RESEARCH ARTICLE
GC-MS Analysis and In Vivo Antimalarial Activities of Seed Extract and Solvent Fractions of Telfairia occidentalis in Plasmodium berghei-infected Mice
Academic Editor: Garnadi Jafar
Sciences of Pharmacy|Vol. 5, Issue 1, pp. 75-84 (2026)
Received
Oct 16, 2025Revised
Jan 13, 2026Accepted
Feb 19, 2026Published
Mar 6, 2026
Abstract
Introduction
Malaria remains one of the major health threats globally. Of the estimated 263 million malaria cases recorded worldwide in 2023, 94% (246 million cases) occurred in Africa. Five African countries; Nigeria (25.9%), the Democratic Republic of the Congo (12.6%), Uganda (4.8%), Ethiopia (3.6%), and Mozambique (3.5%), together accounted for 52% of the global malaria burden (1). Similarly, 95% of the estimated 597, 000 global malaria deaths in 2023 were recorded in the WHO African Region. Four African countries accounted for more than 50% of these deaths, with 39.3% of all global malaria deaths in children under five years occurred in Nigeria (1, 2). Although reasons advanced for the upwards trends in malaria cases and deaths included lack of funding and investment, particularly in high-burden African countries, the main reason might not be far from poverty and poor economic condition. Not only are the major artemisinin combination therapies (ACTs) very costly and beyond the reach of the poor populace, but many people are also financially handicapped to access health facilities for treatment. Quite worrisome is the report of artemisinin partial resistance confirmed in Eritrea, Rwanda, Uganda and Tanzania, and suspected in Ethiopia, Sudan, Namibia and Zambia (1). The emerging piperaquine resistance that has emerged in some South American countries (1), which will likely spread and reduce the efficacy of dihydroartemisinin–piperaquine coupled with the inability of the currently introduced vaccines to provide lasting efficacy and durable protection, poses a serious threat to the control of malaria menace and deadly consequences.
In spite of significant gains achieved in the management of malaria over the past years, the above 2023 malaria data indicate that malaria is a serious threat to lives in Africa, and Nigeria in particular sequel to emergence of partial resistance to artemisinin in some Arican countries (1). Therefore, instigating researchers to search for safe and cost-effective treatments. Medicinal plants provide the reservoirs for potential efficacious antimalarial drugs including the front-line drug, artemisinin and others. Previously we have investigated a number of medicinal plants in Nigeria in vitro and in vivo (3-8) for the development of potential treatment of malaria.
Telfairia occidentalis Hook, is a fluted pumpkin of the Cucurbitaceae family widely consumed as food in Nigeria, especially in the Niger-Delta region and the Eastern part of the country (9). It is a widely consumed vegetable across Nigeria, particularly in the Niger-Delta and Eastern regions, where various meals are prepared from its leaves, stem, and seeds (10). The seeds are very nutritious and are eaten roasted or boiled. The seed has history of being effective in the treatment and prevention of prostrate disorders. The seed extract has been reported to exert antidiabetic (11) cellular antioxidant, immunodulatory, anticancer, antiinflammatory (12), antiplasmodial (13), antioxidant (14), analgesic (14, 15), genotoxic and cytotoxic (16), in vivo inhibitory effect alpha amylase and alpha glucosidase (17), antiulcer (18) and antiprostatic (19) activities. Phytochemical studies of the extract have shown the presence of alkaloid, flavonoid, tannins, terpenes, saponin, and cardiac glycosides (20). Some terpenoids and fatty acid esters including 16-octadecenoic acid methyl ester; octadecanoic acid, terpinen-4-ol; trans-β-ocimene and borneol have previously been reported in the seed extract (12). While HPLC characterisation of the seed extract and fractions revealed the presence of eleven flavonoids; kaempferol, catechin, epicatechin, anthocyanidin, naringenin, flavonones, flavones, rutin, naringin, and resveratol in the seed extract and fractions with butanol fraction having the highest concentration. Also, alkaloids such as ribalinidine, ammodendrine, spartein and lunamarin were found in the seed extract and fractions (20). Although earlier studies (13, 20), had reported on antiplasmodial potential of the T. occidentalis crude seed extract against P. berghei infection in mice, investigation of antimalarial potentials of its solvent fractions to identify the most active fraction has not been carried out. The present study was designed to evaluate the activities of seed extract and fractions of T. occidentalis in Plasmodium berghei-infected mice and to analyse the active antiplasmodial fraction for possible involvement of volatile phytochemical compounds in the antimalarial activities of the seed extract and fractions using GCMS.
Materials and Methods
Plant Collection and Extraction
Fresh seeds of T. occidentalis were purchased from Itam market in Itu L. G. A, Akwa Ibom State, Nigeria, in June, 2023. The seeds were previously identified and authenticated by a taxonomist in the Department of Botany and Ecological Studies, University of Uyo, Uyo, Nigeria. Herbarium specimens (UUPH 1 (b)) were deposited at Department of Pharmacognosy and Natural Medicine Herbarium, University of Uyo. The fresh seeds of the plant were dried on laboratory table for 2 weeks and reduced to powder. The seeds powder (1 kg) was macerated in 50% ethanol (Sigma-Aldrich, USA) (5000 mL) for 72 h, while the remaining part (2 kg) was successively and gradiently macerated for 72 h in 5000 mL of each of, n-hexane, dichloromethane, ethyl acetate and methanol (Sigma-Aldrich, USA) respectively, along their polarity to give the corresponding gradient fractions for each solvent. The liquid filtrate of crude extract and solvent fractions were concentrated and evaporated to dryness in vacuo 40 °C using a rotary evaporator (Yamato scientific co. Ltd, USA). The various yields were calculated and the extract/solvent fractions were stored in a refrigerator at -4 °C, until used for the proposed experiments.
Microorganism
Chloroquine-sensitive strain of Plasmodium berghei (ANKA strain) was obtained from the National Institute of Medical Research (NIMR), Yaba Lagos, Nigeria and maintained by subpassage of blood from infected to healthy mouse once every 7-8 days.
Parasite Inoculation
Each mouse used in the experiment was inoculated intraperitoneally with 0.2 mL of infected blood containing about 1 x 107 P. berghei parasitized erythrocytes collected from an infected-mice with 20-30% parasitemia. The inoculum consisted of 5 x 107 P. berghei infected erythrocytes per milliliter prepared by determining both the percentage parasitemia and the erythrocytes count of the donor mouse and diluting the blood with isotonic saline in proportions indicated by both determinations (7, 21). Parasitemia was monitored by standard methods; thin blood smears were made on glass slides, fixed using methanol, and stained using Giemsa stain (Sigma, USA), and parasitemia was counted using a microscope (Nikon, UK) and was calculated as a percentage of infected red blood cells (RBCs) relative to the total number of cells in a microscopic field at ×100 magnification according to the formula of Peters and Robinson, 1992 (22) as shown in Eq. 1.
Eq, 1 | where parasitemia (%) is the percentage of infected erythrocytes relative to the total erythrocytes counted.
Experimental Animals
Swiss albino mice (18-25 g), male and female, used in the study were obtained from the University of Uyo’s animal house. They were kept in standard plastic cages in a well-ventilated room and left to acclimatized for a period of 10 days before the experiments. The mice were fed on standard pelleted diet and water ad libitum, kept under ambient temperature (28 ± 2 °C) and illuminated environment of 12: 12 h dark/light cycle. The care and use of animals were conducted in accordance with the National Institute of Health Guide for the Care and Use of laboratory Animals (23). Prior ethical approval for the study was obtained from the University of Uyo’s Animal Ethics Committee (UU/CHS/IHREC/23/VOL.1/36).
Drug Administration
In this study, extract and fractions dissolved in 12% Tween 80, chloroquine and pyrimethamine (Sigma, USA) dissolved in distilled water were administered orally with the aid of a stainless metallic feeding cannula.
Evaluation of the In Vivo Antimalarial Activities of Crude Extracts and Solvent Fractions of T. occidentalis Seed:
Evaluation of Suppressive Activities of the Crude Extract and Solvent Fractions of T. occidentalis Seed (4-Day Test)
This test was used to evaluate the schizontocidal activity of the crude extract and solvent fractions as well as chloroquine against early P. berghei infection in mice. This was done as described previously (24, 25). Forty-five mice were randomly divided into nine groups of five (5) mice each. On the first day (D0), the 45 mice were infected with the parasite and randomly divided into various groups based on their body weights. They were administered the crude extract, gradient fractions, chloroquine and distilled water. Based on the previously established LD50 of 3460 mg/kg of the seed extract (25), the administered doses were chosen as fractions (1/6.25, 1/12.5 and 1/25) of the LD50 and the the mice in groups 1-3 were given 138 mg/kg, 276 mg/kg and 553 mg/kg of crude extract respectively, while groups 4, 5, 6, and 7 were administered 276 mg/kg of n-hexane, dichloromethane, ethyl acetate, and methanol solvent fractions respectively. Group 8 was given 5 mg/kg of chloroquine (positive control) and group 9 was given 10 mL/kg of distilled water (negative control) for four consecutive days (D0-D3) between 8am to 9am. On the fifth day (D4), thin films were made from the tail blood. The films were stained with Giemsa stain to reveal parasitized erythrocytes counted out of 500 in a random field of the microscope. The average suppression of parasitemia was calculated (24) as follows: (average % parasitemia positive control – average % parasitemia negative control) / (average % parasitemia negative control). The mean survival time of the mice in each treatment group was determined over a period of 29 days (D0-D28).
Evaluation of Prophylactic Activities of the Crude Extract and Solvent Fractions of T. occidentalis Seed
This was evaluated using a standard method described previously (26, 7). The mice were randomly divided into nine groups of five mice per group. Groups 1-3 were given 138, 276 and 553 mg/kg of crude extract respectively, groups 4, 5, 6, and 7 were given 276 mg/kg of n-hexane, ethyl acetate, dichloromethane, and methanol solvent fractions respectively, group 8 was given 1.2 mg/kg of pyrimethamine (positive control) and group 9 was given 10 mL/kg of distilled water (negative control). Administration of the extract and fractions continued for three consecutive days (D0-D2). On the fourth day (D3), the mice were inoculated with P. berghei. The parasitemia level was assessed by blood smears 72 h later. The mean survival time of the animals was calculated over a period of 29 days.
Evaluation of the Curative Activities of the Crude Extract and Solvent Fractions of T. occidentalis Seed
This test was done according to standard method (27) and used to evaluate the schizontocidal activity of the extract, fractions and chloroquine in established plasmodial infection. P. berghei was injected intraperitoneally into forty five (45) mice on the first day (D0). Seventy two hours later (D3), the mice were divided into nine groups of five mice per group. Groups 1-3 were given different doses of extract, 138, 276 and 553 mg/kg respectively, groups 4-7 were given 276 mg/kg of n-hexane, ethyl acetate, dichloromethane, and methanol solvent fractions respectively, group 8 was given 5 mg/kg chloroquine (positive control) and group 9 was given 10 mL/kg distilled water (negative control). The crude extract, gradient fractions and chloroquine were administered once daily for 5 days. Giemsa stained thin smears were prepared from tail blood samples collected on each day of treatment to monitor the parasitemia level. Rectal temperatures of the mice were taken on days 0, 3, 5, and 7. The mean survival time (MST) of the mice in each group was determined over a period of 29 days (D0-D28).
Gass Chromatography Mass-Spectrometry Analysis
Gas chromatography-mass spectrometry (GC-MS) data of the active solvent fraction (methanol) were recorded on an An Agilent 6890N gas chromatography equipped with an autosampler connected to an Agilent Mass Spectrophotometric Detector. One microliter of the sample was injected in the pulsed spitless mode onto a 30 m x 0.25 mm ID DB 5MS coated fused silica column with a film thickness of 0.15 micrometer. Helium gas was used as a carrier gas, and the column head pressure was maintained at 20 psi to give a constant of 1ml/min. Other operating conditions were preset. The column temperature was initially held at 55 °C for 0.4min, increased to 200 °C at a rate of 25 °C/mins, then to 280 °C at a rate of 8 °C/mins and to a final temperature of 300 °C at a rate of 25 °C/mins, held for 2 mins. The identification time was based on retention time. Components with lower retention time elute first before the ones of higher retention time. The fractions were directly injected in n-hexane as described previously (28). The phytochemicals were identified by comparison of spectra in the NIST 2011 database.
Statistical Analysis
Data collected were analyzed using one way analysis of variance (ANOVA) followed by Tukey’s multiple comparison post-test (Graph pad prism software Inc. La Jolla, CA, USA). Values were expressed as mean ± SEM and significance relative to control were considered at p < 0.05.
Results
Yields of Crude Extract and Solvent Fractions
The percentage yields of the crude extract and solvent fractions were; crude 21.03%, n-hexane 1.6%, DCM 2.82%, ethyl acetate 1.04%, methanol 2.1%.
Suppressive Activities of Ethanol Crude Extract and Solvent Fractions of T. occidentalis Seed
The seed extract and its solvent fractions exerted dose-dependent reductions in parasitemia of the treated mice in various groups. The reductions were statistically significant (p < 0.05) at all doses administered when compared to the negative control. The methanol solvent fraction exerted the highest suppressive effect (65.67%) with MST of 18.33 ± 3.71 days followed by n-hexane fraction (43.27%) with MST of 12.00 ± 1.00 days. Although the extracts’ treatments resulted in considerable prolongation of the mean survival time (m. s. t) of the infected mice, these were only significant (p < 0.01-0.001) in the groups treated with the middle and high doses of the crude extract (276 and 553 mg/kg), ethyl acetate and methanol solvent fractions, when compared to the negative control (Table 1).
| Treatment | Dose (mg/kg) | Parasitaemia | Chemosuppression (%) | MST |
|---|---|---|---|---|
| Control | - | 30.39 ± 1.62 | - | 7.00 ± 0.57 |
| Extract | 138 | 20.56 ± 1.04c | 32.34 | 9.66 ± 0.88 |
| 276 | 15.96 ± 0.95c | 47.48 | 17.00 ± 2.08b | |
| 553 | 13.35 ± 1.22c | 56.07 | 18.33 ± 1.45c | |
| n-hexane | 276 | 17.24 ± 0.66c | 43.27 | 12.00 ± 1.00 |
| Dichloromethane | 276 | 18.03 ± 0.56c | 40.67 | 12.00 ± 1.00 |
| Ethyl acetate | 276 | 19.93 ± 1.62c | 34.41 | 11.33 ± 1.45 |
| Methanol | 276 | 10.43 ± 2.67c | 65.67 | 18.66 ± 3.71c |
| Chloroquine | 5 | 5.04 ± 0.61c | 77.94 | 29.50 ± 0.34c |
| Note: Values are expressed as mean ± SEM. Significant relative to control. ap < 0.05; bp < 0.01; cp < 0.001. n = 5. | ||||
Prophylactic/Repository Activities of Ethanol Crude Extract and Gradient Extractof T. occidentalis Seed
The seed crude extract exerted dose-dependent reductions of parasitemia in the treated groups with the highest dose (553 mg/kg) producing the highest chemosuppression of 55.39% and m. s. t value of 17.66 ± 2.18 days. These reductions were statistically significant relative to the negative control (p < 0.001). The dichloromethane fraction caused a chemosuppressive effect of 46.55% with MST value of 14.33 ± 0.33% followed by ethyl acetate fraction with chemosuppressive activity of 39.21% and m. s. t value of 13.33 ± 0.66 days which was not significant (p > 0.05) when compared with negative control. The standard drug, pyrimethamine, 1.2 mg/kg exhibited a prophylactic activity of 81.82% (Table 2).
| Treatment | Dose (mg/kg) | Parasitaemia | Chemosuppression (%) | MST |
|---|---|---|---|---|
| Control | - | 25.22 ± 2.14 | - | 8.00 ± 0.57 |
| Extract | 138 | 20.64 ± 1.48 | 18.16 | 11.33 ± 0.88 |
| 276 | 16.33 ± 1.32c | 35.24 | 12.66 ± 2.96 | |
| 553 | 11.25 ± 1.54c | 55.39 | 17.66 ± 2.18b | |
| n-hexane | 276 | 22.44 ± 1.20 | 11.02 | 10.00 ± 1.52 |
| Dichloromethane | 276 | 13.48 ± 0.45c | 46.55 | 14.33 ± 0.33 |
| Ethyl acetate | 276 | 15.33 ± 0.82c | 39.21 | 13.33 ± 0.66 |
| Methanol | 276 | 21.33 ± 0.93 | 15.42 | 11.66 ± 1.20 |
| Pyrimethamine | 1.2 | 4.33 ± 0.66c | 81.82 | 25.66 ± 1.22c |
| Note: Values are expressed as mean ± SEM. Significant relative to control ap < 0.05; bp < 0.01; cp < 0.001. n = 5. | ||||
Antiplasmodial Effect of Ethanol Crude Extract and Fractions of T. on Established Infection
There were progressive dose-dependent reductions of parasitemia in all the extract-treated groups relative to negative control. These reductions were statistically significant relative to the negative control (p < 0.001; Figure 1). The methanol fraction had the highest activity with chemosuppressive effect of 77.48% on day 7, this was lower compared with that of the standard, chloroquine, 93.17%. The seed extract demonstrated significant (p < 0.05-0.001) prolongation in the mean survival time of the animals. The groups treated with methanol fraction had a longer mean survival time, 18.00 ± 1.15 days followed by those of crude extract (553 mg/kg) treated mice, 15.66 ± 1.45 days. These were less than that of the standard drug, chloroquine (29.83 ± 0.16 days; Figure 2).
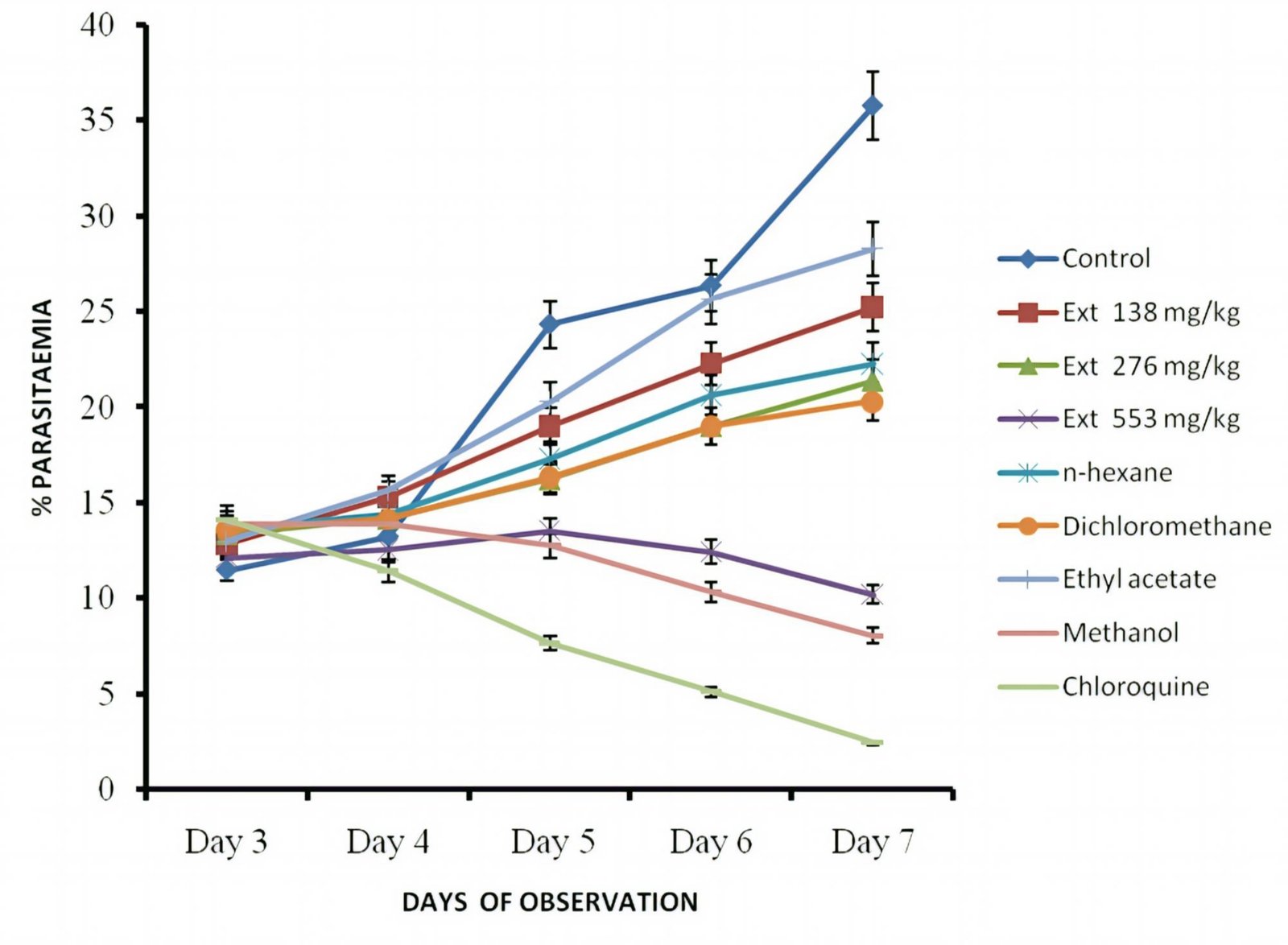
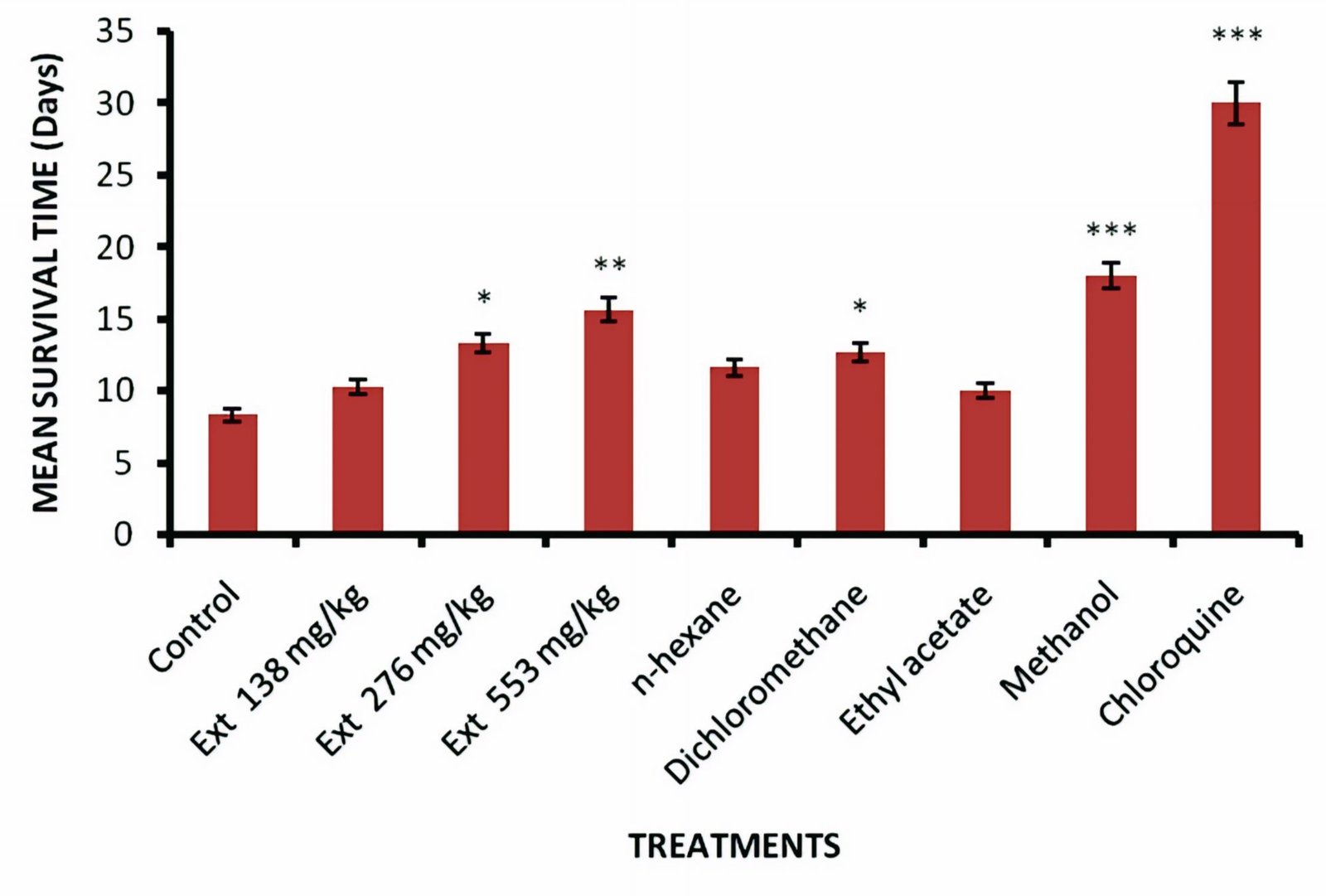
Effect of Seed Extract/Fractions on Rectal Temperatures of Infected Mice
Administration of the seed extract and fractions of T. occidentalis as well as chloroquine to Plasmodium berghei-infected mice in curative test did not cause any significant difference (p > 0.05) in the rectal temperatures of the treated mice when compared with that of controls (Table 3).
| Treatment | Dose (mg/kg) | Rectal Temperature (˚C) | |||
|---|---|---|---|---|---|
| D0 | D3 | D5 | D7 | ||
| Control | - | 36.96 ± 0.34 | 37.31 ± 0.11 | 37.94 ± 0.32 | 38.27 ± 0.16 |
| Extract | 138 | 36.85 ± 0.23 | 35.33 ± 0.20 | 35.73 ± 0.74 | 36.80 ± 0.35 |
| 276 | 36.98 ± 0.19 | 36.70 ± 0.50 | 37.66 ± 0.15 | 37.28 ± 0.15 | |
| 553 | 37.66 ± 0.50 | 36.51 ± 0.35 | 36.86 ± 0.24 | 36.95 ± 0.63 | |
| n-hexane | 276 | 36.46 ± 0.40 | 36.36 ± 0.14 | 36.60 ± 0.40 | 37.40 ± 0.27 |
| Dichloromethane | 276 | 36.85 ± 0.39 | 37.30 ± 0.17 | 36.68 ± 0.60 | 37.40 ± 0.04 |
| Ethyl acetate | 276 | 36.46 ± 0.60 | 36.86 ± 0.18 | 36.36 ± 0.43 | 37.62 ± 0.52 |
| Methanol | 276 | 36.48 ± 0.20 | 36.60 ± 0.08 | 36.40 ± 0.40 | 36.76 ± 0.44 |
| Chloroquine | 5 | 37.20 ± 0.40 | 36.83 ± 0.21 | 37.31 ± 0.25 | 37.65 ± 0.21 |
| Note: Values are expressed as mean ± SEM. | |||||
GC-MS Analysis
GC–MS analysis of the methanol fraction revealed the presence of pharmacologically active compounds such as hexanoic acid, 5, 6-dibromo-6-[p-chlorophenyl]-2, 4-dioxo-, ethyl ester; octanoic acid; palmitic acid, 9-hexadecenyl ester (Z)-; oleic acid, 2-(1-octadecenyloxy)ethyl ester (E)-; 9, 12-octadecadienoic acid (Z, Z)-, 2-[(trimethylsilyl)oxy]-1-[[(trimethylsilyl)oxy]methyl]ethyl ester; α-carotene, 7, 8-dihydro-3, 3', 19-trihydroxy-8-oxo-, triacetate, all-trans-; and eicosanoic acid, octadecyl ester, among others (Table 4; Figure 3).
| Peak | RT (min) | Compound | Peak Area% | Mol. Form | Mol. Mass (amu) |
|---|---|---|---|---|---|
| 1. | 4.733 | Acetic acid, 1, 1-dimethylethyl ester | 11.812 | C6H12O2 | 116.083 |
| 2. | 7.376 | Butanoic acid, 1, 1-dimethyethyl ester | 59.868 | C8H16O2 | 144.115 |
| 3. | 9.721 | Propanoic acid | 1.557 | C3H6O2 | 74.036 |
| 4. | 10.705 | Hexanoic acid, 5, 6-dibromo-6-[p-chlorophenyl]-2, 4-dioxo-, ethyl ester | 1.352 | C14H13Br2ClO4 | 437.886 |
| 5. | 14.983 | Octanoic acid | 1.412 | C8H16O2 | 144.115 |
| 6. | 15.175 | Palmitic acid, 9-hexadecenyl ester, (Z)- | 1.343 | C32H62O2 | 478.474 |
| 7. | 15.477 | Oleic acid, 2-(1-octadecenyloxy)ethyl ester, (E)- | 2.015 | C38H72O3 | 576.548 |
| 8. | 16.606 | α-Carotene, 7, 8-dihydro-3, 3', 19-trihydroxy-8-oxo-, triacetate, all-trans- | 1.534 | C46H62O7 | 726.449 |
| 9. | 19.959 | Eicosanoic acid | 1.891 | C20H40O2 | 312.302 |
| 10. | 20.745 | Eicosanoic acid, octadecyl ester | 1.364 | C38H76O2 | 564.584 |
| 11. | 21.391 | Octanoic acid, 2-hexenyl ester, (E) | 1.655 | C14H26O2 | 226.193 |
| 12. | 22.648 | 1, 3-Dioxolane-2-pentanoic acid, 2-methyl-, ethyl ester | 2.198 | C11H20O4 | 216.136 |
| 13. | 22.770 | 9, 12-Octadecadienal | 1.446 | C18H32O | 264.245 |
| 14. | 22.816 | 1-Dodecyne | 1.723 | C12H22 | 166.172 |
| 15. | 23.788 | 13-Tetradece-11-yn-1-ol | 3.115 | C14H24O | 208.182 |
| 16. | 25.104 | Cyclopropanecarboxylic acid, dodec-9-ynyl ester | 1.366 | C16H26O2 | 250.193 |
| Note: Compounds were presented above based on their retention time (minute). | |||||
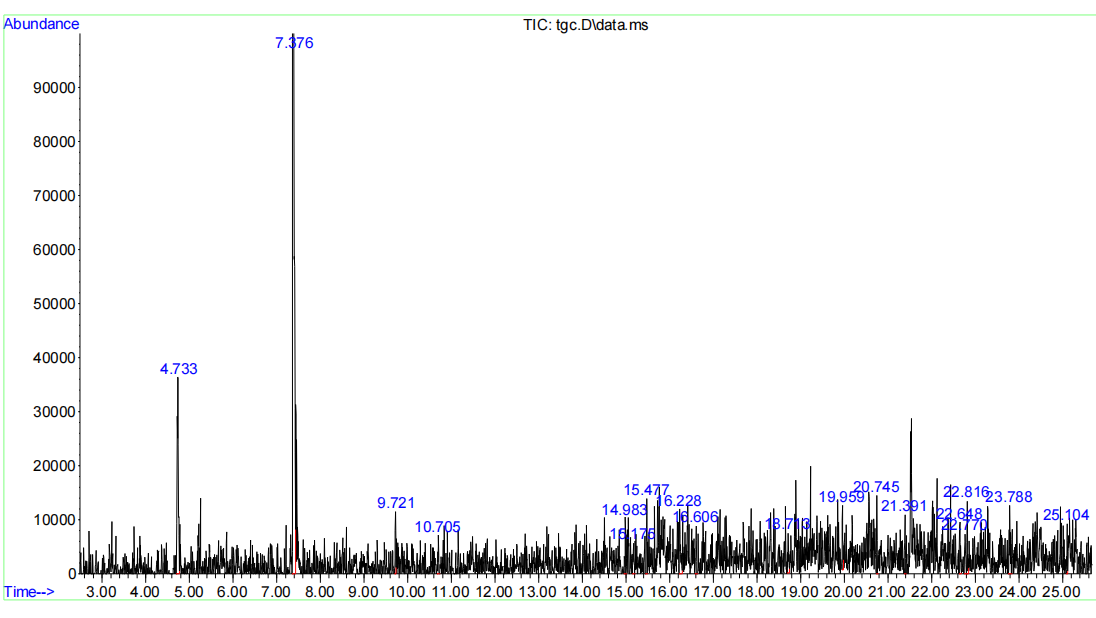
Discussion
The seeds of T. occidentalis are used locally as food and to treat various diseases such as malaria, diabetes, fever, inflammatory diseases and pain among others. This work was designed to confirm and authenticate the antimalarial potentials of the seed extract and solvent fractions of T. occidentalis in order to provide scientific basis for its usage in traditional medicine. The seed extract and solvent fractions of T. occidentalis were investigated for antimalarial activity against rodent malaria parasite, P. berghei infection in mice using standard in vivo models. The extract/fractions significantly reduced the parasitemia in suppressive, prophylactic and curative models in a dose-dependent fashion with methanol fraction exerting the highest suppression and curative activities, while DCM fraction exerted prominent prophylactic activity confirming the antimalarial potential of the seed extract.
The extract and fractions also prolonged the MST of the mice in different models suggesting that they were able to offer certain degree of protection to the mice. This activity could have resulted from plasmodicidal or plasmodistatic activity of the extracts. This further confirms and validates the use of the seed decoctions as malarial remedy. The slight variation in the activities of the extract and fractions suggests the involvement of immunostimulating activity which may be due to the phytochemical constituents in these extract/solvents. Tannins and fatty acids such as linoleic acids have been reported to possess immune-stimulating properties (29, 30).
The strong suppressive activities of the extract and fractions on the development of Plasmodium berghei parasitemia as observed during in the study, suggest that the extract affect the erythrocytic stages of the parasite (31). The inability of the extract and fractions to give complete protection to the infected mice in most cases could have resulted from low doses (138 - 553 mg/kg) used, short half-life/ duration of action of the extract/fractions due to rapid biotransformation processes and subsequent elimination (31). Thus, resulting in further development and multiplication of the parasites as well as short MST of the treated mice as observed in this study. However, these results corroborate previous reports on antimalarial activities of the leaf, seed and root extracts of T. occidentalis (32, 13) where significant suppressive, prophylactic and curative activities were recorded. These results validate the use of the seed of T. occidentalis decoctions as malarial remedy. In this investigation, in vitro study, isolation and characterisation of pure compounds from the active fraction could not be carried out to confirm the activities observed in the in vivo study.
Okokon et al. (12) reported the presence of polyunsaturated fatty acids such as hexadecanoic acid; 16-octadecenoic acid methyl ester; 9, 12-octadecadienoyl chloride (Z, Z); 9-octadecadienoic acid (Z)-, 2, 3-dihydroxypropyl ester; octadecanoic acid; hexadecanoic acid, 2, 3-bis [(trimethylsilyl)oxy]propyl ester; linoleic acid ethyl ester; and hexadecanoic acid methyl ester, as well as monoterpenes such as α-phellandrene; α-campholene aldehyde; terpinen-4-ol; trans-β-ocimene; and borneol in the seed extract. Also, in this study, GC–MS analysis of the active antiplasmodial methanol fraction revealed the presence of polyunsaturated fatty acid compounds such as hexanoic acid, 5, 6-dibromo-6-[p-chlorophenyl]-2, 4-dioxo-, ethyl ester; octanoic acid; palmitic acid, 9-hexadecenyl ester (Z)-; oleic acid, 2-(1-octadecenyloxy)ethyl ester (E)-; 9, 12-octadecadienoic acid (Z, Z)-, 2-[(trimethylsilyl)oxy]-1-[[(trimethylsilyl)oxy]methyl]ethyl ester; α-carotene, 7, 8-dihydro-3, 3', 19-trihydroxy-8-oxo-, triacetate, all-trans-; and eicosanoic acid, octadecyl ester, among others (Figure 4).
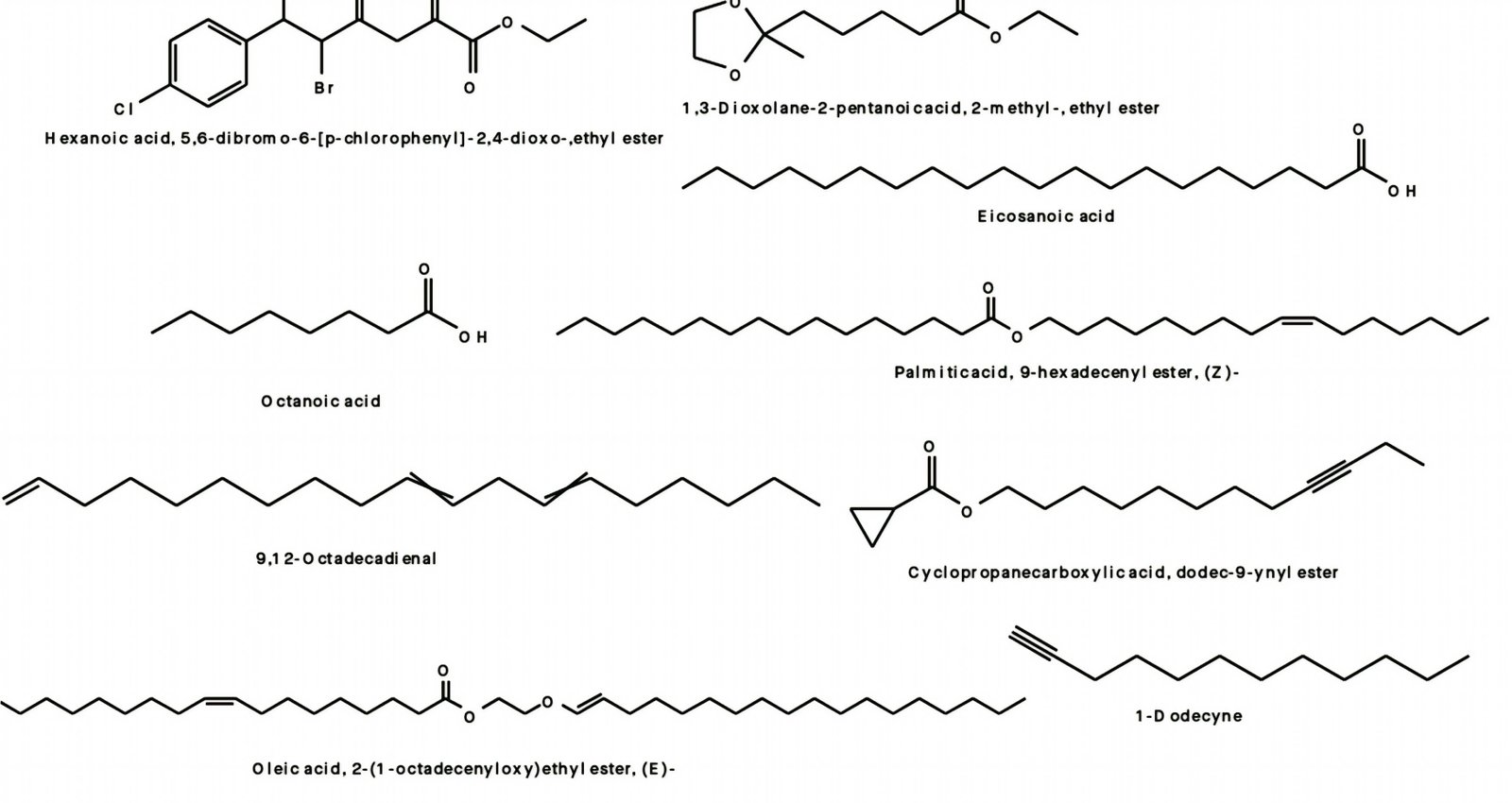
Previous studies of the seed extract and fractions by HPLC indicted flavonoids as the main antiplasmodial agents and revealed the presence of eleven flavonoids; kaempferol, catechin, epicatechin, anthocyanidin, flavonones, flavones, naringenin, rutin, naringin, and resveratol in the extract and fractions (20). Moreso, alkaloids such as spartein, ribalinidine lunamarin and ammodendrine were found to be present in high concentration in the butanol fraction of the seed extract (20). These compounds may be responsible for the observed activities of the extract and fractions. Some secondary metabolites of plants such as alkaloids, flavonoids and triterpenoids have been reported to have antiplasmodial properties (33-35). Polyunsaturated fatty acids (PUFAs) such as hexadecanoic acid, methyl ester, 9, 12-octadecadienoic acid methyl ester (linoleic acid), 9, 12, 15-octadecatrienoic acid, methyl ester (linoleic acid), and 9-octadecenoic acid as found in the active antiplasmodial fractions in this study have been implicated in antiplasmodial activity and this activity has been reported to increase with the degree of unsaturation (36-42). Also, flavonoids in the seed extract have been shown to possess significant antiplasmodial activity against chloroquine sensitive and resistant strains of P. falciparum (43-45). Antioxidant property of flavonoids has been suggested to be responsible for its antiplasmodial activity (46, 44), as elevated free radical levels are common features of malaria disease and are implicated in severe malaria complications. Scavenging of these free radicals could be one of the mechanisms of action of this extract. Other proposed mechanisms of antiplasmodial activity for flavonoids are chelation of nucleic acid base pairing of the parasite (47), modulation of host immunity to tackle disease and inhibition of plasmodial enoyl-ACP reductase (FAB I enzyme) – a key regulator of type II fatty synthases (FAS-II) in P. falciparum (48, 49) and binding to parasite’s serinethreonine kinase with high affinity thereby affecting its development (50). Furthermore, terpenes and their derivatives, monoterpenes and sesquiterpenes, have been implicated in antiplasmodial activity of many plants (34, 35). Monoterpenes such as limonene, trans-β-ocimene and α-pinene found in the dichloromethane fraction of the seed extract have been implicated in endoperoxidation leading to plasmocidal activity (51).
The results from the GCMS analysis of the active fraction revealed the presence of fatty acid esters and monoterpenes, compounds which show active antioxidation (52, 53, 29, 54). The above-mentioned compounds in the extract and active fractions, maybe responsible for the observed antiplasmodial activities in this study.
Fever is one of the cardinal symptoms of malaria especially in humans. However, P. berghei infection in mice is reported to be associated with hypothermia rather than pyrexia (55). Results of rectal temperatures of the infected mice in this study (curative test), showed that there was no significant difference between the mean temperature values of both the treated and untreated infected mice before and after treatment, suggesting that the mice were hypothermic. This hypothermia in mice may have resulted from the serious physiological effects of the malaria parasite on the host, leading to body heat loss and ultimately death of mice (56). Consequently, carbohydrate, lipid, and protein metabolisms of the host are affected negatively by malaria parasite (57, 58). Decreases in metabolic rates of the P. berghei-infected mice have been correlated with decreased body temperatures of mice (59). The extract/fractions however, were unable to attenuate these processes and hence the resultant hypothermia.
Conclusion
The findings of this study further suggest that seed extract and fractions of T. occidentalis possess antimalarial potentials which is due to the activities of its phytochemical constituents especially the polyunsaturated fatty acids among others. This confirms and authenticates its use as malarial remedy in folkloric medicine.
Declarations
Acknowledgment
The authors are grateful to staff of Animal House, Pharmacology and Toxicology Department of University of Uyo for providing technical assistance.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The data generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Statement
All animal experiments were approved by the Animal Research Ethics Committee of the University of Uyo and conducted in accordance with relevant guidelines and regulations.
Funding Information
We did not receive any external funding for this research.
References
- World Health Organisation. World Malaria Report: Addressing inequity in the global malaria response. Geneva: World Health Organization; 2024. Licence: CC BY-NC-SA 3.0 IGO. https:// www. who.int/teams/global-malariaprogramme/reports/worldmalaria-report; 2024.
- Venkatesan P. World Malaria Report 2024. Lancet Microbe.2025; 6:101073.
- Okokon JE,Nkemnele AB,Mohanakrishnan D. Antimalarial, antiplasmodial and analgesic activities of root extract of Alchornea laxiflora, Pharm Biol.2017;55(1):1022-1031.
- Okokon JE, Antia BS, Azare B, Okokon PJ. Antimalarial and antiplasmodial activities of ethanol root extract of Zea mays. Avicenna J Phytomed. 2017;7(3):275-284.
- Okokon JE, Mohanakrishnan D, Dinkar S, Okokon PJ. Antimalarial activities of Zea mays leaf extract in ethanol and selected solvent fractions. J Herbs, Spices Med Plants.2017;23(4): 334 –346.
- Okokon JE, Davis K, Azare B, Okokon P. Analgesic and antimalarial activities of ethanol root extract of Panicum maximum. AfrJ PharmacolTherap.2016; 5(3): 128 -135.
- Okokon JE, Opara KN, Udobang JA, Bankhede H. In vivo antimalarial and antipyretic activities of leaf extract of Ananas comosus.Trop JNatProd Res.2019;3(7):240-245.
- William NB, Bassey AL, Offiong AU, Otuekong E, Okokon JE. In vivo antimalarial activities of leaf extract and fractions ofSetaria megaphylla (Wild.) Loes. in Plasmodium berghei infected mice. Trop J Nat Prod Res.2022;6(9):1504-1510.
- Eseyin OA, Igboasoiyi AC, Oforah E, Nkop N, Agboke A. Hypoglycaemic activity of Telfairia occidentalis in rats. JPharmBiores. 2005;12;2(1):36-42.
- Usunomena U, Okpiabhele A. Telfairia occidentalis Hook f. mitigates Carbon Tetrachloride induced Nephrotoxicity in Rat. J Res Appl Basic Med Sci.2023; 9 (3):130-137.
- Eseyin OA, Ebong P, EkpoA, IgboasoiyiA, Oforah E. Hypoglycemic effect of the seed extract of Telfairia occidentalis in rat. Pak J Biol Sci.2007;10(3), 498–501.
- Okokon, J. E., Antia, B. S., Dar, A., and Choudhary, M. I. Immunomodulatory, anticancer and antiinflammatory activities of Telfairia occidentalis seed extract and fractions. Int J Food Nutr Safety. 2012;2(2):72–85.
- OkokonJE, Ekpo AJ, Eseyin OA. Evaluation of in vivo antimalarial activities of ethanolic leaf and seed extractsof Telfairia occidentalis. J Med Food. 2009;12(3):649–653.
- Osukoya OA, Adegbenro D, Onikanni SA, Ojo OA, Onasanya A. Antinociceptive and antioxidant activities of the methanolic extract of Telfairia occidentalis Seeds. Anc Sci Life. 2016; 36(2):98-103.
- Okokon, J. E., Dar, A., and Choudhary, M. I. Chemical constituents and analgesic activity of Telfairia occidentalis. Phytopharmacol. 2012;3(2):359–366.
- Magnus SP, AnagbosoMO, Johnny II, Ise U P, OkokonJE. Evaluation of genotoxic and cytotoxic activities of leaf and seed extracts of Telfairia occidentalis. J Comple Alt Med Res. 2024; 25(3):7–16.
- Enin GN, Okokon JE, Odokwo BO, Antia BS. Preliminary phytochemical screening and in vivo Inhibitory study of Telfaira occidentalis Hook f. seeds extract on alpha amylase and alpha glucosidase of rats. J Sci Tech Res.2023;5(4):26-35.
- Umoh UF, Ubengama EE, Udofia EU, Obasi OI, Okonna UK, Umanah ES, Etefia JE, Okokon JE. HPLC characterization and anti-ulcer effects of methanol seed extract and fractionated components of Telfairia occidentalis in rodents. Nig J Pharm Appl Sci Res. 2025: 14(2):111-117.
- Fabian UA, Anagboso MO, Samuel AE, Okokon JE. Effect of seed extract and fractions of Telfairia occidentalis on liver and kidney functions and histologies of rats with testosterone-induced benign prostatic hyperplasia in rats. J Compl Alt Med Res.2025; 26(6): 1 -18.
- Ebong AS, Eseyin OA, Etim EI, Okokon JE. Telfairia occidentalis potentiates antiplasmodial activity of artemisinin and amodiaquine combination therapy. Anti-Infective Agents.2020;18(2): 152-159.
- AtanuFO, IdihFM, NwonumaCO, HettaHF, AlameryS, BatihaGE. Evaluation of antimalarial potential of extracts from Alstonia boonei and Carica papaya in Plasmodium berghei-infected mice. Evidence-Based Compl Alt Med Article ID 2599191, 2021;11.
- Peters W, Robinson BL. The chemotherapy of rodent malaria. Studies on pyronaridine and other Mannich base antimalarials. Ann Trop Med Parasitol.1992;86, 455–465.
- National Institutes of Health (US). Division of Public Information, National Institutes of Health (US). Editorial Operations Branch. NIH almanac. US Department of Health, Education and Welfare, Public Health Service, National Institutes of Health; 1998.
- Knight DJ, Peters W. The antimalarial activity of N-benzyloxydihydrotriazines. I. The activity of clociguanil (BRL 50216) against rodent malaria, and studies on its mode of action. Ann Trop Med Parasitol.1980; 74:393–404.
- Okokon JE, Nwafor PA.Antiplasmodial activity of root extract and fractions ofCroton zambesicus. J Ethnopharm.2010; 121:74-78.
- Peters W. Drug resistance in Plasmodium berghei Vincke and Lips, I. Chloroquine resistance. Expl Parasitol.1965;17:80–89.
- Ryley JF, Peters W. The antimalarial activity of some quinolone esters. Ann Trop Med Parasitol.1970; 84:209–222.
- Okokon JE, Davies K, Lekara J, Iwara K, Thomas P, Li W-W. Antidiabetic and hypolipidemic activities of solvents fractions of Setaria megaphylla and its phytochemical characterisation. Phytomed Plus. 2022:100182.
- KhanSL, Siddiqui FA. Beta-Sitosterol: As Immunostimulant, Antioxidant and Inhibitor of SARS-CoV-2 Spike Glycoprotein. Arch Pharmacol Therapeut 2020; 2(1):12-16.
- Kumaradevan G, Damodaran R, Mani P, Dineshkumar G, Jayaseelan T. Phytochemical screening and GC–MS analysis of bioactive components of ethanol leaves extract of Clerodendrum phlomidis (L.). Am J Biol Pharm Res.2015; 2:142–148.
- Waako PJ,Gumede B.,SmithP,Folb PI. The in vitro and in vivo Antimalarial activity of Cardiospermum halicacabum L. and Momordica foetida Schumch. et Thonn. J Ethnopharmacol.2005;99(1):137-143.
- Okokon JE,Ekpo AJ,Eseyin OA. Antiplasmodial activity of ethanolic root extract of Telfairia occidentalis.Research Journal of Parasitology.2007; 2(2):94-98.
- Kirby GC.Plants as source of Anti-malarial Drugs. Tropical Doctor. 1989;27(1):7-13.
- Philipson JD, Wright CW. Antiprotozoal compounds from plants sources. Planta Medica.1991;57:553–559.
- Christensen SB, KharazmiA. Antimalarial natural products. Isolation, characterization and biological properties. In: Tringali C, editor. Bioactive compounds from natural sources: isolation, characterization and biological properties. London: Taylor & Francis; p. 379–432,2001.
- Kumaratilake LM, Robinson BS, Ferrante A, Poulos A. Antimalarial properties of n-3 and n-6 polyunsaturated fatty acids: in vitro effects on Plasmodium falciparum and in vivo effects on P. berghei. J Clin Investig.1992;89:961–967.
- Krugliak M, Deharo E, Shalmiev G, Sauvain M, Moretti C, GinsburgH. Antimalarial effects of C18 fatty acids on Plasmodium falciparum in culture and on Plasmodium vinckei petteri and Plasmodium yoelii nigeriensis in vivo. Expl Parasitol.1995; 81:97–105.
- Suksamrarn A, BuapromM, UdtipS, Nuntawong N,Haritakun, R, Kanokmedhakul S. Antimycobacterial and antiplasmodial unsaturated carboxylic acid from the twigs of Scleropyrum wallichianum. Chem. Pharmaceut. Bull. 2005; 53:1327–1329.
- AttiouaB, Weniger B, Chabert P. Antiplasmodial activity of constituents isolated from Croton lobatus. Pharm Biol. 2007; 45:263–266.
- MelaririP, CampbellW, EtusimP, SmithP. Antiplasmodial properties and bioassay-guided fractionation of ethyl acetate extracts from Carica papaya leaves. JParasitolRes, 2011; Article ID 104954, 7pages.
- MelaririP, Campbell W, Etusim P, Smith P. In vitro and in vivo antimalarial activity of linolenic and linoleic acids and their methyl esters. Advanced Studies in Biology.2012; 4:333–349.
- Enyiekere VJ, Asanga EE, Okokon JE, Ekeleme CM,Anagboso MO, Ise UP. Fatty acid esters and acyclic monoterpenoid from Justicia insularis leaf fractions attenuated malaria pathogenesis through docking with PFSHMT and PFEMP-1 proteins. Nat Prod Comm. 2024;19(7):1-14.
- Attioua B, Lagnikab L, Yeoc D, AntheaumedC, Kaisere M, Wenigerf B, LobsteinfA, Vonthron-S_en_echeauf C. In vitro antiplasmodial and antileishmanial activities of flavonoids from Anogeissus leiocarpus (Combretaceae). Int J Pharm Sci Rev Res, 2011; 11:1–6.
- GaneshD, FuehrerH, StarzengruberP, SwobodaP, Khan WA, ReismannJA, MuellerMS, Chiba P, Noedl H. Antiplasmodial activity of flavonol quercetin and its analogues in Plasmodium falciparum: evidence from clinical isolates in Bangladesh and standardized parasite clones. Parasitol Res.2012;110:2289–2295.
- Ezenyi IC, Salawu, OA, Kulkarni R Emeje M. Antiplasmodial activity aided isolation and identification of quercetin-40-methyl ether in Chromolaena odorata leaf fraction with high activity against chloroquine-resistant Plasmodium falciparum. Parasitol Res.2014;113:4415–4422.
- Cimanga RK, Tona GL, Kambu OK, Mesia GK, Muyembe JJ, Apers S. (2009). Antimalarial, antiamoebic and cytotoxic activities of some extracts and isolated constituents from the leaves of Morinda morindoides (Baker) Milne- Redh. (Rubiaceae).Rec Progr Med Plants. 2009;5:225–242.
- Lui KC, Yang SC, Roberts MF. Antimalarial activity of Artemisia annua flavonoids from whole plants and cell cultures. Plants Cell.1992; II: 637–40.
- Kirmizibekmez H, Calis I, Perozzo R, Brun R, Donmez AA, Linden A, Ruedi P, Tasdemir D. Inhibiting activities of the secondary metabolites of Phlomis brunneogaleata against parasitic protozoa and plasmodial enoyl-ACP reductase, a crucial enzyme in fatty acid biosynthesis. Planta Med.2004;70:711.
- Teffo LS, Aderogba MA, Eloff JN. Antibacterial and antioxidant activities of four kaempferol methyl ethers isolated from Dodonaea viscose Jacq. Var. angustifolia leaf extracts. South Afr J Botany. 2010;76:25–29.
- Ferreira JF, Luthria DL, Sasaki T, Heyerick A. Flavonoids from Artemisia annua L. as antioxidants and their potential synergism with artemisinin against malaria and cancer. Molecules. 2010; 15:3135–3170.
- Hatzakis E, Opesenica I, Solaja BA, Stratakis M. Synthesis of novel polar derivatives of the antimalarial endoperoxides. Arch Org Chem. 2007; VIII: 124–125.
- Kohno Y, EgawaY, Itoh S, Nagaoka S, TakahashiM, MukaiK. Kinetic study of quenching reaction of singlet oxygen and scavenging reaction of free radical by squalene in n-butanol. Biochimica et Biophysica Acta. 1995;1256: 52–56.
- Ponnamma SU, Manjunath K.GC-MS analysis of phytocomponents in the methanolic extract of Justicia wyaadensis (NEES) T. Anders. Int J Pharm Biol Sci. 2012;3:570-576.
- MareiGI, Rabea EI, Badawy ME. In vitro antimicrobial and antioxidant activities of monoterpenes against some food-borne pathogens. J Plant Protection and Pathology, Mansoura University, 2019;10(1): 87-94.
- Belay WY, Gurmu AE, Wubneh ZB. Antimalarial activity of stem bark of Periploca linearifolia during early and established Plasmodium infection in mice. Evidence-Based Compl Altern Med. Volume 2018, Article ID 4169397, 7 pages
- Jennings G, Elia M (1987). “Effect of E. coli endotoxin on temperature, oxygen consumption and brown adipose tissue thermogenesis in rats and mice,” Bioscience Reports.1987;7(6):517–523.
- Siddiqui WA, Trager W. Free amino-acids of blood plasma and erythrocytes of normal ducks and ducks infected with malarial parasite, Plasmodium lophurae,” Nature.1967; 214(5092):1046-1047.
- Angus MG Fletcher KA, Maegraith BG. Studies on the lipids of Plasmodium knowlesi-infected rhesus monkeys (Macaca mulatto) I: Changes in serum lipids,” Annals Trop Med Parasitol.1971; 65(2): 135–154.
- Briand H, PeterW. Acute malaria: effects of P. berghei on the metabolic rate of mice,” The Ohio J Sci.1977;77–189.