REVIEW
Harnessing Plant Secondary Metabolites for Vaccine Development in Africa: Current Insights and Future Prospects
Sciences of Phytochemistry|Vol. 5, Issue 1, pp. 64-80 (2026)
Received
Dec 7, 2025Revised
Feb 9, 2026Accepted
Mar 3, 2026Published
Apr 20, 2026
Abstract
Introduction
Secondary metabolites are biological compounds that are not directly involved in an organism’s intrinsic functions but perform essential ecological and relational roles. The absence of secondary metabolites does not cause immediate death but can lead to long‑term impairment of survivability, fecundity, or other fitness attributes (1). These compounds are often restricted to a narrow set of species within a phylogenetic group and are produced by plants (plant secondary metabolites, PSMs) as well as by microorganisms such as bacteria, fungi, algae, and actinobacteria (2). This review is explicitly focused on PSM evidence that bears on vaccine translation namely antigen‑specific adjuvant activity, formulation or excipient utility, delivery strategies, and operational feasibility for deployment in African settings. To preserve conceptual focus, class‑level descriptions are presented only where they directly inform mechanisms or formulation properties relevant to adjuvant design, antigen stability, or delivery; fuller taxonomic and chemical background is provided in Supplemental Table 1. Table 1 delineates representative PSM candidates positioned across the clinical development continuum, encompassing early-stage preclinical assessments through to established, licensed adjuvant systems. A dedicated section on definitions “definition of terms” systematically clarifies key terminology and explicates the evidence‑tagging convention applied throughout the manuscript.
Among the major classes of PSMs, alkaloids constitute a prominent group of nitrogen‑containing compounds with considerable biogenetic and structural diversity. Alkaloids are commonly classified by biogenetic origin rather than solely by structural features; examples that have been retained in this review for their reported biological activity include berberine, solasodine (family Solanum), ricinine (genus Ricinus), and tomatidine (from Lycopersicon pimpinellifolium) (3). Discussion of specific alkaloids is limited to instances in which mechanistic or experimental evidence links the compound to immunomodulation, adjuvant‑like activity, or formulation benefits. Flavonoids are ubiquitous phenolic compounds classified into flavones, flavonols, isoflavones, and anthocyanins; many flavonoids have reported antimicrobial and antioxidant properties. Where relevant to vaccine science, flavonoids such as quercetin and catechin are considered only when studies directly assess immune modulation or antigen‑specific outcomes (4). Terpenoids are a large class of isoprene‑derived natural products, classified by carbon number into hemiterpenes (C5), monoterpenes (C10), sesquiterpenes (C15), diterpenes (C20), sesterterpenes (C25), triterpenes (C30), carotenoids (C40), and polyisoprenoids (Cn, n > 40) (5). Saponins are glycosides with triterpenoid or steroidal aglycones and commonly exhibit surfactant properties; selected saponins (for example, Quillaja‑derived QS-21) have documented adjuvant activity when co‑formulated with antigens and are therefore discussed as vaccine‑relevant examples (6). Other saponins are considered only where comparable evidence exists. Polysaccharides and lectins are additional PSM classes of interest. Polysaccharides such as arabinogalactan and certain β‑glucans have been reported to modulate innate immune pathways, and lectins are carbohydrate‑binding proteins with diverse biological activities. Statements about lectins (for example, Concanavalin A) are confined to reported immunostimulatory mechanisms and explicitly note translational limitations, since potent in vitro mitogens are not necessarily practical or safe vaccine adjuvants without further evidence (7). Although numerous in vitro and animal studies report PSM‑associated immune effects, systematic evidence from controlled co‑formulation studies demonstrating antigen‑specific adjuvant activity is limited, and adequately powered clinical trials or operational evaluations relevant to deployment are scarce. Vaccine development in African settings is further constrained by cold‑chain requirements, high costs, limited local manufacturing and quality‑control capacity, and the need for clinical evaluation in representative populations. These operational and scientific considerations are therefore incorporated into the criteria used to appraise the evidence.
This review summarizes mechanistic, preclinical, and formulation evidence for PSMs insofar as such evidence bears directly on vaccine translation (antigen‑specific outcomes, formulation stability, delivery, and thermostability). The objectives are to (i) identify PSMs and characterized extracts with the strongest mechanistic and preclinical evidence for adjuvant or formulation utility, (ii) evaluate formulation and stability data relevant to field conditions, and (iii) delineate the experimental, regulatory, and implementation steps required to advance promising candidates toward clinical evaluation in target populations, with explicit attention to Africa‑relevant constraints and priorities.
Definition of terms
Adjuvant: A substance co‑formulated or co‑administered with a vaccine antigen to enhance or direct antigen‑specific immune responses; adjuvant activity is demonstrated by increased antigen‑specific immunogenicity (8).
Immunostimulant: A compound that boosts immune function, either in a targeted (antigen‑specific) or broad, non‑specific way. They range from vaccine adjuvant molecules and bacterial products to synthetic drugs and herbal extracts, and are used to prevent infections, support cancer therapy, and treat immune deficiencies (9).
Nutraceutical: A food‑derived product consumed primarily for health promotion or disease prevention (dietary supplements, fortified foods). Nutraceuticals are regulated differently from vaccines and therapeutics and should be described as such; reported immunomodulatory effects from nutraceuticals should be labelled as nutraceutical (observational or experimental) and not conflated with formal adjuvant activity (10).
Therapeutic agent: A compound intended to treat or cure disease through a defined pharmacological mechanism and evaluated by therapeutic endpoints in clinical trials (distinct from prophylactic vaccine adjuvants). Therapeutic claims require evidence from appropriately designed clinical studies (11).
Usage Note
Throughout the manuscript we append an evidence tag at first mention (for example, “adjuvant (preclinical)”, “immunostimulant (in vitro)”, “nutraceutical (observational human data)”) and reserve the term “adjuvant” for studies that co‑administer an antigen and report antigen‑specific outcomes.
Methods
Review Type and Scope
This manuscript is a narrative, evidence‑focused review that synthesizes mechanistic, preclinical, and formulation evidence on plant secondary metabolites (PSMs) insofar as that evidence bears directly on vaccine translation (antigen‑specific outcomes, adjuvant‑like activity, formulation performance, and operational feasibility). The review is purposive: the objective was to identify and interpret studies that directly inform whether and how PSMs might function as adjuvants, immunostimulants with vaccine relevance, or formulation/excipient components that improve antigen stability or delivery.
Literature Identification
Targeted literature searches were performed in major bibliographic databases and supplemented by hand searches of reference lists and selected grey literature. Primary sources consulted included PubMed/MEDLINE, Web of Science, Scopus, and Google Scholar. Search terms combined plant‑derived keywords (for example, phytochemical, plant secondary metabolite, alkaloid, flavonoid, saponin, terpenoid, lectin, polysaccharide) with vaccine‑relevant terms (for example, adjuvant, vaccine formulation, antigen co‑formulation, thermostability, immunostimulant). Searches were iterative and purposive: initial searches identified exemplar studies and reviews, which guided follow‑up searches for mechanistic or formulation evidence.
Inclusion and Exclusion Criteria
Inclusion Criteria
Articles published between 2000 and 2025 was prioritized to capture contemporary vaccinology and formulation science while excluding largely historical chemical reports that predate modern immunological methods. Older, seminal works were considered only when directly relevant to mechanistic interpretation. Primary research (in vitro, animal, human) that met one or more of the following: (a) co‑formulation or co‑administration of a defined antigen with a characterized PSM reporting antigen‑specific outcomes (antigen‑specific IgG/IgA, neutralization, T‑cell phenotype, or protection in challenge models); (b) mechanistic studies that identify pathways plausibly mediating adjuvant activity (for example, innate receptor engagement, antigen uptake/presentation); or (c) formulation and stability studies that assess antigen integrity, thermostability, or delivery performance under conditions relevant to deployment.
Exclusion Criteria
Non‑primary reports lacking sufficient methodological detail (editorials, opinion pieces) except were used for regulatory context; chemical or phytochemical surveys without immunological or formulation data; studies reporting only general toxicity or unrelated pharmacology. Grey‑literature items (conference abstracts, technical reports) were consulted selectively for formulation or operational data and are explicitly identified and qualified in the text.
Study Selection and Data Handling
Records identified through database searches and reference checks were screened for relevance by the authors. Full texts of potentially relevant items were retrieved and assessed against the inclusion/exclusion criteria above. Key information from included primary studies was recorded in a structured internal table: citation, study design (in vitro, animal, human), compound identity and characterization, extract standardization (where reported), antigen identity and co‑formulation details (route, dose, formulation matrix), primary endpoints and time points, formulation/stability metrics, principal findings, and notable methodological limitations. Data extraction was performed manually and iteratively; items raising uncertainty about inclusion or interpretation were discussed among the authors until consensus was reached.
Synthesis and Interpretation
Synthesis was narrative and organized by compound class (alkaloids, flavonoids, terpenoids, saponins, polysaccharides/lectins), by evidence domain (mechanistic, antigen‑specific immunogenicity/protection, formulation/stability), and by model (in vitro, animal, human). Where multiple studies addressed the same compound–antigen pairing with comparable endpoints, results were compared and described together. Heterogeneity in antigens, formulations, and endpoints precluded formal meta‑analysis; quantitative pooling was not attempted. Interpretive emphasis was placed on studies that reported antigen‑specific outcomes and on mechanistic data that plausibly explain observed effects.
Consideration of Study Quality and Conflicting Evidence
A formal, uniform risk-of-bias scoring system was not applied across all study types in this narrative review; instead, study quality and relevance were considered qualitatively, with controlled co-formulation studies featuring clear compound characterization and antigen-specific endpoints being given greater interpretive weight than isolated in vitro reports or poorly characterized extracts. This nuanced prioritization ensured that the synthesis remained rooted in methodologically robust evidence while still acknowledging the preliminary insights offered by smaller-scale mechanistic studies. Conflicting findings are presented alongside a detailed discussion of likely explanatory factors—such as differences in compound purity or standardization, antigen identity, dose/regimen, route of administration, model species, or endpoint selection—to provide a transparent view of the current scientific landscape. By dissecting these variables, the review highlights how subtle technical discrepancies often underpin broader clinical contradictions. Where discordant results could not be reconciled due to significant data gaps or methodological heterogeneity, conclusions were framed conservatively and specific experimental work was recommended to address these remaining ambiguities and strengthen the weight of evidence in future investigations.asd
Grey Literature, Transparency, and Limitations
Selected grey literature (conference abstracts, technical reports, regulatory guidance) was consulted to capture formulation and operational data that may not appear in indexed journals; such items were used primarily to inform practical considerations and are explicitly identified and qualified in the text. The purposive narrative approach limits reproducibility and may introduce selection bias; these limitations are acknowledged and discussed in the manuscript. To aid reader appraisal, the Methods list the databases and general search terms used, the inclusion rationale, the timeline applied, and the criteria used to distinguish adjuvant‑relevant evidence from general immunomodulatory findings. Key decisions about study inclusion and the basis for interpretive weighting are described in the limitations section so readers can assess how evidence informed conclusions.
Results and Discussion
Plant Secondary Metabolites as Vaccine Adjuvants and Immunomodulators
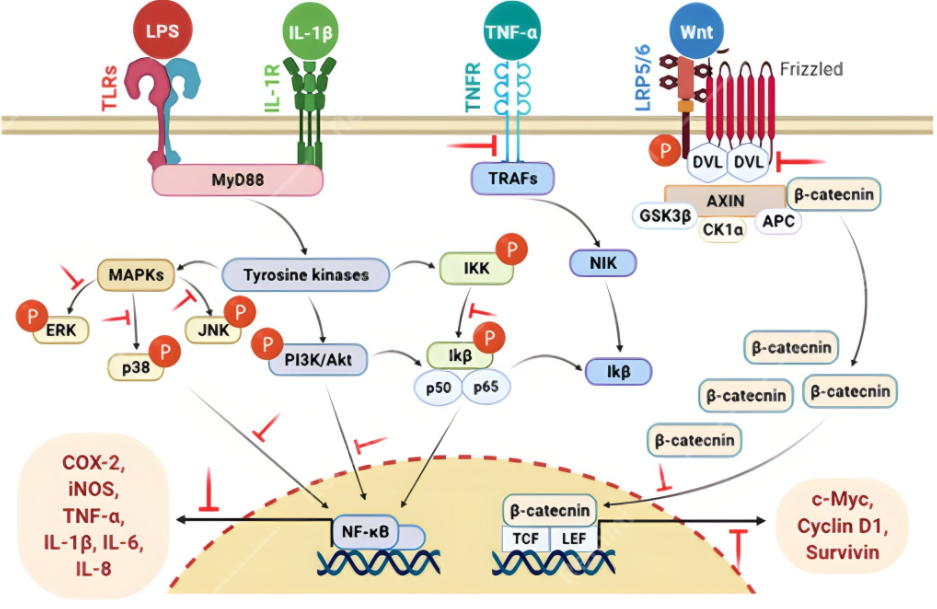
For clarity, mechanisms are classified into three categories: (a) adjuvant‑like activity, (b) direct antiviral activity, and (c) anti‑inflammatory/modulatory effects. Each cited study is labelled accordingly in tables and discussion. Immunomodulators are agents that can regulate an organism’s immune system; those that enhance or activate the immune response are known as immunostimulants, while those that lessen or inhibit the immune response are known as immunosuppressants (9). These agents are most commonly used in various viral diseases (HIV/AIDS, SARS-Cov-2/COVID-19), autoimmune diseases, allergic reactions, inflammatory conditions, and cancer. There are diverse groups of medicinal plants and plant-based products that have potent immunomodulating activities, and these compounds have been found to modulate immune functions through diverse mechanisms, including MAPK, PI3K/Akt, NF-κB, and Wnt signaling pathways (12). These findings are derived from in vitro systems or animal models and should be interpreted as hypothesis‑generating rather than evidence of human vaccine efficacy. Translational limitations include species differences, dose scaling, and absence of antigen‑specific clinical endpoints. Figure 1 illustrates the principal cellular and molecular pathways through which PSMs exert immunomodulatory effects. It highlights activation of MAPK, PI3K/Akt, NF‑κB, and related cascades, providing a visual overview of how PSMs influence antigen presentation and immune cell function.
Advances in the development of saponins as human vaccine adjuvants have occurred in the last decade, leading to the development of a new generation of vaccines against cancer and infectious diseases at various phases of clinical trials. Many Quillaja saponins (from the plant Quillaja saponaria) are used as an adjuvant (13-15). Saponin QS-21 promotes the immune system without cytotoxic effects and is currently being developed as an auxiliary AIDS vaccine (16), and another is the saponin aglycone called Quillaic acid (17). Curcumin has shown potential as a complementary supplementation during the vaccination period, as it can increase antibodies produced post-vaccination (18). Curcumin intake confers anti-inflammatory activity and may be a promising prophylactic nutraceutical strategy for COVID-19 (19). Limited in vitro and computational studies suggest Withania somnifera may interact with SARS‑CoV‑2 targets, but current evidence does not support clinical equivalence to hydroxychloroquine. These findings should be considered preliminary and hypothesis‑generating, requiring controlled pharmacological and clinical studies before therapeutic claims can be made. W. somnifera can bind to the key targets (Spike protein, ACE-2, RdRp, 3CLpro, and PLpro) of SARS-CoV-2, indicating it may be a good potential therapeutic candidate for COVID-19 treatment (20, 21).
African Medicinal Plants with Potential for Vaccine Development
Many traditional medicinal plants have been shown to possess immuno-potentiating activities, validating their use in folk medicine (22, 23). Moringa oleifera, as a new plant food, has received attention from researchers due to its rich variety of nutrients. It has been reported that M. oleifera leaf polysaccharide (MOLP) possesses outstanding bioactive functions, including immunoregulatory, glycolipid-lowering, and antioxidant activities. Recently, a polysaccharide (MOLP-PE) was verified to possess acceptable homogeneity and high proportions of arabinose (Ara) and galactose (Gal), which indicated that MOLP-PE was a type of arabinogalactan (AG) which has shown stronger ability in stimulating proliferation, phagocytosis and cytokines release of macrophages and bound with Toll-like receptor (TLR) closer via more binding sites (24).
Artemisia annua, a well-known traditional medicinal plant, has been extensively used as an antimalarial and anticancer agent. Recently, the in vitro and in vivo efficacy of artemisinin (a sesquiterpene trioxane lactone) against hepatocellular carcinoma and experimental visceral leishmaniasis has been reported. Flavonoids of A. annua have been linked to beneficial immunomodulatory activities in subjects affected by parasitic and chronic diseases (23, 25-28).
Aspilia africanahas been used by many African communities in the treatment of inflammatory conditions as well as osteoporosis, stomach ache, diarrhea, measles, malaria, tuberculosis, cough, gastric ulcers, sores, diabetes, rheumatic pains, bee, scorpion, and wasp stings, ear infections, febrile headaches, and gonorrhea. It is used as a contraceptive and for wound healing. The plant, often known as the hemorrhage plant or wild sunflower, is referred to by various local names by different communities, such as Makayi in Luganda (Uganda), Orangila in Igbo (Nigeria), Nyana in Kissi (Sierra Leone), Fofo in Akan-akyem (Ghana), Mbnaso in Kpe (Cameroon), Soumadibrouin among the Malinke (Côte d’Ivoire), and Winnih in Mano (Liberia) (28-30). Phytochemical analysis indicates that the species is rich in a broad range of PSMs, explaining its efficacy in treating various African communities' inflammation and other health conditions. The therapeutic PSMs in A. africana include flavonoids, alkaloids, tannins, saponins, terpenoids, sterols, phenolic compounds, and glycosides. Essential oils from the plant leaves are rich in monoterpenes, sesquiterpenes, 5-pinene, and germacrene, which are also therapeutic (30). The broad range of antimicrobial and biological activities, including anti-inflammatory, haemostatic, oxytocic, gastroprotective, antiulcer, wound healing, anticancer, antihypertensive, and antidiabetic potentials, could be attributed to these therapeutic PSMs (31). Typical PSMs in A. africana that exhibit great anti-inflammatory potential include monoterpenes, e.g., carene, terpenes, e.g., 5-pinene, and sesquiterpenes, e.g., 6-caryophyllene and tannins (32, 33). Supplemental Table 1 summarizes selected African medicinal plants, their key secondary metabolites, and reported immunological mechanisms relevant to vaccine development. It also includes a critical appraisal of study quality and reproducibility to guide interpretation of translational potential. Supplemental Table 1 is available in the supplementary data.
While Supplemental Table 1 summarizes African medicinal plants and their mechanistic immunological properties, several PSMs have progressed further along the translational pipeline. Table 1 highlights representative PSMs that are at different stages of clinical development, ranging from preclinical evaluation to licensed adjuvant systems. This overview provides context for understanding which compounds have achieved regulatory‑grade validation and which remain at the experimental stage, thereby linking mechanistic promise to translational readiness.
| PSM / class | Source | Vaccine | Development phase | Reference |
|---|---|---|---|---|
| QS‑21 (AS01, AS02, ALFQ) | Triterpenoid saponin, Q. saponaria bark | Shingrix (zoster), RTS, S/AS01 (malaria), many prophylactic/therapeutic vaccines | Licensed (post‑Phase III) | (40-42) |
| Matrix‑M | Q. saponaria saponin fractions in ISCOM‑like nanoparticles | NVX‑CoV2373 (Novavax COVID‑19), influenza, malaria, others | Licensed/Phase III | (43-45) |
| Quil‑A / other Q. saponaria bark mixtures | Crude triterpenoid saponin mixtures | Early human and veterinary subunit vaccines | Phase I-II | (42-46) |
| Quillaja brasiliensis leaf saponins (QB‑90, IQB‑90) | Leaf triterpenoid saponins, Q. brasiliensis | Experimental viral and influenza vaccines (incl. intranasal) | Preclinical | (44, 45) |
| Plant‑cell‑culture QS‑21 (ccQS‑21) | QS‑21 from Q. saponaria cell culture | Compared with clinical QS‑21; proposed to supply AS‑type systems | Preclinical (manufacturing platform) | (47) |
| Synthetic / semisynthetic QS‑21 analogues | QS‑21 variants by chemical synthesis | Cancer and infectious‑disease subunit vaccines | Preclinical | (41, 48) |
| Nasal saponin adjuvants | Saponins from several plants | Nasal vaccines for respiratory viruses | Preclinical | (42, 26) |
| Advax (delta‑inulin) | Crystalline inulin (plant fructan) | Influenza, hepatitis B, coccidial vaccines | Phase I-II | (36, 49, 50) |
| TCM plant polysaccharides (APS, RGP, IRPS, etc.) | Polysaccharides from Astragalus, Rehmannia, Isatis, others | Infectious disease and cancer subunit vaccines (mostly animal) | Preclinical | (50-52) |
| General plant polysaccharides (glucans, mannans, inulin variants) | Various plant‑derived polysaccharides | Subunit and nano‑vaccines | Some in human use / trials | (49, 53, 54) |
| Plant phenolics (flavonoids etc.) in nanoadjuvants | Phenolics/flavonoids from multiple plants | Experimental cancer vaccines | Preclinical | (55, 56) |
| Plant virus‑like particles (pVLPs) | Cowpea mosaic virus, tobacco mosaic virus VLPs | Cancer and infectious‑disease vaccines | Preclinical; some early clinical candidates | (55) |
| Plant lectins (mistletoe lectin, garlic lectins, others) | Glycoprotein lectins from plants | Experimental viral and cancer vaccines, incl. COVID‑19 concepts | Preclinical / limited non‑vaccine clinical use | (45, 56) |
Together, Supplemental Table 1 and Table 1 illustrate the continuum from mechanistic discovery to clinical application, underscoring both the promise of PSMs and the need for rigorous evaluation before widespread adoption.
Formulation and Delivery Of PSM-based Vaccines
Practical feasibility (formulation and carriers): Encapsulation within biodegradable polymers (e.g., PLGA, chitosan) and inorganic matrices (e.g., mesoporous silica, calcium phosphate) can improve antigen protection and immunogenicity in preclinical models, but each carrier class imposes distinct analytical and manufacturing demands (residual solvent control, particle characterization, endotoxin testing). Technologies that enable thermostability, simple aseptic fill‑finish, and minimal cold‑chain dependence are favoured for low‑resource settings.
Nanoformulations and Stability
Encapsulating PSMs into nanoparticles enhances vaccine stability and immunogenicity, improving antigen delivery and avoiding degradation. Natural adjuvants in vaccine formulations, such as saponins, alkaloids, and flavonoids, have shown immunostimulatory effects. Bioactive compounds have been encapsulated using poly (D, L-lactide-co-glycolide) (PLGA) and chitosan-based nanoparticles, preventing enzymatic degradation and ensuring controlled antigen release for prolonged immune activation (57). Moreover, enhancing antigen stability and uptake by antigen-presenting cells is achieved using mesoporous silica and calcium phosphate nanoparticles, promoting robust humoral and cellular responses (58).
Saponin-based adjuvants such as QS-21 enhance dendritic cell activation and T-cell responses; moreover, the co-encapsulation of PSMs with antigens further augments immunogenicity (59); however, challenges such as cytotoxicity, inconsistent bioavailability, and nanoparticle aggregation need meticulous optimization of nanoformulation procedures (60).
Practical feasibility (manufacturing, regulation, deployment): Near‑term applicability depends on validated manufacturing platforms, standardized phytochemical characterization, GLP toxicology, and regulatory alignment. Key operational constraints include scale‑up complexity, quality‑control capacity, cold‑chain logistics, and cost. Prioritise candidates with clear supply‑chain pathways, scalable extraction or synthetic routes, and existing regulatory precedents to accelerate clinical translation in African contexts.
Oral and Intranasal Delivery
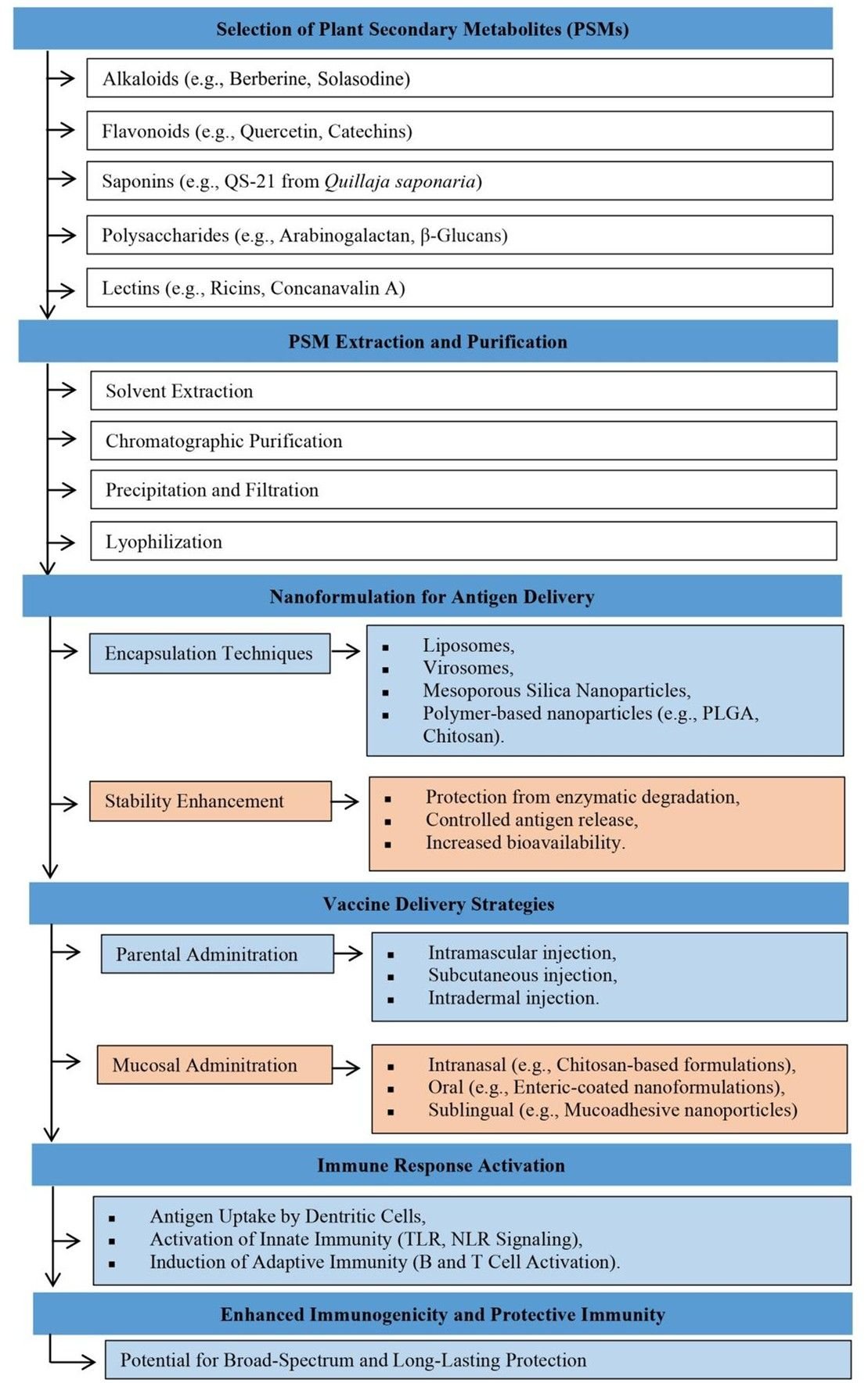
The potential for needle-free vaccination with plant-derived adjuvants is gaining attention, especially in mucosal immunization; oral and Intranasal vaccines protect pathogen entry sites and may enhance systemic and mucosal immunity. Current vaccine formulations include polysaccharides, saponins, lectins, and PSMs that enhance mucosal immune responses (61). Enhanced IgA and IgG responses result from increased antigen absorption by mucosal-associated lymphoid tissues using liposomal and virosomal delivery systems (62). Chitosan-based nanoparticles, recognized for their mucoadhesive properties, enhance antigen retention and immune activation in oral and nasal vaccines (63). Using flagellin, a Toll-like receptor 5 agonist, enhances the immunogenicity of Intranasal vaccines by augmenting dendritic cell activation and cytokine production as a mucosal adjuvant (64). Despite these advancements, challenges such as mucosal tolerance and enzymatic degradation in the gastrointestinal tract must be addressed to optimize plant-based adjuvants for oral and Intranasal vaccine delivery. Figure 2 presents a flowchart depicting PSM-based vaccine formulation and delivery strategies, from selection of plant secondary metabolites such as alkaloids, flavonoids, saponins, polysaccharides, and lectins, to PSM extraction and purification to nanoformulation for antigen delivery, to vaccine delivery strategies such as parental and mucosal administration to immune response activation, and finally to enhanced immunogenicity and protective immunity.
Current Challenges and Gaps
PSMs are a potential new area for vaccine research. Still, many challenges must be solved before they can be used widely in Africa; the main problems are a lack of standardization and dosage consistency. PSMs are complicated combinations of bio-active substances; their chemical profiles depend on the species, how they were grown, where they came from, and how they were extracted. Despite this heterogeneity, replicating data and establishing safe, effective dose schedules isn't easy. Comparing various research outcomes is difficult without strong quality control procedures and standardized reporting standards; obtaining regulatory approval is therefore challenging (65).
Issues concerning the toxicity and safety of PSMs are also equally important. In contrast, many plants containing PSMs are used traditionally and are presumed safe; however, controlled biomedical applications demand more rigorous evaluation (66). Heavy metals, adulterants, or environmental contaminants can make safety assessments even harder. Recent reviews emphasize the importance of thorough toxicological profiling, encompassing authentication and contaminant screening, to ensure consumer safety (67, 68). However, toxicological research capacity remains limited in most African nations, and few PSMs have undergone the systematic pre-clinical, and clinical trials necessary to establish safe therapeutic windows (69-71).
Regulatory integration and staged evidence pathway: Scientific claims about PSMs must align with regulatory expectations. Evidence gaps map directly to requirements: (i) compound characterization for CMC dossiers; (ii) controlled preclinical safety/immunogenicity for first‑in‑human studies; (iii) GLP toxicology and stability for trial authorization; and (iv) clinical trial designs with predefined endpoints. A staged pathway is recommended, with early engagement of regional regulators and WHO prequalification advisors to align evidence packages with African deployment goals.
Future Prospects and Research Directions
An adjuvant is a substance that can augment an increase in the immunogenicity of a vaccine. The traditional starting point has been to evaluate substances to increase the antibody titer to a given antigen in an animal model. Lately, this has been broadened to include immunogenicity measurements such as an increase in specific antibody isotypes, in cell-mediated immune responses, e.g., IFN-γ Elispot responses, Th1 and Th2 cytokine responses, as well as to provoke a reaction of the innate immune system, including activation and differentiation of dendritic cells. These functional assays define an adjuvant's qualitative and quantitative profiles. An example includes studies of lipopeptides, which are known to bind to toll-like receptor 2 (TLR2) and other pattern recognition receptors (PRRs) on macrophages. The most potent of these compounds, P3CSK4, causes NF-κB p65 translocation and enhanced tyrosine protein phosphorylation, confirming the involvement of the TLR NF-κB translocation pathway. Specific genes induced or repressed by P3CSK4 treatment of bone marrow-derived macrophages were examined in a cDNA expression array that monitored 140 genes. Activation of IL-1β, IL-7, and IL-15 genes was noted, consistent with activation of the NF-κB translocation pathway (72). Another gene expression array analysis example examined the effect of imiquimod derivative S-28463 on macrophages. Thirteen genes were induced from the 588 screened genes, which are known to be involved in macrophage activation and inflammation, and included IL-1β (73). Hence, using such tools as gene activation may provide a more precise characterization of the cellular action of adjuvants. However, it remains challenging to correlate these results with immunological outcome in vivo. The effects of the well-known adjuvants aluminum phosphate (Th2) and Freund’s complete adjuvant (Th1/Th2) on gene expression were examined in splenocytes from mice immunized two weeks earlier with toxoid antigens (36, 74). These adjuvants have been found to activate 61 genes, with primary differences in the kinetics of the responses, where aluminum phosphate was shown to activate the genes earlier. Genes involved in immune function, apoptosis, and signal transduction were identified (75).
Microneedle drug delivery systems have been widely used to treat various diseases due to their advantages of being minimally invasive, non-invasive, easy to administer, improving patient compliance, reducing side effects, and penetrating the stratum corneum. Because of the benefits of the microneedle drug delivery system, the microneedle drug delivery system has been the focus of pharmaceutical and material science research in recent years. With the development of microneedle technology, new microneedles emerge as time requires, and researchers will face new challenges. The biocompatibility of these new materials and whether there are other side effects on organisms needs to be determined by further research. Meanwhile, most of these materials have not been approved by regulators (76). The use of biodegradable polymers for the administration of pharmaceuticals and biomedical devices has increased dramatically. Biodegradable polymers' most important biomedical applications are in controlled drug delivery systems, such as implants and devices for bone and dental repairs (77).
Clinical and Public Health Implementations
PSMs may potentially offer opportunities for regionally relevant vaccine research; however, evidence for routine efficacy, cost‑effectiveness, and operational advantages remains preliminary and requires formal clinical and economic evaluation (78). They may also lessen the need for pricey synthetic adjuvants, which could reduce production costs overall (79). Such tactics could lessen dependency on cold-chain-dependent formulations and imported vaccine components in settings with limited resources, resolving significant distribution and accessibility issues. Additionally, using medicinal plants readily available in the area can strengthen communities and encourage self-sufficient vaccine development (80).
Clinically, PSM-based adjuvants have increased vaccine shelf life, strengthened humoral and cellular immune responses, and enhanced antigen stability (81). For instance, it has been demonstrated that formulations based on saponins from Q. saponaria can produce strong immune responses with few adverse effects, suggesting they are suitable for human applications (82). Similarly, curcumin and other bioactive substances can support long-term immunological memory, improve dendritic cell activation, and modify inflammatory pathways, all of which may contribute to improved vaccine responsiveness (83, 84). However, clinical translation necessitates thorough preclinical and phase I-III trials to assess safety, optimal dosage, and immunogenicity in various populations.
Increasing local capacity in phytomedicine research, production, quality control, and integrating PSM-based vaccines could improve healthcare systems at the public health level (85). Programs that teach scientists, chemists, and medical professionals how to formulate natural products and administer vaccines can help to develop long-lasting expertise. Additionally, vaccination acceptance and coverage can be increased by using needle-free delivery methods, such as oral or Intranasal vaccines enhanced with adjuvants derived from plants (86). This is especially true for children and populations with limited access to healthcare facilities.
The cost benefits, immunogenic potential, and safety profiles of conventional and PSM-based adjuvants are contrasted in Table 3. All things considered, PSM-based vaccines offer scalable, reasonably priced, and successful interventions for preventing infectious diseases in Africa by combining traditional knowledge with contemporary immunology. These strategies can support resilient immunization programs and equitable global health outcomes by bridging ethnobotany and clinical practice.
| S/N | Parameter | Conventional Adjuvants (e.g., Alum, MF59) | PSM-Based Adjuvants (e.g., Quillaja saponins, Curcumin, Withania extracts) | References |
|---|---|---|---|---|
| 1 | Cost of Production | High - relies on synthetic or imported components. | Moderate to Low - can be sourced locally from medicinal plants. | (87) |
| 2 | Immunogenicity | Moderate - may require multiple doses. | High - can enhance humoral and cellular immune responses. | (81) |
| 3 | Safety Profile | Established but may cause local inflammation or reactogenicity. | Generally safe if standardized; minimal adverse effects reported in pre-clinical studies. | (88) |
| 4 | Stability | Requires a strict cold chain for some formulations. | Can improve antigen stability through nano-encapsulation and natural stabilizing properties. | (89) |
| 5 | Ease of Administration | Injectable (IM or SC) only. | Potential for oral, intranasal, or transdermal delivery. | (88) |
| 6 | Scalability in Africa | Limited by import dependency and cost. | High - local sourcing of plant materials allows scalable production. | (89) |
| 7 | Regulatory Approval Status | Fully approved and widely used. | Emerging - requires rigorous preclinical and clinical testing | (90) |
| 8 | Sustainability | Depends on industrial production. | Environmentally sustainable; supports local communities and biodiversity. | (82) |
| 9 | Immunomodulatory Effects | Primarily, the adjuvant effect. | Multifaceted - adjuvant plus immunomodulatory, anti-inflammatory, and antiviral properties. | (91) |
Policy Recommendations and Capability Building
Robust capacity enhancement and targeted policy measures are crucial for integrating PSMs into African vaccine research and development. Prioritizing the support and preservation of indigenous knowledge systems, which have historically underpinned the utilization of medicinal plants in African communities, is essential (92). Policymakers must establish frameworks that safeguard intellectual property rights, provide equitable benefit-sharing, and recognize the contributions of traditional healers (93). Governments can foster innovation in modern vaccine science and protect cultural heritage by implementing community-based bio-prospecting (94). It is equally crucial to sustain infrastructure and funding to advance vaccine development and phytopharmaceutical research (95). The absence of professional facilities for systematically extracting, characterizing, and testing PSMs in most African countries limits their potential application as efficient adjuvants in vaccines. The foundation for large-scale production will be formed by creating regional centers of excellence equipped with cutting-edge laboratories, bioreactors, and quality control equipment. Funding sources should prioritize multidisciplinary initiatives integrating immunology, pharmacology, and ethnobotany to bridge the divide between traditional plant utilization and biomedical application (96). This infrastructure will enhance domestic vaccine production and reduce dependence on imported adjuvants and formulations.
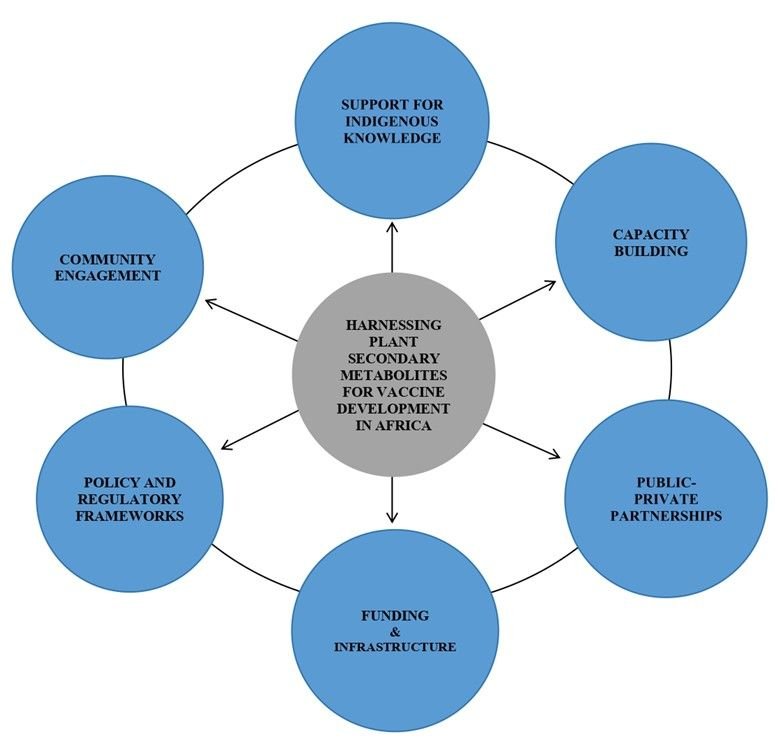
To ensure sustainability and stimulate innovation, public-private partnerships (PPPs) are essential (97). Strategic partnerships among academic institutions, research centers, pharmaceutical companies, and traditional medicine practitioners can improve knowledge sharing, technology transfer, and market integration (98). Educational institutions provide training and research proficiency, but industry involvement presents production scale and regulatory navigation opportunities. Collaborations with traditional practitioners at the community level foster trust and facilitate culturally sensitive implementation. PPPs can establish a sustainable framework for vaccine development in Africa by aligning public health priorities with indigenous knowledge systems and private sector innovation. By fortifying health systems and promoting scientific autonomy, these policy measures and capacity-building endeavors will enable African nations to use their biodiversity to develop innovative vaccines. A conceptual framework is shown in Figure 3 to allow the harnessing of PSMs for vaccine development in Africa; capacity building, community engagement, public-private partnerships, and improvement of policy and regulatory frameworks should be included.
Ethical and Regulatory Considerations
Nonclinical and early clinical evaluation of candidate PSMs intended as vaccine adjuvants should follow staged, evidence‑based requirements. A complete nonclinical dossier is expected to include GLP‑compliant pharmacology and toxicology studies that characterize dose-response relationships, local reactogenicity, systemic toxicity, and genotoxicity where relevant (99). Bridging studies should translate mechanistic in vitro and animal findings into defined biomarkers and antigen‑specific endpoints to justify first‑in‑human dosing and species selection; dose‑scaling rationale must be documented (100). Manufacturing and quality control must provide reproducible chemical definition of botanical starting materials and final adjuvant preparations. Batch release criteria should include validated assays for identity, purity, residual solvents, endotoxin, and quantification of key phytochemical markers. Process validation for extraction, purification, and downstream processing is required to support stability and comparability claims used in nonclinical and clinical studies (101).
Regulatory review typically follows a staged pathway: compound characterization, nonclinical safety package, phased clinical trials with predefined immunological and clinical endpoints. For multi‑country trials, regional joint review and reliance mechanisms can harmonize data expectations and streamline ethics and regulatory approvals; early scientific advice from national regulators or regional platforms is recommended to clarify adjuvant‑specific data requirements (102, 103).
Ethical oversight and trial safety monitoring must be explicit in protocols. First‑in‑human and early efficacy trials should include independent data‑safety monitoring boards, predefined stopping rules for reactogenicity, and justification of risk-benefit for participant populations (104). Pharmacovigilance planning should be defined prior to large‑scale trials or deployment, including active surveillance strategies and regional signal‑evaluation pathways for rare adverse events (100, 105).
Limitations
The review is a structured narrative synthesis rather than a systematic review and was limited to English‑language publications and the databases specified in Methods, which may omit relevant regional or non‑English sources. Evidence is heterogeneous, comprising in vitro studies, diverse animal models, formulation experiments and a limited number of human trials; therefore, many conclusions are provisional and primarily preclinical. Chemical variability of plant extracts and inconsistent phytochemical characterization reduce reproducibility and complicate dose‑response and safety assessment. Clinical data are uneven across compound classes and few studies report protective clinical endpoints. Formulation claims (shelf life, field stability) are largely supported by preclinical data and require clinical validation.
Recommendations
In advancing plant‑derived vaccine adjuvants or immunomodulators, it is essential to prioritize candidates that demonstrate clear antigen‑specific evidence. Only partially purified substances or characterized extracts that reproducibly elicit antigen‑specific immunogenicity or protection in controlled co‑formulation studies should be taken forward, while signals observed solely in vitro must remain hypothesis‑generating rather than definitive. To ensure rigor and comparability, chemical characterization must be standardized across all reports, encompassing botanical identification, extraction methodology, marker‑compound quantification, and validated analytical assays in both preclinical and formulation studies.
For each candidate, developers should provide a transparent account of the anticipated manufacturing pathway, including key analytical requirements such as sterility testing, particle characterization, and residual solvent analysis. Cost drivers must be identified early, alongside a realistic plan for regional manufacture or fill‑finish capacity. Formulations that demonstrate thermostability or reduced dependence on cold‑chain logistics, validated through accelerated and real‑time stability studies under African climatic conditions, should be prioritized.
Nonclinical safety and bridging studies must be staged strategically, with targeted GLP‑compliant toxicology or bridging experiments designed to translate mechanistic endpoints into human‑relevant biomarkers before initiating first‑in‑human trials. Equally important is early engagement with regional regulators and partners, leveraging joint review or reliance pathways where possible, and aligning data packages with both regulatory expectations and local manufacturing capabilities.
Clinical development must be guided by predefined endpoints and safety monitoring frameworks. Immunological primary and secondary endpoints should be established in advance, together with arrangements for independent data safety monitoring boards, stopping rules for reactogenicity, and a pharmacovigilance plan tailored to multi‑site trials in target populations.
Conclusion
Plant secondary metabolites (PSMs) demonstrate measurable immunological effects that are relevant to vaccine science, but the current evidence base is predominantly preclinical. In vitro and animal studies consistently show that PSMs can activate MAPK, PI3K/Akt and NF‑κB signalling, promote dendritic cell maturation, and improve antigen stability when incorporated into delivery systems such as PLGA or chitosan nanoparticles; these data establish mechanistic plausibility but do not permit direct extrapolation to human vaccine efficacy. Clinical validation is limited: QS‑21‑containing saponin systems remain the clearest example of a PSM‑derived component that has advanced through Phase I-III evaluation within licensed adjuvant platforms, whereas candidates such as arabinogalactan, curcumin and quercetin show reproducible preclinical effects but lack robust human data. Compared with conventional adjuvants (e.g., alum, MF59), which benefit from extensive clinical experience, reproducibility and established safety‑monitoring frameworks, PSM‑based approaches offer chemical diversity and novel mechanisms but face substantial translational barriers. Key constraints include inconsistent compound characterization, variable bioavailability, dose‑finding uncertainty, incomplete toxicological profiling, manufacturing and analytical capacity gaps, cold‑chain and fill‑finish considerations, and regional regulatory readiness. Safety and immunogenicity are highly compound‑ and formulation‑dependent; reports of cytotoxicity, nanoparticle aggregation and inconsistent reporting further increase uncertainty. Given these realities, advancement should follow staged, evidence‑driven milestones: standardized botanical identification and phytochemical characterization; controlled antigen co‑formulation studies with predefined immunological endpoints; GLP‑compliant toxicology and validated stability testing under field‑relevant conditions; and adequately powered clinical trials in representative populations. Importantly, the methodological scope of this review imposes constraints. As a purposive narrative synthesis, it draws on heterogeneous studies with variable endpoints, limited antigen‑specific data, and selective use of grey literature. These factors restrict causal inference and highlight the need for cautious interpretation. Policy or clinical endorsement should be conditional on meeting these thresholds. In sum, PSMs remain promising mechanistic leads for adjuvant discovery, but responsible translation requires conservative, stepwise development grounded in standardization, rigorous safety evaluation, manufacturability assessment and regionally appropriate regulatory engagement.
Abbreviations
A549 cells = Adenocarcinomic Human Alveolar Basal Epithelial Cells; AG = Arabinogalactan; AMA = African Medicines Agency; AP-1 = Activator Protein; APC = Antigen Presenting Cell; Art = Artemisinin; Bcl-xL = B-cell Lymphoma-Extra Large; CD11b = Cluster of Differentiation Molecule 11b; cDNA = Complementary DNA; Con-A = Concanavalin A; COX-2 = Cyclooxygenase-2; ENL = Erythema Nodosum Leprosum; FLS = Fibroblast-Like Synoviocytes; HCQ = Hydroxychloroquine; HMGB1 = High Mobility Group Box 1; HO-1 = Heme Oxygenase-1; HUVECs = Human Umbilical Vein Endothelial Cells; IAP1 = Inhibitor of Apoptosis Protein 1; ICAM-1 Intercellular Adhesion Molecule 1; IKK = Inhibitor of Nuclear Factor Kappa-B Kinase; IL = Interleukin; IM = Intramuscular; iNOS = Inducible Nitric Oxide Synthase; JAK-STAT = Janus Kinase-Signal Transducer and Activator of Transcription; JNK = c-Jun N-terminal Kinases; LAK = Lymphokine-Activated Killer Cells; LPS = Lipopolysaccharide; MAPK = Mitogen-Activated Protein Kinase; ML3 = Maturity Level 3; MMP-1 = Matrix Metalloproteinase-1; MMP-3 = Matrix Metalloproteinase-3; MOLP = Moringa Oleifera Leaf Polysaccharide; MOLP-PE = Moringa Oleifera Leaf Polysaccharide-Purified Extract; NFAT = Nuclear Factor of Activated T-Cells; NF-κB = Nuclear Factor Kappa-Light-Chain-Enhancer of Activated B Cells; NLRP3 = NOD-Like Receptor Protein 3; NOS-2 = Nitric Oxide Synthase 2; Nrf2 = Nuclear Factor Erythroid 2-Related Factor 2; PAMPs = Pathogen-Associated Molecular Patterns; PBMCs = Peripheral Blood Mononuclear Cells; PHA = Phytohemagglutinin; PI3K/Akt = Phosphatidylinositol 3-Kinase / Protein Kinase B; PLGA = Poly (D, L-Lactide-Co-Glycolide); PPPs = Public-Private Partnerships; PRRs = Pattern Recognition Receptors; PSMs = Plant Secondary Metabolites; QS-21; Quillaja Saponin 21; RA = Rheumatoid Arthritis; RT = Rutin; SC = Subcutaneous; SOCS-3 = Suppressor of Cytokine Signaling 3; STAT-1 = Signal Transducer and Activator of Transcription 1; TGF-β1 = Transforming Growth Factor Beta 1; Th1 = T-helper Type 1; TLR4 = Toll-Like Receptor 4; TNF-α = Tumor Necrosis Factor Alpha; Tregs = Regulatory T Cells; WHO = World Health Organization; Wnt = Wingless/Integrated Signaling Pathway; XIAP = X-Linked Inhibitor of Apoptosis Protein
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
Publicly available
Ethics Statement
Not applicable.
Funding Information
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Supplemental Material
The <a class="cursor-pointer" href="https://etflin.com/file/document/20260406072604_19260_82581387.docx">Supplementary Data</a> contains <b>Supplemental Table 1</b>. Summary of African plants with PSMs and their pharmacological properties relevant to vaccine development. <br />
References
- Jan R, Asaf S, Numan M, Lubna, Kim KM. Plant Secondary Metabolite Biosynthesis and Transcriptional Regulation in Response to Biotic and Abiotic Stress Conditions. Agronomy. 2021;11(5):968. doi: https://doi.org/10.3390/agronomy11050968
- Yeshi K, Crayn D, Ritmejerytė E, Wangchuk P. Plant Secondary Metabolites Produced in Response to Abiotic Stresses Has Potential Application in Pharmaceutical Product Development. Molecules. 2022;27(1):313. doi: https://doi.org/10.3390/molecules27010313
- Gutiérrez-Grijalva EP, López-Martínez LX, Contreras-Angulo LA, Elizalde-Romero CA, Heredia JB. Plant Alkaloids: Structures and Bioactive Properties. Singapore: Springer Singapore; 2020. doi: https://doi.org/10.1007/978-981-15-2361-8_5
- Martínez G, Mijares MR, De Sanctis JB. Effects of Flavonoids and Its Derivatives on Immune Cell Responses. Iad. 2019;13(2):84-104. doi: https://doi.org/10.2174/1872213x13666190426164124
- Brahmkshatriya PP, Brahmkshatriya PS. Terpenes: Chemistry, Biological Role, and Therapeutic Applications. Berlin, Heidelberg: Springer Berlin Heidelberg; 2013. doi: https://doi.org/10.1007/978-3-642-22144-6_120
- Hu Y, Wu Y, Liu H, Cheng S, Wen F, Wan L. QS-21: Structural insights, immunological mechanisms, and prospects for rational design of safer vaccine adjuvants. European Journal of Medicinal Chemistry. 2026;301:118223. doi: https://doi.org/10.1016/j.ejmech.2025.118223
- Murphy EJ, Fehrenbach GW, Abidin IZ, Buckley C, Montgomery T, Pogue R, et al. Polysaccharides—Naturally Occurring Immune Modulators. Polymers. 2023;15(10):2373. doi: https://doi.org/10.3390/polym15102373
- Wang ZB, Xu J. Better Adjuvants for Better Vaccines: Progress in Adjuvant Delivery Systems, Modifications, and Adjuvant–Antigen Codelivery. Vaccines. 2020;8(1):128. doi: https://doi.org/10.3390/vaccines8010128
- Shahbazi S, Bolhassani A. Immunostimulants: types and functions. Journal of Medical Microbiology and Infectious Diseases. 2016 Jul 10;4(3):45-51.
- Shahidi F. Nutraceuticals, functional foods and dietary supplements in health and disease. Journal of Food and Drug Analysis. 2020;20(1). doi: https://doi.org/10.38212/2224-6614.2144
- Downing NS, Aminawung JA, Shah ND, Krumholz HM, Ross JS. Clinical Trial Evidence Supporting FDA Approval of Novel Therapeutic Agents, 2005-2012. Jama. 2014;311(4):368. doi: https://doi.org/10.1001/jama.2013.282034
- Ali Reza ASM, Nasrin MS, Hossen MA, Rahman MA, Jantan I, Haque MA, et al. Mechanistic insight into immunomodulatory effects of food-functioned plant secondary metabolites. Critical Reviews in Food Science and Nutrition. 2021;63(22):5546-5576. doi: https://doi.org/10.1080/10408398.2021.2021138
- Arslan I. Quillaic Acid–Containing Saponin-Based Immunoadjuvants Trigger Early Immune Responses. Rev. Bras. Farmacogn. 2020;30(4):467-473. doi: https://doi.org/10.1007/s43450-020-00080-y
- Wallace F, Fontana C, Ferreira F, Olivaro C. Structure Elucidation of Triterpenoid Saponins Found in an Immunoadjuvant Preparation of Quillaja brasiliensis Using Mass Spectrometry and 1H and 13C NMR Spectroscopy. Molecules. 2022;27(8):2402. doi: https://doi.org/10.3390/molecules27082402
- O’Hagan DT, Lodaya RN, Lofano G. The continued advance of vaccine adjuvants – ‘we can work it out’. Seminars in Immunology. 2020;50:101426. doi: https://doi.org/10.1016/j.smim.2020.101426
- Ragupathi G, Gardner JR, Livingston PO, Gin DY. Natural and synthetic saponin adjuvant QS-21 for vaccines against cancer. Expert Review of Vaccines. 2011;10(4):463-470. doi: https://doi.org/10.1586/erv.11.18
- Naji E, Abdulfatah HF, AlZawi K. Plant Secondary Metabolites, Their Classification and Biological Roles: A Review. Juaps. 2024;18(1):106-115. doi: https://doi.org/10.37652/juaps.2023.144549.1164
- Widjaja SS, Rusdiana R, Amelia R. Curcumin. Journal of Advanced Pharmaceutical Technology & Research. 2022;13(3):187-190. doi: https://doi.org/10.4103/japtr.japtr_54_22
- Fessler SN, Chang Y, Liu L, Johnston CS. Curcumin Confers Anti-Inflammatory Effects in Adults Who Recovered from COVID-19 and Were Subsequently Vaccinated: A Randomized Controlled Trial. Nutrients. 2023;15(7):1548. doi: https://doi.org/10.3390/nu15071548
- Kulkarni-Munshi R, Talmohite D, More A, Chakravarty J, Kamat S, Khobragade A, et al. Ashwagandha, Withania somnifera (L.) Dunal, for the prophylaxis against SARS-CoV-2 infection: A multicentric randomized hydroxychloroquine controlled clinical trial in Indian health care workers. Journal of Ayurveda and Integrative Medicine. 2025;16(3):101135. doi: https://doi.org/10.1016/j.jaim.2025.101135
- Kashyap VK, Dhasmana A, Yallapu MM, Chauhan SC, Jaggi M. Withania somnifera as a potential future drug molecule for COVID-19. Future Drug. Discov. 2020;2(4). doi: https://doi.org/10.4155/fdd-2020-0024
- Zebeaman M, Tadesse MG, Bachheti RK, Bachheti A, Gebeyhu R, Chaubey KK. Plants and Plant‐Derived Molecules as Natural Immunomodulators. BioMed Research International. 2023;2023(1). doi: https://doi.org/10.1155/2023/7711297
- Li T, Chen H, Wei N, Mei X, Zhang S, Liu DL, et al. Anti-inflammatory and immunomodulatory mechanisms of artemisinin on contact hypersensitivity. International Immunopharmacology. 2012;12(1):144-150. doi: https://doi.org/10.1016/j.intimp.2011.11.004
- Gu J, Yang X, Lin L, Zhao M. Identification of an arabinogalactan with special structure from Moringa Oleifera leaf and exploration of its immunomodulatory activity. International Journal of Biological Macromolecules. 2024;279:134616. doi: https://doi.org/10.1016/j.ijbiomac.2024.134616
- Islamuddin M, Chouhan G, Farooque A, Dwarakanath BS, Sahal D, Afrin F. Th1-Biased Immunomodulation and Therapeutic Potential of Artemisia annua in Murine Visceral Leishmaniasis. PLoS Negl Trop Dis. 2015;9(1):e3321. doi: https://doi.org/10.1371/journal.pntd.0003321
- Suyama TL, Gerwick WH, McPhail KL. Survey of marine natural product structure revisions: A synergy of spectroscopy and chemical synthesis. Bioorganic & Medicinal Chemistry. 2011;19(22):6675-6701. doi: https://doi.org/10.1016/j.bmc.2011.06.011
- Shahrajabian MH, Sun W, Cheng Q. Exploring Artemisia annua L., artemisinin and its derivatives, from traditional Chinese wonder medicinal science. Not Bot Horti Agrobo. 2020;48(4):1719-1741. doi: https://doi.org/10.15835/nbha48412002
- Mathur S, Hoskins C. Drug development: Lessons from nature. Biomedical Reports [Internet]. 2017 May 9;6(6):612–4. https://doi.org/10.3892/br.2017.909
- Ashong GW, Kwaansa – Ansah EE, Alhassan M, Adofo PN, Sarkodie JA, Ababio BA. Comparative analysis of phytochemical constituents in different parts of Aspilia africana: A potential medicinal plant for therapeutic applications. Phytomedicine Plus. 2024;4(4):100656. doi: https://doi.org/10.1016/j.phyplu.2024.100656
- Okello D, Kang Y. Exploring Antimalarial Herbal Plants across Communities in Uganda Based on Electronic Data. Evidence-Based Complementary and Alternative Medicine. 2019;2019:1-27. doi: https://doi.org/10.1155/2019/3057180
- K.O. Ajeigbe, A.A. Onifade, D.R. Omotoso, S.S. Enitan, Olaleye SB. Anti-ulcerogenic Activity of Aspilia Africana Leaf Extract: Roles of Gastric Acid, Oxidative Stress and Neutrophil Infiltration. African Journal of Biomedical Research [Internet]. 2016 [cited 2025 Sep 4];17(3):193–201. Available from: https://www.ajol.info/index.php/ajbr/article/view/135739
- Okoli C, Akah P, Nwafor S, Anisiobi A, Ibegbunam I, Erojikwe O. Anti-inflammatory activity of hexane leaf extract of Aspilia africana C.D. Adams. Journal of Ethnopharmacology. 2007;109(2):219-225. doi: https://doi.org/10.1016/j.jep.2006.07.037
- Okello D, Lee J, Kang Y. Ethnopharmacological Potential of Aspilia africana for the Treatment of Inflammatory Diseases. Evidence-Based Complementary and Alternative Medicine. 2020;2020(1). doi: https://doi.org/10.1155/2020/8091047
- Lin WC, Lin JY. Berberine down-regulates the Th1/Th2 cytokine gene expression ratio in mouse primary splenocytes in the absence or presence of lipopolysaccharide in a preventive manner. International Immunopharmacology. 2011;11(12):1984-1990. doi: https://doi.org/10.1016/j.intimp.2011.08.008
- Banik U, Parasuraman S, Adhikary AK, Othman NH. Curcumin: the spicy modulator of breast carcinogenesis. J Exp Clin Cancer Res. 2017;36(1). doi: https://doi.org/10.1186/s13046-017-0566-5
- Yuandani, Jantan I, Rohani AS, Sumantri IB. Immunomodulatory Effects and Mechanisms of Curcuma Species and Their Bioactive Compounds: A Review. Front. Pharmacol. 2021;12. doi: https://doi.org/10.3389/fphar.2021.643119
- Kim JH, Nam GS, Kim SH, Ryu DS, Lee DS. Orostachys japonicus exerts antipancreatic cancer activity through induction of apoptosis and cell cycle arrest in PANC‐1 cells. Food Science & Nutrition. 2019;7(11):3549-3559. doi: https://doi.org/10.1002/fsn3.1207
- Hong Y, Lee J, Moon H, Ryu CH, Seok J, Jung YS, et al. Quercetin Induces Anticancer Activity by Upregulating Pro-NAG-1/GDF15 in Differentiated Thyroid Cancer Cells. Cancers. 2021;13(12):3022. doi: https://doi.org/10.3390/cancers13123022
- Thomazelli APFDS, Tomiotto-Pellissier F, Miranda-Sapla MM, da Silva SS, Alvarenga DS, Panis C, et al. Concanavalin-A displays leishmanicidal activity by inducing ROS production in human peripheral blood mononuclear cells. Immunopharmacology and Immunotoxicology. 2018;40(5):387-392. doi: https://doi.org/10.1080/08923973.2018.1510960
- Morais V, Suarez N, Cibulski S, Silveira F. Leaf Saponins of Quillaja brasiliensis as Powerful Vaccine Adjuvants. Pharmaceutics. 2025;17(8):966. doi: https://doi.org/10.3390/pharmaceutics17080966
- Sander VA, Corigliano MG, Clemente M. Promising Plant-Derived Adjuvants in the Development of Coccidial Vaccines. Front. Vet. Sci. 2019;6. doi: https://doi.org/10.3389/fvets.2019.00020
- Kumar A, Sharma A, Tirpude NV, Padwad Y, Hallan V, Kumar S. Plant-derived immuno-adjuvants in vaccines formulation: a promising avenue for improving vaccines efficacy against SARS-CoV-2 virus. Pharmacol. Rep. 2022;74(6):1238-1254. doi: https://doi.org/10.1007/s43440-022-00418-4
- Lacaille-Dubois MA, Wagner H. New perspectives for natural triterpene glycosides as potential adjuvants. Phytomedicine. 2017;37:49-57. doi: https://doi.org/10.1016/j.phymed.2017.10.019
- Lv X, Martin J, Hoover H, Joshi B, Wilkens M, Ullisch DA, et al. Chemical and biological characterization of vaccine adjuvant QS-21 produced via plant cell culture. iScience. 2024;27(3):109006. doi: https://doi.org/10.1016/j.isci.2024.109006
- Lacaille-Dubois MA. Updated insights into the mechanism of action and clinical profile of the immunoadjuvant QS-21: A review. Phytomedicine. 2019;60:152905. doi: https://doi.org/10.1016/j.phymed.2019.152905
- Jia Y, Zhu H, Cai X, Sun C, Ye Y, Cai D, et al. Plant-Derived Immunomodulatory Nanoadjuvants for Cancer Vaccines: Current Status and Future Opportunities. Vaccines. 2025;13(4):378. doi: https://doi.org/10.3390/vaccines13040378
- Wan X, Yin Y, Zhou C, Hou L, Cui Q, Zhang X, et al. Polysaccharides derived from Chinese medicinal herbs: A promising choice of vaccine adjuvants. Carbohydrate Polymers. 2022;276:118739. doi: https://doi.org/10.1016/j.carbpol.2021.118739
- Zou M, Lei C, Huang D, Liu L, Han Y. Application of plant-derived products as adjuvants for immune activation and vaccine development. Vaccine. 2024;42(25):126115. doi: https://doi.org/10.1016/j.vaccine.2024.07.016
- Feng L, Zhang J, Ma C, Li K, Zhai J, Cai S, et al. Application prospect of polysaccharide in the development of vaccine adjuvants. International Journal of Biological Macromolecules. 2025;297:139845. doi: https://doi.org/10.1016/j.ijbiomac.2025.139845
- Li L, Honda‐Okubo Y, Khanna V, Sajkov D, Petrovsky N. Phase 2 randomized controlled trial of seasonal influenza vaccine shows Advax® delta inulin adjuvant accelerates the humoral anti‐influenza response. Immunol Cell Biol. 2025;103(8):794-808. doi: https://doi.org/10.1111/imcb.70050
- Sun B, Yu S, Zhao D, Guo S, Wang X, Zhao K. Polysaccharides as vaccine adjuvants. Vaccine. 2018;36(35):5226-5234. doi: https://doi.org/10.1016/j.vaccine.2018.07.040
- Wang D, Liu Y, Zhao W. The Adjuvant Effects on Vaccine and the Immunomodulatory Mechanisms of Polysaccharides From Traditional Chinese Medicine. Front. Mol. Biosci. 2021;8. doi: https://doi.org/10.3389/fmolb.2021.655570
- Liang X, Zhou J, Wang M, Wang J, Song H, Xu Y, et al. Progress and prospect of polysaccharides as adjuvants in vaccine development. Virulence. 2024;15(1). doi: https://doi.org/10.1080/21505594.2024.2435373
- Pifferi C, Fuentes R, Fernández-Tejada A. Natural and synthetic carbohydrate-based vaccine adjuvants and their mechanisms of action. Nat Rev Chem. 2021;5(3):197-216. doi: https://doi.org/10.1038/s41570-020-00244-3
- Reyna-Margarita HR, Irais CM, Mario-Alberto RG, Agustina RM, Luis-Benjamín SG, David PE. Plant Phenolics and Lectins as Vaccine Adjuvants. Cpb. 2019;20(15):1236-1243. doi: https://doi.org/10.2174/1389201020666190716110705
- Sharma T, Singh D, Mahapatra A, Mohapatra P, Sahoo S, Sahoo SK. Advancements in clinical translation of flavonoid nanoparticles for cancer treatment. OpenNano. 2022;8:100074. doi: https://doi.org/10.1016/j.onano.2022.100074
- Zhang W, Wang L, Liu Y, Chen X, Liu Q, Jia J, et al. Immune responses to vaccines involving a combined antigen–nanoparticle mixture and nanoparticle-encapsulated antigen formulation. Biomaterials. 2014;35(23):6086-6097. doi: https://doi.org/10.1016/j.biomaterials.2014.04.022
- Mody KT, Popat A, Mahony D, Cavallaro AS, Yu C, Mitter N. Mesoporous silica nanoparticles as antigen carriers and adjuvants for vaccine delivery. Nanoscale. 2013;5(12):5167. doi: https://doi.org/10.1039/c3nr00357d
- Talukdar P, Winegar PH, Hudson GA, Astolfi MCT, Inman JL, Keasling JD, et al. From bark to bench: innovations in QS-21 adjuvant characterization and manufacturing. Front. Immunol. 2025;16. doi: https://doi.org/10.3389/fimmu.2025.1677995
- Shen H, Ackerman AL, Cody V, Giodini A, Hinson ER, Cresswell P, et al. Enhanced and prolonged cross‐presentation following endosomal escape of exogenous antigens encapsulated in biodegradable nanoparticles. Immunology. 2005;117(1):78-88. doi: https://doi.org/10.1111/j.1365-2567.2005.02268.x
- Cox E, Verdonck F, Vanrompay D, Goddeeris B. Adjuvants modulating mucosal immune responses or directing systemic responses towards the mucosa. Vet. Res. 2006;37(3):511-539. doi: https://doi.org/10.1051/vetres:2006014
- Henriksen-Lacey M, Korsholm KS, Andersen P, Perrie Y, Christensen D. Liposomal vaccine delivery systems. Expert Opinion on Drug Delivery. 2011;8(4):505-519. doi: https://doi.org/10.1517/17425247.2011.558081
- Prego C, Paolicelli P, Díaz B, Vicente S, Sánchez A, González-Fernández Á, et al. Chitosan-based nanoparticles for improving immunization against hepatitis B infection. Vaccine. 2010;28(14):2607-2614. doi: https://doi.org/10.1016/j.vaccine.2010.01.011
- Huleatt JW, Jacobs AR, Tang J, Desai P, Kopp EB, Huang Y, et al. Vaccination with recombinant fusion proteins incorporating Toll-like receptor ligands induces rapid cellular and humoral immunity. Vaccine. 2007;25(4):763-775. doi: https://doi.org/10.1016/j.vaccine.2006.08.013
- Heinrich M, et al. Best Practice in the Chemical Characterisation of Extracts Used in Pharmacological and Toxicological Research The ConPhyMP Guidelines. Front Pharmacol. 2022;13:953205. https://doi.org/10.3389/fphar.2022.953205
- Karnwal A, Jassim AY, Mohammed AA, Sharma V, Al-Tawaha ARMS, Sivanesan I. Nanotechnology for Healthcare: Plant-Derived Nanoparticles in Disease Treatment and Regenerative Medicine. Pharmaceuticals. 2024;17(12):1711. doi: https://doi.org/10.3390/ph17121711
- Ullah A, Lim SI. Plant extract‐based synthesis of metallic nanomaterials, their applications, and safety concerns. Biotech & Bioengineering. 2022;119(9):2273-2304. doi: https://doi.org/10.1002/bit.28148
- Ciara MD, et al. Current trends in toxicity assessment of herbal medicines: a narrative review. Processes. 2023;11(1):83. https://doi.org/10.3390/pr11010083
- Hewitt P, Hartmann A, Tornesi B, Ferry-Martin S, Valentin JP, Desert P, et al. Importance of tailored non-clinical safety testing of novel antimalarial drugs: Industry best-practice. Regulatory Toxicology and Pharmacology. 2024;154:105736. doi: https://doi.org/10.1016/j.yrtph.2024.105736
- Duga A, Dereje N, Fallah MP, Angasa T, Bayih AG, Agbenu E, et al. Strengthening National Regulatory Authorities in Africa: A Critical Step Towards Enhancing Local Manufacturing of Vaccines and Health Products. Vaccines. 2025;13(6):646. doi: https://doi.org/10.3390/vaccines13060646
- Market Access Africa. Africa’s WHO Maturity Level 3 National Regulatory Authorities sign Memorandum of Understanding to establish a structured framework for reliance on regulatory decisions. Market Access Africa Regulatory Spotlight. 2025. Available from: https://www.marketaccess.africa/blogs/maa-regulatory-spotlight-12 (Accessed 2 September 2, 2025 at 09:48 am SAT)
- Esche U, Ayoub M, Pfannes S, Müller M, Huber M, Wiesmüller KH, et al. Immunostimulation by bacterial components: I. Activation of macrophages and enhancement of genetic immunization by the lipopeptide P3CSK4. International Journal of Immunopharmacology. 2000;22(12):1093-1102. doi: https://doi.org/10.1016/s0192-0561(00)00069-2
- Buates S, Matlashewski G. Identification of Genes Induced by a Macrophage Activator, S-28463, Using Gene Expression Array Analysis. Antimicrob Agents Chemother. 2001;45(4):1137-1142. doi: https://doi.org/10.1128/aac.45.4.1137-1142.2001
- Lindblad EB. Aluminum Adjuvants: Basic Concepts and Progress in Understanding. New York, NY: Springer New York; 2014. doi: https://doi.org/10.1007/978-1-4939-1417-3_3
- Zhao T, Cai Y, Jiang Y, He X, Wei Y, Yu Y, et al. Vaccine adjuvants: mechanisms and platforms. Sig Transduct Target Ther. 2023;8(1). doi: https://doi.org/10.1038/s41392-023-01557-7
- Zhang R, Miao Q, Deng D, Wu J, Miao Y, Li Y. Research progress of advanced microneedle drug delivery system and its application in biomedicine. Colloids and Surfaces B: Biointerfaces. 2023;226:113302. doi: https://doi.org/10.1016/j.colsurfb.2023.113302
- Aoki K, Saito N. Biodegradable Polymers as Drug Delivery Systems for Bone Regeneration. Pharmaceutics. 2020;12(2):95. doi: https://doi.org/10.3390/pharmaceutics12020095
- Díaz-Dinamarca DA, Salazar ML, Castillo BN, Manubens A, Vasquez AE, Salazar F, et al. Protein-Based Adjuvants for Vaccines as Immunomodulators of the Innate and Adaptive Immune Response: Current Knowledge, Challenges, and Future Opportunities. Pharmaceutics. 2022;14(8):1671. doi: https://doi.org/10.3390/pharmaceutics14081671
- Wang P. Natural and Synthetic Saponins as Vaccine Adjuvants. Vaccines. 2021 Mar 5;9(3):222. https://doi.org/10.3390/vaccines9030222
- Adhi Iman Sulaiman, Toto Sugito, Bambang Suswanto, Tri Nugroho Adi, Sri Weningsih. Community Empowerment Program in Management of Health Clinic Development and Herbal Tourism. icistech. 2024;1(1):82-92. doi: https://doi.org/10.62951/icistech.v1i1.21
- Singh AK, Yadav TP, Singh G, Pritam M, Pandey B, Ansari MI, et al. Nanosize Carriers for Drug and Vaccine Delivery: Advances and Challenges. Nanoasia. 2021;11(6). doi: https://doi.org/10.2174/2210681211666210505115217
- Cibulski S, Rivera-Patron M, Suárez N, Pirez M, Rossi S, Yendo AC, et al. Leaf saponins of Quillaja brasiliensis enhance long-term specific immune responses and promote dose-sparing effect in BVDV experimental vaccines. Vaccine. 2018;36(1):55-65. doi: https://doi.org/10.1016/j.vaccine.2017.11.030
- Therapeutic Implications of Natural Bioactive Compounds. Frontiers in bioactive compounds. 2022. https://doi.org/10.2174/97898150800251220301
- Canistro D, Chiavaroli A, Cicia D, Cimino F, Curro D, Dell Agli M, et al. The pharmacological basis of the curcumin nutraceutical uses: an update. Pharm Adv. 2021;03(02):421. doi: https://doi.org/10.36118/pharmadvances.2021.06
- El-Saadony MT, Zabermawi NM, Zabermawi NM, Burollus MA, Shafi ME, Alagawany M, et al. Nutritional Aspects and Health Benefits of Bioactive Plant Compounds against Infectious Diseases: A Review. Food Reviews International. 2021;39(4):2138-2160. doi: https://doi.org/10.1080/87559129.2021.1944183
- Nordin AH, Husna SMN, Ahmad Z, Nordin ML, Ilyas RA, Azemi AK, et al. Natural Polymeric Composites Derived from Animals, Plants, and Microbes for Vaccine Delivery and Adjuvant Applications: A Review. Gels. 2023;9(3):227. doi: https://doi.org/10.3390/gels9030227
- Firdaus FZ, Skwarczynski M, Toth I. Developments in Vaccine Adjuvants. Vaccine Design. 2021 Dec 17;145–78. https://doi.org/10.1007/978-1-0716-1892-9_8
- Jiang L, Zhang G, Li Y, Shi G, Li M. Potential Application of Plant-Based Functional Foods in the Development of Immune Boosters. Front. Pharmacol. 2021;12. doi: https://doi.org/10.3389/fphar.2021.637782
- Bouazzaoui A, Abdellatif AAH, Al-Allaf FA, Bogari NM, Al-Dehlawi S, Qari SH. Strategies for Vaccination: Conventional Vaccine Approaches Versus New-Generation Strategies in Combination with Adjuvants. Pharmaceutics. 2021;13(2):140. doi: https://doi.org/10.3390/pharmaceutics13020140
- Pulendran B, S. Arunachalam P, O’Hagan DT. Emerging concepts in the science of vaccine adjuvants. Nat Rev Drug Discov. 2021;20(6):454-475. doi: https://doi.org/10.1038/s41573-021-00163-y
- Banerjee K, Madhyastha R, Nakajima Y, Maruyama M, Madhyastha H. Nanoceutical Adjuvants as Wound Healing Material: Precepts and Prospects. Ijms. 2021;22(9):4748. doi: https://doi.org/10.3390/ijms22094748
- Njoroge GN, Kaibui IM, Njenga PK, Odhiambo PO. Utilisation of priority traditional medicinal plants and local people's knowledge on their conservation status in arid lands of Kenya (Mwingi District). J Ethnobiology Ethnomedicine. 2010;6(1). doi: https://doi.org/10.1186/1746-4269-6-22
- Willcox M, Diallo D, Sanogo R, Giani S, Graz B, Falquet J, et al. Intellectual property rights, benefit-sharing and development of “improved traditional medicines”: A new approach. Journal of Ethnopharmacology. 2015;176:281-285. doi: https://doi.org/10.1016/j.jep.2015.10.041
- Morgera E. Fair and Equitable Benefit-Sharing at the Cross-Roads of the Human Right to Science and International Biodiversity Law. Laws. 2015;4(4):803-831. doi: https://doi.org/10.3390/laws4040803
- Sinumvayo JP, Munezero PC, Tope AT, Adeyemo RO, Bale MI, Nyandwi JB, et al. Advancing Vaccinology Capacity: Education and Efforts in Vaccine Development and Manufacturing across Africa. Vaccines. 2024;12(7):741. doi: https://doi.org/10.3390/vaccines12070741
- Mukti BH. Ethnobotanical studies of medicinal plants in Borneo. Health Sciences International Journal. 2024 Aug 31;2(2):154–68. https://doi.org/10.71357/hsij.v2i2.41
- Sikombe S, Muleya F, Phiri J, Zulu S, Simasiku P, Situtu M. Key elements for promoting public-private partnerships in research and innovation. Cogent Business & Management. 2024;11(1). doi: https://doi.org/10.1080/23311975.2024.2401627
- Ma Z, Augustijn K, de Esch IJ, Bossink B. Collaborative university–industry R&D practices supporting the pharmaceutical innovation process: Insights from a bibliometric review. Drug Discovery Today. 2022;27(8):2333-2341. doi: https://doi.org/10.1016/j.drudis.2022.05.001
- Moorthy V, Abubakar I, Qadri F, Ogutu B, Zhang W, Reeder J, et al. The future of the global clinical trial ecosystem: a vision from the first WHO Global Clinical Trials Forum. The Lancet. 2024;403(10422):124-126. doi: https://doi.org/10.1016/s0140-6736(23)02798-8
- Shen J, Swift B, Mamelok R, Pine S, Sinclair J, Attar M. Design and Conduct Considerations for First‐in‐Human Trials. Clinical Translational Sci. 2018;12(1):6-19. doi: https://doi.org/10.1111/cts.12582
- Demir F, Albarri R, Unal DO. An Overview of Biotechnological Drug’s Various Techniques of Downstream Process, Guideline’s and Different Chromatographic Analysis. Cpa. 2024;20(8):729-742. doi: https://doi.org/10.2174/0115734129317408240903150800
- Cavaleri M, de Sousa CMA, Hacker A, Higgs ES, Lumpkin MM, Maia CS, et al. A roadmap for fostering timely regulatory and ethics approvals of international clinical trials in support of global health research systems. The Lancet Global Health. 2025;13(4):e769-e777. doi: https://doi.org/10.1016/s2214-109x(24)00515-1
- Sharma M, Grover M, Suryavanshi SJ, Sharma N, Shukla VK. Comparative Review of Clinical Trial Regulations in Different Countries: Current Scenario and Future Prospect. Rrct. 2025;20. doi: https://doi.org/10.2174/0115748871365320250529125930
- Van Norman GA. Data Safety and Monitoring Boards Should Be Required for Both Early- and Late-Phase Clinical Trials. JACC: Basic to Translational Science. 2021;6(11):887-896. doi: https://doi.org/10.1016/j.jacbts.2021.09.005
- Fikes J, Kapeghian J, Wojcinski ZW. Overview of Drug Development and Nonclinical Safety: Role of the Toxicologic Pathologist and Regulatory Aspects. Boca Raton: CRC Press; 2024. doi: https://doi.org/10.1201/9780429354861-2
