RESEARCH ARTICLE
In Vitro Antioxidant, Anti-Inflammatory, Anthelmintic, Anti-Microbial, FTIR and TLC Analysis of the Leaves of Memecylon amoenum
Academic Editor: Sanchaita Rajkhowa
Sciences of Phytochemistry|Vol. 5, Issue 1, pp. 88-96 (2026)
Received
Dec 11, 2025Revised
Sep 2, 2026Accepted
Feb 4, 2026Published
Apr 27, 2026
Abstract
Introduction
The concept that plants serve as valuable sources of biologically active compounds with therapeutic potential is widely accepted (1). Throughout history, plant-based remedies have been used across the world to manage numerous health conditions, including asthma, gastrointestinal disorders, skin diseases, respiratory and urinary complications, and cardiovascular and hepatic problems (2). Interest in plant-derived medicines often regarded as safer than synthetic drugs continue to grow rapidly (3). Medicinal plants, defined broadly as plant species used in herbal medicine due to their healing properties, form the backbone of traditional healthcare systems (4). Majority of people in countries like Ghana depend on these plants for their primary healthcare needs (5).
In Africa alone, about 70% of the population relies on traditional remedies (6). A World Health Organization (WHO) report likewise indicates that 80% of the global population outsource traditional medicine (7). Phytochemicals are naturally occurring bioactive compounds in plants that provide health benefits beyond those offered by essential nutrients (8). Many modern drugs originated from plant sources, reflecting the progression of conventional medicine from herbal traditions (9). Atropine also forms part of the antidiarrheal drug Lomotil when combined with diphenoxylate, a morphine derivative (10). Ghana possesses a vast diversity of medicinal plants capable of treating a wide range of conditions from minor ailments such as colds and digestive problems to chronic and infectious diseases such as hypertension, cancer, viral infections, schistosomiasis, and other parasitic diseases. Despite this wealth of resources, many of these plants have not yet been subjected to rigorous scientific investigation to document their phytochemical profiles, antioxidant, anti-inflammatory, and anthelmintic activities, and their therapeutic potential (11).
This study focuses on Memecylon amoenum, using the pulverized leaves of the plant for extraction and subsequent bioassays. The species grows along the Nkawkaw–Adawso road in Ghana’s Eastern Region and is locally known as “Sa.” Taxonomically, it belongs to the family Melastomataceae, genus Memecylon, species M. amoenum, within the sector Madagascar (Africa), kingdom Plantae, phylum Spermatophyta, class Magnoliopsida, and order Myrtales. Only limited scientific information is currently available on this plant, indicating a strong need for further research and detailed investigation. Traditionally, it has been used to treat various ailments, including wounds, diarrhoea, skin infections, and toothache (12). Traditional medicine remains an essential component of healthcare in many developing countries, shaped by local beliefs and cultural practices. Medicinal plants play a vital role in supporting this ideal, contributing significantly to the WHO’s objective of enabling people worldwide to lead healthy, productive, and sustainable lives (5).
Methodology
Collection and Identification of Plant Samples
In March 2022, leaves of M. amoenum were collected from Nkawkaw (Latitude: 6°33'4.36"N; Longitude: 0°45'58.32"E) in the Kwahu South District of the Eastern Region of Ghana. The plant specimen was identified and authenticated by Mr. Asare of the Department of Herbal Medicine, Faculty of Pharmacy and Pharmaceutical Sciences, Kwame Nkrumah University of Science and Technology.
Processing of the Plant Samples
The leaves were washed and left to air-dry at room temperature in the laboratory for 14 days. Once dried, they were ground into a fine powder, stored in a desiccator, and kept in the laboratory until needed for use.
Extraction of Plant Sample
The extraction of bioactive compounds was carried out using a sequential solvent extraction approach to capture constituents of varying polarities. A total of 100 g of pulverized M. amoenum leaves were carefully packed into a cellulose thimble and placed within a Soxhlet apparatus. The process was initiated by extracting the plant material with 700 mL of methanol for a duration of three days, followed by a subsequent extraction using 700 mL of petroleum ether. This sequential method ensures the systematic recovery of both polar and non-polar phytochemicals. After extraction, each crude extract was transferred to a rotary evaporator and concentrated under reduced pressure at a controlled temperature of 50 °C. The solvent was evaporated until the extracts reached a completely dry state, yielding the concentrated crude material used for the subsequent biological assays.
Phytochemical Screening
The dried sample and extracts were all subjected to phytochemical screening using standard protocols (13).
DPPH Assay
Concentrations of 100, 50, 25, 12.5 and 6.250 μg/mL extracts were made. DMSO was used to dissolve petroleum ether extract. A solution of 150 µL of the DPPH was added to 50 µL of the solution of the extracts in a microtiter plate. Ascorbic acid was used as the standard which was also prepared in the same concentrations as the extract. A solution of 150 µL of the DPPH was added to 50 µL of the solution of the standard in different wells on the same microtiter plate. Some of the wells of the microtiter plate were filled with only DPPH solution and distilled water was used as the control. The samples were in dark chamber for 30 min. The color changed from deep violet to light yellow. Absorbances were then taken at 517 nm with a microtiter plate reader. The results were recorded in IC50 (13).
Total Antioxidant Capacity Assay
Total Antioxidant Capacity (TAC) was conducted using the phosphomolybdenum method, which is based on the principle of antioxidant-driven reduction. In this assay, the plant extracts (methanol and petroleum ether) were combined with a specific reagent solution under acidic conditions to facilitate the reduction of Mo (VI) to Mo (V). This chemical reaction results in the formation of a characteristic green-to-blue phosphomolybdenum (V) complex. The intensity of this color, which reflects the antioxidant strength, was quantified by measuring the absorbance at 695 nm using a UV-visible spectrophotometer. To determine the final values, a calibration curve was constructed using ascorbic acid as a standard, allowing the results to be expressed as ascorbic acid equivalents (AAE) per weight of the extract (6).
Anthelmintic Activity
The In vitro anthelmintic assay was performed to evaluate the efficacy of the methanol and petroleum ether extracts against adult worms, using Mebendazole as a standard reference drug. The procedure involved exposing the worms to varying concentrations of the extracts (ranging from 0.375 mg/mL to 6 mg/mL) and monitoring their physiological responses. The anthelmintic potential was quantified based on two primary criteria: paralysis time, recorded when the worms showed no movement unless shaken vigorously, and death time, recorded when the worms exhibited no motility even after being dipped in warm water. (13).
Anti-inflammatory Assay
In separate test tubes, 0.2 mL of egg albumin solution, 2.8 mL of phosphate-buffered saline (PBS), and 2.0 mL of each extract or standard drug were mixed, with distilled water serving as the control. The mixtures were incubated at 37 °C for 15 min, followed by heating at 70 °C for 5 min. After cooling, the absorbance of each sample was measured at 660 nm, using Aspirin as the reference standard. The % inhibition of denaturation by the extracts and standard drugs were calculated using the Eq. 1.
Antimicrobal Activity Test
The antimicrobial activity of the extracts was evaluated using the broth dilution method to determine both the zones of inhibition and the Minimum Inhibitory Concentration (MIC). In this procedure, various concentrations of the methanol and petroleum ether extracts, alongside the reference antibiotic Ciprofloxacin, were tested against five different microorganisms.
The assay involved observing the inhibitory effects of these crude mixtures, with the methanol extract demonstrating the highest potency. The MIC, defined as the lowest concentration of the extract that completely inhibits visible microbial growth, was recorded and compared to the standard drug (6).
Thin Layer Chromatography (TLC)
Thin-layer chromatography (TLC) was conducted to identify the number of components in the petroleum ether and methanol crude extracts and to determine the optimal solvent system for separation. A fluorescent TLC plate (10 cm × 2 cm) was used, and different solvent systems were tested in the development tank, allowing each to saturate for about 5 min. Small amounts of the extracts were dissolved in their respective solvents and spotted 2 cm from the base of the plate. After drying, the plate was developed until the solvent had travelled 80–90% of its length, then removed and dried. The solvent front was marked, and the separated spots were visualized under UV light, measured, and their retardation factors (Rf) calculated. A solvent mixture of ethyl acetate and petroleum ether (1: 4) produced the clearest separation.
Results and Discussion
Percentage Yield
The percentage yield represents the overall efficiency of the extraction process and serves as a key indicator of the amount of crude extract recovered relative to the initial mass of the plant material used. Data regarding the specific percentage yields of M. amoenum extracts obtained across different solvent systems are comprehensively presented in Table 1.
| Crude extract | Mass (g) | % yield (%) |
|---|---|---|
| Methanolic extract | 26.708 | 16.385 |
| Petroleum ether extract | 18.231 | 10.128 |
Phytochemical Screening
The pulverized plant sample of the leaves and the two crude extracts were taken through various phytochemical screening tests to investigate the phytochemicals present in them. Out of thirteen phytochemicals tested for, the phytochemical screening revealed the presence of twelve, eleven, and seven secondary metabolites respectively for the pulverized sample, the methanol extract and the petroleum ether extract. The results are shown in Table 2.
| Phytochemicals | Pulverized Sample | Methanol Extract | Pet. Ether Extract |
|---|---|---|---|
| Alkaloids | + | + | + |
| Flavonoids | - | - | - |
| Phenols | + | + | - |
| Tannins | + | + | - |
| Terpenoids | + | + | - |
| Polyphenols | + | + | + |
| Steroids | + | + | + |
| Phytosterols | + | + | + |
| Saponins | + | + | - |
| Anthraquinones | + | + | - |
| Glycosides | + | + | + |
| Quinones | + | + | + |
| Carotenoids | + | - | + |
| Description: Key:(+) = presence of secondary metabolites, (-) = secondary metabolites not detected. | |||
The polarity of saponins because of their hydrophilic glycoside moieties (5) was affirmed in this study. In other words, saponins were absent in the non-polar solvent (petroleum ether) but present in the polar solvent (methanol), which may be because of the polarity of saponins and the rule of thumb ‘like dissolves like’. Tannins, phenols, terpenoids, alkaloids and flavonoids, according to literature, influence different types of biological activities, including antioxidant, antispasmodic, antifungal, antimicrobial, anthelmintic and anti-inflammatory effects. Phenols are represented by their anti-inflammatory, antioxidant, and other biological properties and can protect against oxidative pressure and some infections (14). Alkaloids such as isoquinoline, indole and diterpene are known to have good anti-inflammatory effects, for example bisbenzyl isoquinoline alkaloids, as stated in (15). Phytochemicals: tannins, phenols, steroids, and terpenoids, as found in crude extracts from the leaves of M. amoenum, indicate that the extract could serve as a potential remedy for cancer treatment, microbial infections, oxidative stress, helminthic infections and inflammatory disorder.
Antioxidant Activity
A lot of normal cellular metabolism in the body produces free radicals as by-products. These free radicals go on to cause extensive damage to fundamental molecular building blocks like DNA, proteins, and lipids in the human system. Damages to these blocks lead to diseases like cancer and cardiovascular disease. Hence these antioxidant assays seek to evaluate the strength of our crude extract to mop-up these free radicals.
DPPH Radical Scavenging Assay
The DPPH radical scavenging activity of the methanol and petroleum ether extracts of M. amoenum, along with the standard ascorbic acid, expressed as IC₅₀ values, is presented in Table 3.
| Samples | IC50 (ppm) |
|---|---|
| Standard (Ascorbic acid) | 58.46 ± 0.88 |
| Methanol crude extract | 11.42 ± 0.05 |
| Petroleum ether crude extract | 43.42 ± 0.62 |
DPPH is a free radical and purple in colour. Antioxidants reduce DPPH to DPPH-H by transfer of a hydrogen radical. This reduction changes the colour of the DPPH to pale yellow or whitish. The scavenging activity of the M. amoenum crude extract through the annihilation of the DPPH radicals was investigated. From the results of the DPPH scavenging assay, the IC50 values recorded were 11.42 ± 0.05 ppm for the methanol crude extract, 43.42 ± 0.62 ug/mL for the petroleum ether crude extract and 58.46 ± 0.88 ug/mL for the ascorbic acid. The extracts may contain compounds that neutralize free radicals by donating hydrogen atoms or electrons, leading to the formation of DPPH-H, a yellow-colored product. Among the two extracts, methanol have good activity than petroleum ether extracts due to the low IC50 value obtained.
Total Antioxidant Capacity
The results of total antioxidant capacity of the methanol and petroleum ether extracts have been presented in Table 4.
| Sample | TAC (gAAE/100g) |
|---|---|
| Methanol extract | 36.04 ± 1.43 |
| Petroleum Ether extract | 31.35 ± 3.98 |
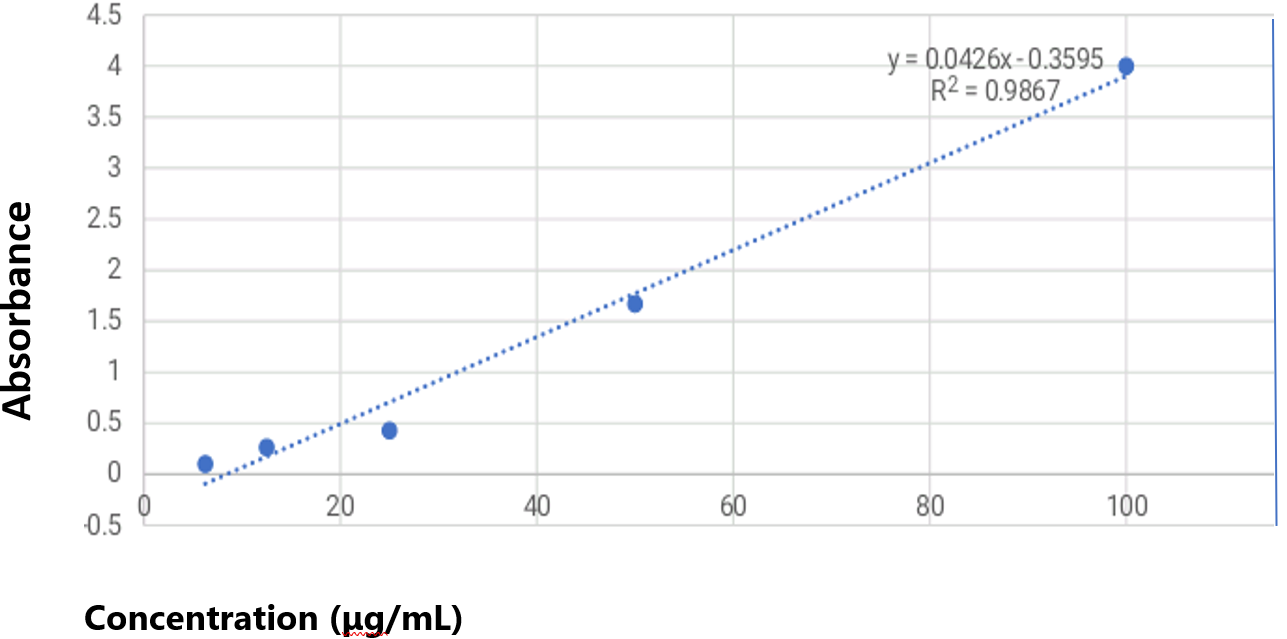
The total antioxidant capacity (TAC) assay quantifies the overall antioxidants present in plant extracts and was conducted using the phosphomolybdenum method, with ascorbic acid as the standard. The calibration curve constructed from the relationship between concentration and absorbance of ascorbic acid is presented in Figure 1. This assay relies on the reduction of Mo (VI) to Mo (V) by the extract, leading to the formation of a blue phosphomolybdenum (V) complex under acidic conditions (13) absorbance of the resulting complex was measured at 695 nm using a UV–visible spectrophotometer. The TAC values obtained were 36.04 ± 1.43 g for the methanol extract and 31.35 ± 3.98 g for the petroleum extract, indicating that 36.04 ± 1.43 g of the methanol extract and 31.35 ± 3.98 g of the petroleum extract exhibit antioxidant activity equivalent to ascorbic acid per 100 g of extract. Previous studies show that phytochemicals in medicinal plants, such as phenols (16) and terpenoids (17), possess strong antioxidant properties and may be beneficial in managing diseases associated with oxidative stress. Moreover, phenols and vitamins such as C and E are known to delay aging by neutralizing free radicals. These findings indicate that the antioxidant potential of this plant may be attributed to the presence of phytochemicals, including terpenoids and phenols, in its extracts.
Anti-inflammatory Activity
Protein (egg albumin) denaturation assay was used in showcasing the crude extracts of M. amoenum anti-inflammatory potentialand comparing the anti-inflammatory potency of the crude extracts of M. amoenum, against standard drugs aspirin. The results are depicted in Figure 2.
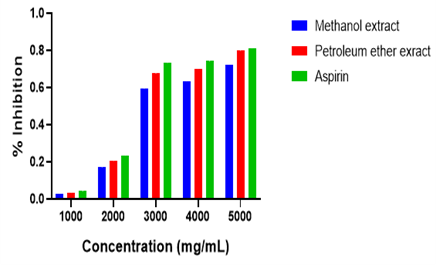
The crude extracts and standard anti-inflammatory drug (Aspirin) exhibited concentration-dose-dependent ability in preventing thermally induced protein denaturation. Thus, the higher the concentration of the test sample, the higher the mean % in protecting the egg albumin (protein) from denaturing. Among the crude extracts, petroleum ether recorded a higher potency, with better % inhibition at all concentrations compared to methanol extract and was closer to even the standard drug aspirin. This may be due to the difference in the phytoconstituents present in each extract. According to Soliman (18), much examinations have demonstrated that secondary metabolites present in different plants apply helpful effects for human wellbeing, for example, cardiovascular protection, as well as anti-inflammatory effects. As indicated by Magangana (19), Phenolic compounds are known for their notable pharmacological activities, particularly their anti-inflammatory effects. Flavonoids, condensed tannins, and gallotannins, which are different types of phenolics, can modulate molecular targets involved in inflammation, while saponins and terpenoids have also been shown to exhibit anti-inflammatory effects. (20). Therefore, the presence of phytoconstituents like terpenoids, phenolics, and tannins in the crude extracts may account for their anti-inflammatory potential.
Anthelminthic Activity
The adult worm mortality assay was employed in assessing the anthelminthic activity of the methanol and petroleum ether extracts as well as that of the standard drug, Mebendazole. In vitro anthelmintic assays allow for the rapid evaluation of plant extracts for potential anthelmintic activity and help elucidate the mechanisms through which their active compounds interact with parasites. The paralysis and death (mortality) time were used as the criterion for the anthelmintic activity. The results of the anthelminthic activities of the methanol and petroleum ether extracts are shown in Figure 3 and Figure 4.
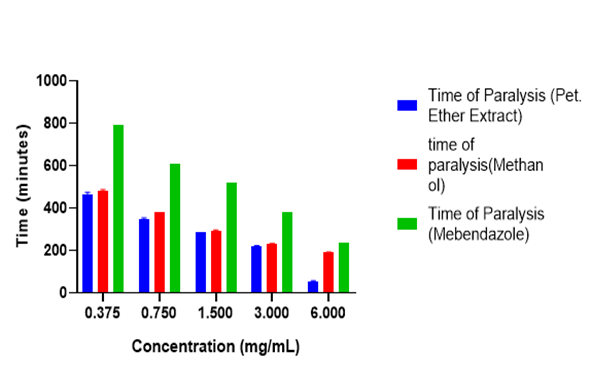
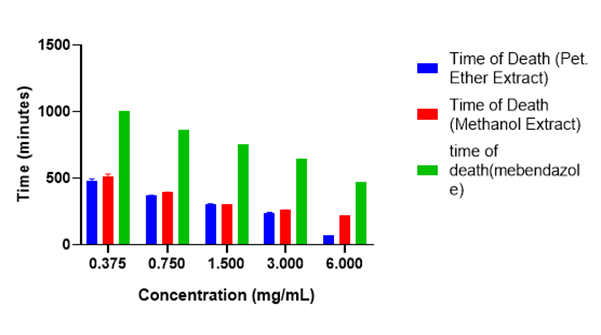
Shorter paralysis and death time signifies how powerful and promising an extract is as an anthelmintic agent. From the results obtained, petroleum ether crude extract at a concentration of 6 mg/mL gave a shorter paralysis and death time compared to the methanol extract and standard drug, mebendazole. Even at lower concentrations (3, 1.5, 0.750 and 0.375 mg/mL) the petroleum extract gave a shorter paralysis and death time as compared to the methanol extract and mebendazole. Petroleum ether extract exhibited anthelmintic activity because of the phytochemicals like tannins and terpenoids presents in the extracts. The petroleum ether showed high and remarkable anthelmintic activity compared to the standard drug, mebendazole because it contains different phytochemicals, and these phytoconstituents may work together in a synergistic manner. Also, the flukeworm is the right worm to use since it is the parasitic worm in the human body, but earthworm was rather used since it was the available worm we could get, and the earthworm also has the same physical characteristics compared to the ascaris (flukeworm).
Antimicrobial Activity
Broth dilution method was used to determine the zone of Inhibition and the Minimum Inhibitory Concentration (MIC) respectively. The minimum inhibitory concentrations of methanol and petroleum ether extracts of M. amoenum, along with ciprofloxacin as the reference drug against various microorganisms, are presented in Table 5.
| Organisms | Minimum inhibitory concentration (ug/mL) | ||
|---|---|---|---|
| Methanol Extract (ug/mL) | Pet. Ether Extract (ug/mL) | Ciprofloxacin (µg/mL) | |
| E. feacalis | 25 | 50 | 1.953 |
| S. typhi | 25 | 50 | 0.977 |
| S. aureus | 50 | 50 | 0.977 |
| P. aeruginosa | 25 | 25 | 1.953 |
| K. pneumoniae | 50 | 25 | 0.977 |
The assay showed that the two crude extracts of M. amoenum displayed varying inhibitory effects against the five tested microorganisms, with the methanol extract demonstrating the strongest activity. Unlike mebendazole, which are single compounds, the extracts are complex mixtures, meaning the observed effects could be due to one or more active constituents, while other compounds might mask or antagonize their activity. The higher MIC values of the extracts, compared to the lower MIC values of the reference drugs, may reflect the lower concentration of the active compounds within the crude mixtures. To achieve the full antimicrobial potential, isolation and purification of the active substance (s) may be necessary. Previous studies have shown that phytochemicals such as terpenoids, saponins, tannins and alkaloids (8) possess antimicrobial properties, suggesting that these constituents in M. amoenum may be responsible for the observed activity.
Thin Layer Chromatography
TLC was conducted to identify the number of components in the methanol and petroleum ether extracts and to select an appropriate solvent system for their separation. The technique separates compounds based on differences in polarity (21). Aluminium foil coated with a thin layer of silica gel served as the stationary phase, while a 1: 4 mixture of ethyl acetate and petroleum ether was used as the mobile phase. Silica gel consists of a silicon–oxygen network with surface hydroxyl groups, whose polarity allows them to form dipole–dipole interactions with polar compounds, facilitating separation (22). In carrying out the experiment, a faint line in pencil was drawn 1 cm from the bottom on the TLC plate directly on the silica as the baseline. Each of the crude extracts was dissolved in methanol a drop of it placed on the baseline with the help of a dropper. The drop was allowed to dry while the chamber containing the mobile phase was made to get saturated, for the development of the plate. The mobile phase rose up the TLC plate as the plate was placed in the chamber by capillary action, dragging the components of the mixture up with it. As the components moved up the plate, they reacted with the surface of the silica, causing them to move at different rates. Once the mobile phase travelled to almost the topmost part of the plate, the TLC plate was removed and the solvent front marked and allowed to dry. Visualization of the spots were done visibly and under UV light (both short and long). Figure 5 shows the visualization of the components of the extracts that travelled on the plate under visible, long UV light and short UV light.

Identified spots were marked and their respective distance travelled measured, together with solvent front. The retardation factor (RF) values were then determined and represented in table. Using the Eq. 2.
The calculated Rf values for the components of the methanol and petroleum ether extracts are presented in Table 6.
| Components | Methanol extract | Petroleum ether extract |
|---|---|---|
| A | 0.33 | 0.33 |
| B | 0.46 | 0.46 |
| C | 0.55 | 0.55 |
| D | 0.64 | 0.64 |
| E | - | 0.71 |
| F | - | 0.77 |
| G | - | 0.86 |
The methanolic crude extract showed four spots whereas the petroleum ether extract showed seven spots with Rf values between 0.33 to 0.86. Comparing the number of spots of the separated compounds and the phytochemicals recognized in the crude extracts, the spots were less than the phytochemicals indicated to be present. This could be because some components exist naturally as isomers or co-elute in mixtures.
Fourier Transform Infrared (FTIR) Spectroscopic Analysis
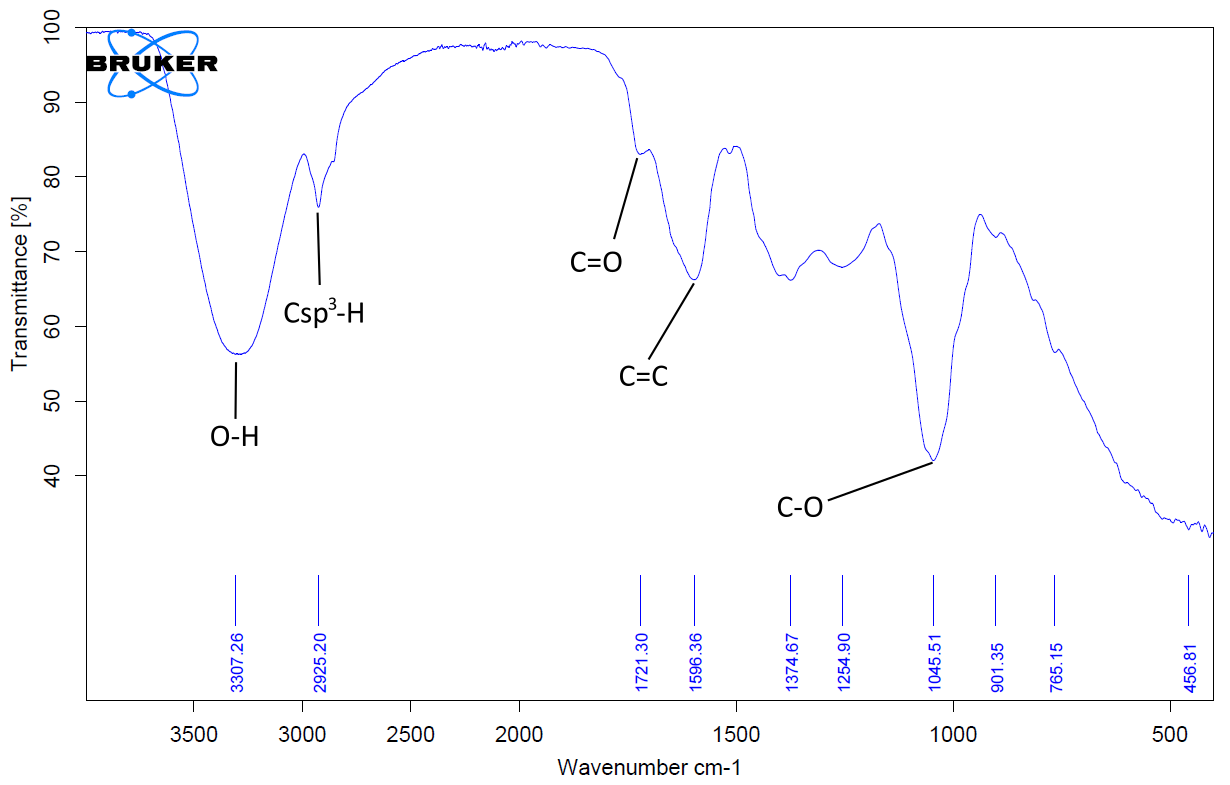
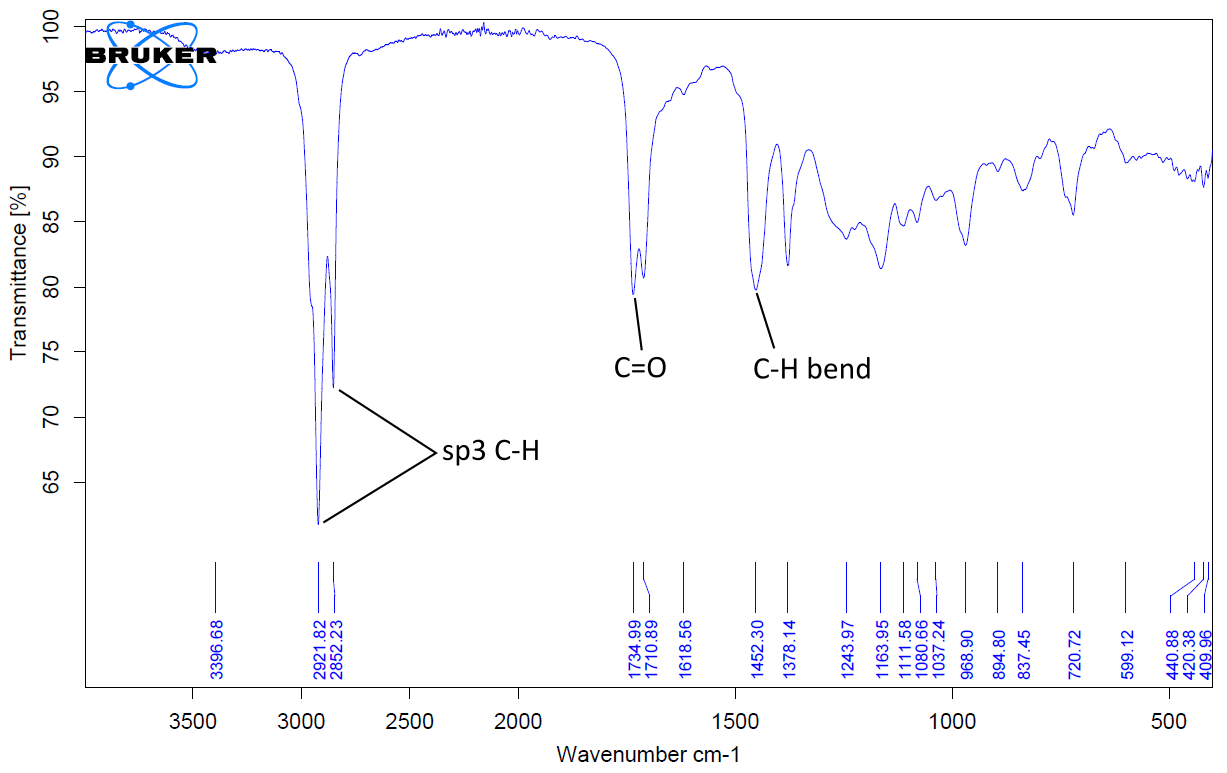
FTIR analysis was performed on both crude extracts. The results are depicted in Figure 6 and Figure 7, and Table 7. For the crude extracts, the wave numbers were around 3307.64 cm-1, 2925.20 cm-1, 1721.30 cm-1, 1696.36 cm-1 and 1045.51 cm-1 positions of the spectra are characteristic of alcohol (O-H), sp3C-H, carbonyl (C=O), and alkene stretch (C=C) respectively. These functional groups confirm the presence of saponins, steroids and terpenoids identified in the screening test conducted.
| Methanol crude extract | Petroleum ether crude extract | ||
|---|---|---|---|
| Functional group | Wavenumber (cm-1) | Functional group | Wavenumber (cm-1) |
| O-H | 3307.26 | sp3C-H | 2921.82 |
| sp3C-H | 2925.20 | C=O | 1710.89 |
| C=O | 1721.30 | C-H bend | 1378.14 |
| C=C | 1696.36 | - | - |
| C=O | 1045.51 | - | - |
Conclusion
Soxhlet extraction of M. amoenum using methanol and petroleum ether yielded appreciable percentages, with petroleum ether producing a slightly higher extraction yield under the applied conditions, suggesting differences in solvent polarity influencing compound recovery. Phytochemical screening revealed a wider range of bioactive constituents in the methanol extract compared to the petroleum ether extract, indicating the greater efficiency of methanol in extracting chemically diverse phytochemicals, particularly polar and semi-polar compounds. The extracts exhibited notable biological activities, including antimicrobial effects against both Gram-positive and Gram-negative bacteria, with MIC values ranging from 25 to 50 mg/mL, demonstrating moderate inhibitory potential against tested microbial strains. Additionally, significant antioxidant, anti-inflammatory, and anthelmintic properties were observed, further highlighting the therapeutic potential of the plant and supporting its pharmacological relevance in traditional medicine. FTIR spectroscopic analysis confirmed the presence of key functional groups corresponding to the identified phytoconstituents, providing molecular-level evidence of the detected compounds, while thin-layer chromatography demonstrated the presence of multiple well-separated compounds within the extracts, indicating chemical complexity and diversity. Overall, this work authenticates and scientifically substantiates the ethnobotanical use of the plant by indigenous communities for the management of various ailments and provides a foundation for future phytochemical isolation and pharmacological investigations.
Declarations
Acknowledgment
The authors would like to acknowledge Stepanie Adu Amoako of the microbiology laboratory of the Pharmaceutics Department, and the Organic Chemistry laboratory of the Department of Chemistry, KNUST. We would also like to acknowledge the technicians of the KNUST Central laboratory for the immense role they played in the research.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
All the data has been included in the manuscript.
Ethics Statement
This study did not involve human participants or animal subjects, and no ethical approval was required.
Funding Information
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
References
- Dar RA, Shahnawaz M, Ahanger MA, Majid IU. Exploring the diverse bioactive compounds from medicinal plants: a review. J Phytopharm. 2023; 12 (3): 189-195.
- Cousins D, Huffman MA. Medicinal properties in diet an ethnopharmacological evaluation. Afr Study Monogr. 2002; 23: 65–68.
- Nasim N, Sandeep IS, Mohanty S. Plant-derived natural products for drug discovery: current approaches and prospects. Nucleus. 2022; 65 (3): 399-411.
- Manisha DRB, Begam AM, Chahal KS, Ashok MA. Medicinal plants: traditional uses and modern applications. J Neonatal Surg. 2025; 14 (3).
- Singh R. Medicinal plants: a review. J Plant Sci. 2015; 3 (1): 50-55.
- Acheampong A, Buah P, Frimpong SO, Tima L, Sarfo-Mainoo AOB, Mensah W, Odei J. In vitro anti-helminthic, antimicrobial, and antioxidant activity of methanolic extract of Aspilia silphioides leaves. J Pharmacogn Phytochem. 2024; 13 (2): 151-161.
- Pathania S, Ramakrishnan SM, Bagler G. Phytochemica: a platform to explore phytochemicals of medicinal plants. Database (Oxford). 2015; 2015: bav075.
- Kurmukov AG. Phytochemistry of medicinal plants. In: Medicinal plants of Central Asia: Uzbekistan and Kyrgyzstan. 2013. p. 13-14.
- Buah P, Odei J, Oti-Boakye A, Sarfo-Mainoo LT, Acheampong A, Baah KA, et al. Phytochemical screening and investigation of pharmacological properties of ethanol extract of the leaves of Justicia pectoralis.
- Ginsburg CM, Angle CR. Diphenoxylate-atropine (Lomotil) poisoning. Clin Toxicol. 1969; 2 (4): 377-382.
- Jamtsho T, Loukas A, Wangchuk P. Pharmaceutical potential of remedial plants and helminths for treating inflammatory bowel disease. Pharmaceuticals. 2024; 17 (7): 819.
- Amaranathan S, Murugesan A, Govindharaj A, Yogeshwaran A, Perumal A. Taximetric studies of the genus Memecylon L. and their phytoconstituents. J Res Siddha Med. 2023; 6 (2): 52-57.
- Buah P, Acheampong A, Oti-Boakye A, Ofori S, Frimpong WM, Odei J, Sarfo-Mainoo LT. In vitro antimicrobial, antioxidant, and anti-helminthic activities and GC-MS analysis of fractions of the leaves of Aspilia silphioides. J Pharmacogn Phytochem. 2024; 13 (5): 201-210.
- Ogidi OI, Emaikwu NG. Plant phenolic compound isolation and bioinformatics approaches. In: Computational approaches in biotechnology and bioinformatics. Boca Raton: CRC Press; 2024. p. 188-215.
- Wang R, Mao Y, Yu C, Rong Z, Wang R, Wang Y, et al. Natural products with activity against nonalcoholic steatohepatitis. Mini Rev Med Chem. 2024; 24 (21): 1894-1929.
- Jan R, Asaf S, Numan M, Lubna, Kim KM. Plant secondary metabolite biosynthesis under stress conditions. Agronomy. 2021; 11 (5): 968.
- Kolesnikova AI, Kayumov AR, Gilfanov IR, Frolova LL, Nikitina LE, Trizna EY. Antimicrobial activity of terpenes against Staphylococcus aureus. Microbiology. 2024; 93 (3): 385-388.
- Soliman SA, Khaleil MM, Metwally RA. Antifungal activity of Bacillus species and their metabolites. Biology. 2022; 11 (10): 1390.
- Magangana TP, Makunga NP, La Grange C, Stander MA, Fawole OA, Opara UL. Bioactive compounds and antioxidant activity of pomegranate peel extracts. Antioxidants. 2021; 10 (7): 1119.
- Chauke S. Antimicrobial properties and phytochemical analysis of medicinal plants used for ear infections [dissertation]. 2023.
- Dyguła P, Kucharska K, Kamiński M. Composition analysis of lubricating oils using chromatography. J Chromatogr B. 2022; 1192: 123137.
- Liu H, Wang Y, Xue X, Liu Y, Chen P, Wang P, Yin SF. Hydrogen bonding interactions in polymers for photocatalysis. J Colloid Interface Sci. 2024; 669: 393-401.