt) | 17.6 | 17.8 | 17.9 | 17.4 | 17.3 | 17.3 | 10-35 |
| DO (mg/L) | 5.71 | 5.29 | 5.76 | 5.87 | 5.68 | 5.54 | 4-8 |
Although temporal microbial monitoring was conducted across multiple sampling weeks, the physicochemical measurements presented in Table 3 represent the general environmental conditions of the ponds during the study period. The relatively narrow ranges observed for temperature (31.4-32.0 °C), pH (7.29-7.80), salinity (17.3-17.9 ppt), and dissolved oxygen (5.29-5.87 mg/L) indicate that water quality remained relatively stable throughout the culture cycle. Such environmental stability may partly explain the weak correlations observed between Vibrio abundance and the measured physicochemical variables.
All recorded parameters remained within the optimal ranges for P. monodon culture, including temperature (28-32 °C), pH (6.8-8.7), salinity (10-35 ppt), and dissolved oxygen (4-8 mg/L). These conditions indicate that both ponds provided a generally favorable environment for shrimp growth and survival. Nevertheless, Vibrio spp. were consistently detected throughout the culture period, suggesting that their presence in traditional ponds in Tarakan is not solely determined by physicochemical water quality conditions (25).
These findings support previous studies reporting that Vibrio spp. may persist at sub-pathogenic levels even under optimal water quality conditions, driven by factors such as organic matter availability, plankton dynamics, and sediment-associated processes.
To further evaluate the relationship between environmental conditions and bacterial abundance, Pearson correlation analysis was conducted between Vibrio counts and the measured water quality parameters (Table 4) to statistically assess possible environmental influences on bacterial populations.
| Parameter | r | p-value |
|---|---|---|
| Temperature | 0.18 | 0.62 |
| pH | -0.21 | 0.55 |
| Salinity | 0.12 | 0.73 |
| Dissolved Oxygen | -0.27 | 0.44 |
| Notes: p > 0.05 indicates non-significant correlation. | ||
Pearson correlation analysis showed weak relationships between Vibrio abundance and all measured water quality parameters (Table 4). Temperature showed a weak positive correlation with Vibrio abundance (r = 0.18, p = 0.62), while salinity also exhibited a weak positive association (r = 0.12, p = 0.73). In contrast, pH (r = −0.21, p = 0.55) and dissolved oxygen (r = −0.27, p = 0.44) showed weak negative correlations with Vibrio counts. However, none of these relationships were statistically significant (p > 0.05), indicating that variations in physicochemical parameters within the observed ranges did not strongly influence Vibrio population dynamics.
Although the Vibrio levels observed in this study remained non-lethal, unfavorable changes in water quality may exacerbate bacterial proliferation and increase disease risk. Kopprio et al. (2017) reported that Vibrio abundance is closely linked to broader biogeochemical cycles and environmental variability (26). Furthermore, Vibrio infections may become pathogenic when dissolved oxygen declines below 6 mg/L, salinity drops to 10-15 ppt, or temperature deviates substantially from optimal ranges, particularly affecting shrimp larvae (27). These findings emphasize that routine Vibrio monitoring should be integrated with water quality management as a preventive strategy rather than a reactive measure. These findings emphasize that routine Vibrio monitoring remains essential as an early warning measure, even when conventional water quality indicators appear optimal.
Conclusion
This study confirmed the consistent presence of Vibrio spp. in traditional shrimp ponds in Tarakan, with bacterial abundance fluctuating throughout the culture period but remaining below the pathogenic threshold of 1 × 10⁴ CFU/mL thereby indicating relatively low microbiological risk under the observed culture conditions. Although temperature, salinity, pH, and dissolved oxygen values were within optimal ranges for P. monodon culture, the correlation between Vibrio abundance and these water quality parameters was weak and not statistically significant suggesting that physicochemical stability alone does not fully explain bacterial population variability. This limited relationship reflects the relatively stable physicochemical conditions observed during the study and indicates that Vibrio population dynamics in traditional ponds are influenced more strongly by other factors, such as organic load, sediment-associated processes, and pond management practices that collectively shape the microbial ecology of extensive aquaculture environments. These findings underscore the importance of continuous Vibrio monitoring as part of routine pond management, even under favorable water quality conditions to support early detection of potential ecological imbalance. Strengthening biosecurity measures, including regular water exchange, sediment management, and improved farm monitoring, is essential to reduce potential disease risks, prevent unexpected outbreaks, and enhance the long-term sustainability of traditional shrimp aquaculture systems particularly in low-input traditional farming systems where environmental control is limited.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
All data generated or analyzed during this study are included in this published article.
Ethics Statement
Ethical approval was not required for this study.
Funding Information
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
References
- Shulhan S. Dampak sosial ekonomi pengembangan budidaya tambak udang. Karat J Pembang Sumenep. 2021; 1 (1): 1–8.
- Ananta N, Rachmawani D, Iromo H. Analisis Usaha Tambak Tradisional di Kalimantan Utara (Studi Kasus Petambak di Kota Tarakan). Pros Semin Nas Teknol Komput dan Sains. 2025; 3 (1): 109–114.
- Felix F, Nugroho TT, Silalahi S, Octavia Y. Skrining bakteri Vibrio sp asli Indonesia sebagai penyebab penyakit udang berbasis teknik 16S ribosomal DNA. J Ilmu dan Teknol Kelaut Trop. 2011; 3 (2): 85–99.
- Austin B, Zhang XH. Vibrio harveyi: a significant pathogen of marine vertebrates and invertebrates. Lett Appl Microbiol. 2006 Aug; 43 (2): 119–24.
- Putra SF. , Tumbol RA, Undap SL, Kreckhoff RL, Pangemanan PN. Identifikasi bakteri Vibrio spp. penyebab vibriosis pada udang vanname (Litopenaeus vannamei) di PT. Pilar Persada Parigi. e-Journal Budid Perair. 2024; 12 (2): 2024.
- Vezzulli L, Grande C, Reid PC, Hélaouët P, Edwards M, Höfle MG, et al. Climate influence on Vibrio and associated human diseases during the past half-century in the coastal North Atlantic. Proc Natl Acad Sci. 2016 Aug 23; 113 (34).
- Apriliani M, Sarjito, Haditomo AHC. Keanekaragaman agensia penyebab vibriosis pada udang vaname (Litopenaeus vannamei) dan sensitivitasnya terhadap antibiotik. J Aquac Manag Technol. 2016; 5 (1): 98–107.
- De Schryver P, Defoirdt T, Sorgeloos P. Early mortality syndrome outbreaks: a microbial management issue in shrimp farming? PLoS Pathog. 2014 Apr; 10 (4): e1003919.
- M. Prakash D, Karmegam N. A study on bacterial flora associated with fresh water prawn, Macrobrachium rosenbergii. Int J Curr Res Acad Rev. 2013; 1 (1): 25–32.
- Kautsky N, Rönnbäck P, Tedengren M, Troell M. Ecosystem perspectives on management of disease in shrimp pond farming. Aquaculture. 2000 Nov; 191 (1–3): 145–61.
- Boyd CE. Environmental management of shrimp farms in Asia to promote healthy shrimp and reduce negative impacts. Asian Fish Sci. 2011; 23 (4): 463–81.
- Holmström K, Gräslund S, Wahlström A, Poungshompoo S, Bengtsson BE, Kautsky N. Antibiotic use in shrimp farming and implications for environmental impacts and human health. Int J Food Sci Technol. 2003 Mar 7; 38 (3): 255–66.
- Baker-Austin C, Oliver JD, Alam M, Ali A, Waldor MK, Qadri F, et al. Vibrio spp. infections. Nat Rev Dis Prim. 2018 Jun 21; 4 (1): 1–19.
- Thompson JR, Polz MF. Dynamics of Vibrio Populations and Their Role in Environmental Nutrient Cycling. In: The Biology of Vibrios. Washington, DC, USA: ASM Press; 2014. p. 190–203.
- Handayani DW, Diansyah G, Isnaini. Analisis koloni bakteri Vibrio sp dan kualitas air pada air budidaya Juwana kuda laut (Hippocampus sp). Maspari J. 2020; 12 (1): 1–8.
- Pfeffer C, Oliver JD. A comparison of thiosulphate-citrate-bile salts-sucrose (TCBS) agar and thiosulphate-chloride-iodide (TCI) agar for the isolation of Vibrio species from estuarine environments. Lett Appl Microbiol. 2003; 36 (3): 150–1.
- Syafrina Sari Lubis, Arif Sardi, Nailul Muna. Enumeration and pathogenicity test of Vibrio sp on green mussels (Perna viridis) from the Krueng Cut area of Aceh Besar. KENANGA J Biol Sci Appl Biol. 2022 Apr 30; 2 (1).
- Beveridge TJ. Structures of Gram-Negative Cell Walls and Their Derived Membrane Vesicles. J Bacteriol. 1999 Aug 15; 181 (16): 4725–33.
- Colwell RR. Global Climate and Infectious Disease: The Cholera Paradigm. Science (80-). 1996 Dec 20; 274 (5295): 2025–31.
- Lutz C, Erken M, Noorian P, Sun S, McDougald D. Environmental reservoirs and mechanisms of persistence of Vibrio cholerae. Front Microbiol. 2013 Dec 16; 4: 375.
- Brumfield KD, Chen AJ, Gangwar M, Usmani M, Hasan NA, Jutla AS, et al. Environmental Factors Influencing Occurrence of Vibrio parahaemolyticus and Vibrio vulnificus. Semrau JD, editor. Appl Environ Microbiol. 2023 Jun 28; 89 (6).
- Shakibazadeh S, Saad CR, Christianus A, Kamarudin MS, Sijam K, Nor Shamsudin M, et al. Bacteria flora associated with different body parts of hatchery reared juvenilePenaeus monodon, tanks water and sediment. Ann Microbiol. 2009 Sep; 59 (3): 425–30.
- Avnimelech Y. Shrimp and fish pond soils: processes and management. Aquaculture. 2003 Apr 14; 220 (1–4): 549–67.
- Le Roux F, Blokesch M. Eco-evolutionary Dynamics Linked to Horizontal Gene Transfer in Vibrios. Annu Rev Microbiol. 2018 Sep 8; 72 (1): 89–110.
- Gusman E, Firman. Identifikasi bakteri Vibrio sp. pada udang windu (Penaeus monodon) di tambak tradisional Kota Tarakan. J Harpodon Borneo. 2012; 5 (2): 173–183.
- Kopprio GA, Streitenberger ME, Okuno K, Baldini M, Biancalana F, Fricke A, et al. Biogeochemical and hydrological drivers of the dynamics of Vibrio species in two Patagonian estuaries. Sci Total Environ. 2017 Feb; 579: 646–56.
- Bhassu S, Shama M, Tiruvayipati S, Soo TCC, Ahmed N, Yusoff K. Microbes and pathogens associated with shrimps - implications and review of possible control strategies. Front Mar Sci. 2024 Oct 7; 11.