REVIEW
Analgesic Mechanisms of Lawsone (2-hydroxy-1, 4-naphthoquinone) from Lawsonia inermis: A Comprehensive Review of Cyclooxygenase Modulation and Other Molecular Pain Targets
Sciences of Pharmacy|Vol. 5, Issue 2, pp. 162-168 (2026)
Received
Jan 2, 2026Revised
Feb 5, 2026Accepted
Feb 17, 2026Published
Apr 28, 2026
Abstract
Introduction
The management of chronic pain and inflammation, particularly in autoimmune multisystem diseases such as rheumatoid arthritis (RA), remains one of the most significant global health burdens, contributing substantially to disability, impaired productivity, and diminished quality of life (1, 2). A critical feature of inflammatory nociception is the upregulation of cyclooxygenase-2 (COX-2), which catalyzes the synthesis of prostaglandin E2 (PGE2), a major mediator of peripheral and central sensitization (3, 4). Conventional non-steroidal anti-inflammatory drugs (NSAIDs) primarily exert their therapeutic effect by inhibiting the COX enzyme, thereby preventing the biosynthesis of PGE2 (1). However, their long-term use is associated with considerable systemic risks, including concerns over gastrointestinal, cardiovascular, and renal (1, 5). Inflammation is further perpetuated by a cascade of molecular signaling events involving key transcription factors and signaling molecules, including increased COX-2 activity (induced by cytokines) and downstream production of PGE2 (3–5). Furthermore, the production of Reactive Oxygen Species (ROS) and Nitric Oxide (NO) is well known to induce inflammation (2, 4–6). The complexity of these intertwined inflammatory pathways has intensified interest in bioactive natural compounds that offer safer pharmacological profiles while modulating key inflammatory pathways.
Natural products offer a rich reservoir of bioactive molecules suitable for new drug development (5–7). Among these, Lawsonia inermis, traditionally used for medicinal and cosmetic purposes, contains lawsone (2-hydroxy-1, 4-naphthoquinone) as its principal bioactive naphthoquinone (1, 3, 8, 9). Historically, L. inermis has been extensively used in traditional medicine for its analgesic, anti-inflammatory, and antipyretic properties (3, 10). Lawsone has demonstrated significant in vivo efficacy, with analgesic and anti-inflammatory effects that are equipotent or even more potent than those of aspirin in preclinical models (11). Critically, lawsone has also shown a favorable safety profile concerning hepatotoxicity and nephrotoxicity at effective anti-arthritic doses, unlike many NSAIDs (1). Lawsone is classified as a natural 1, 4-naphthoquinone derivative (3, 10). A clear delineation of lawsone’s analgesic mechanisms is essential to establish its clinical value, guide rational drug development, and validate its suitability as a safer therapeutic option for chronic pain conditions.
While studies confirm lawsone's potential, as evidenced by its significant reduction in serum PGE2 levels (indicating COX-2 modulation) and the suppression of the inflammatory transcription factor NF-κB and associated cytokines (TNF-α, IL-1β, IL-6) (1), a comprehensive understanding of its multi-target action is still emerging. Specifically, the involvement of lawsone with novel pain targets needs systematic validation. The P2X7 purinergic receptor (P2X7R), a ligand-gated ion channel activated by ATP, has gained attention as a critical therapeutic target in chronic inflammation and neuropathic pain (5–7). Given that 1, 4-naphthoquinone has been identified as a valuable molecular scaffold for the development of potent P2X7R antagonists (4–6), this comprehensive review aims to synthesize and critically evaluate existing pharmacological and in silico data on isolated lawsone and related naphthoquinone analogues. By specifically integrating evidence supporting lawsone's modulation of COX-2, NF-κB, and the hypothetical targeting of P2X7R, this review aims to provide a comprehensive evaluation of lawsone's analgesic potential by integrating evidence from studies on L. inermis, pure lawsone, and related naphthoquinone derivatives.
Methods
Search Strategy and Data Sources
A literature search was conducted to provide a comprehensive narrative review, focusing on establishing the direct molecular mechanisms of lawsone. Searches were conducted across key scientific databases, including PubMed, ScienceDirect, and Google Scholar. The search scope prioritized publications from 2020 onwards to ensure the synthesis of the most contemporary mechanistic insights, complementing earlier foundational studies.
The search strategy employed a combination of terms addressing the compound's identity, source, therapeutic effects, and hypothesized molecular targets. The comprehensive keywords utilized were: "lawsone", "lawsonia inermis", "2-hydroxy-1, 4-naphthoquinone", "analgesic", "anti-inflammatory", "COX-2", "PGE2", "NF-κB", "P2X7R", "naphthoquinone", and "LOX (Lipoxygenase)", applied individually and in Boolean combinations to maximize relevant results.
Inclusion and Exclusion Criteria
The selection process applied strict criteria to maintain focus on the core mechanistic hypothesis concerning isolated lawsone and its structural analogues:
Inclusion Criteria: Studies were included only if they investigated pure, isolated lawsone (2-hydroxy-1, 4-naphthoquinone) or structurally relevant naphthoquinone derivatives. Primary preference was given to studies that provided explicit mechanistic data, including the quantification or direct analysis of molecular targets such as PGE2, COX-2, NF-κB, or the direct functional inhibition of P2X7R. Studies confirming the 1, 4-naphthoquinone core as a potential scaffold for P2X7R antagonism were also included to underscore the review's key novelty.
Exclusion Criteria: Studies focused primarily on crude extracts of Lawsonia inermis, purely descriptive reviews, or those lacking quantitative pharmacological mechanisms were excluded. Articles that were inaccessible as full text or deemed irrelevant to the anti-inflammatory or analgesic pathways were also filtered out.
Data Selection and Synthesis
The initial search yielded approximately 186 entries, which were then preliminarily screened for titles and abstracts. A systematic filtering process, applying the established inclusion criteria, resulted in the selection of 14 accessible, highly relevant publications. These selected studies constituted the exclusive evidentiary basis for the qualitative synthesis and discussion presented in the subsequent sections of this review. The overall literature screening process, including the stages of identification, screening, eligibility assessment, and the specific reasons for exclusion at each stage, is summarized in Figure 1.
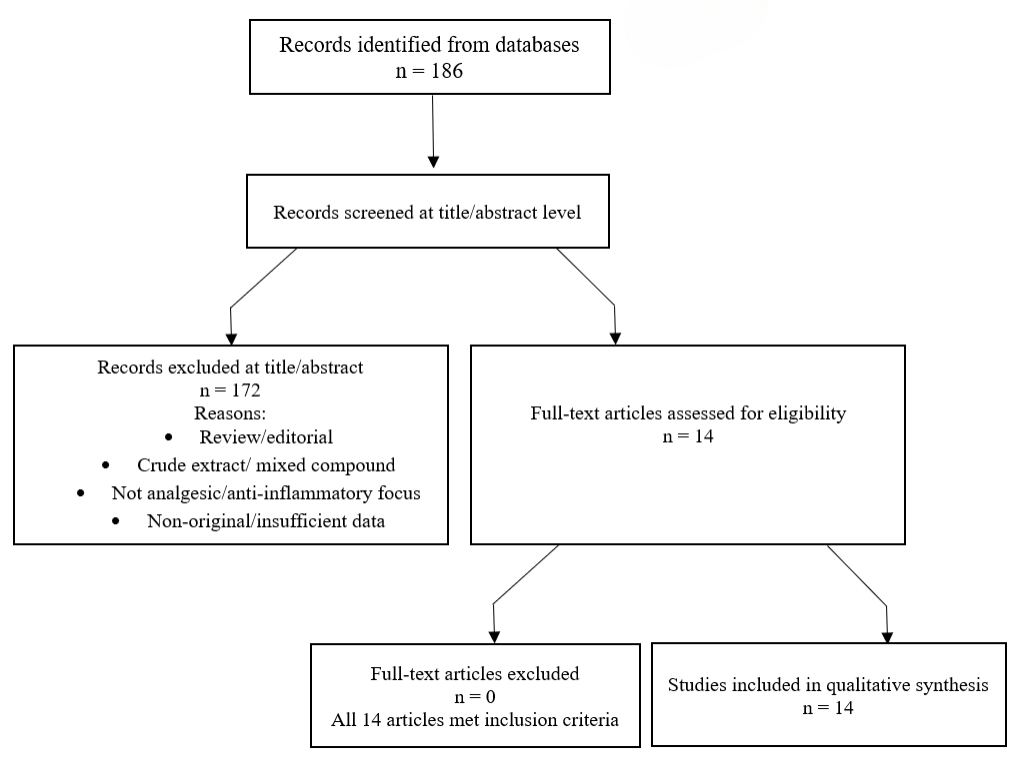
Results and Discussion
To enhance conceptual clarity and avoid over-interpretation, this section adopts an evidence-graded narrative. Direct evidence refers to studies evaluating lawsone itself in biological models with measurable changes in analgesic/inflammatory endpoints and pathway readouts (notably the COX-2/PGE2 axis and NF-κB–associated cytokine signalling). Indirect evidence refers to mechanistic plausibility inferred from structurally related 1, 4-naphthoquinone analoguesand computational/pharmacological studies, particularly those suggesting potential P2X7 receptor (P2X7R) involvement. Accordingly, the discussion first synthesises direct lawsone evidence, followed by indirect (hypothesis-generating) evidence.
In Vivo Analgesic and Anti-inflammatory Efficacy of Lawsone
The pharmacological characterisation of isolated lawsone (2-hydroxy-1, 4-naphthoquinone) confirms its traditional application by demonstrating significant anti-inflammatory and analgesic efficacy in established preclinical models (11). In the acute pain assessment model using the hot-plate test, oral administration of lawsone (80 mg/kg) showed a more potent analgesic effect (p < 0.1) than the reference drug, aspirin (200 mg/kg) (11). In models of acute inflammation, specifically carrageenan-induced hind paw edema, lawsone (80 mg/kg) demonstrated an anti-inflammatory effect equipotent to that of aspirin (200 mg/kg) (11).
Furthermore, in the chronic inflammation model of Freund's Complete Adjuvant (FCA)-induced rheumatoid arthritis (RA) in Sprague-Dawley rats, lawsone (100 and 200 mg/kg) significantly inhibited arthritic progression, ameliorating paw edema and improving haematological markers (1). This efficacy was validated by histopathological evaluation, which showed attenuation of the arthritic score comparable to that of the reference drug, piroxicam (1). Lawsone treatment also improved hepatic function markers (ALT and AST) and showed no signs of nephrotoxicity in the tested doses, suggesting a favourable safety profile compared to conventional NSAIDs (1).
Modulation of Prostaglandin Synthesis and COX-2 Pathway
A primary mechanism underlying the anti-inflammatory activity of lawsone involves the modulation of the cyclooxygenase (COX) pathway, leading to the inhibition of prostaglandin production (11). Consistent with its efficacy in edema models, lawsone administration in FCA-induced RA rats led to a significant reduction in serum PGE2 levels (p < 0.001) across the treated groups compared to the arthritic control (1). PGE2 is a central lipid mediator derived primarily from COX-2 activity, driving inflammation and acute pain (1, 5). This reduction in PGE2 strongly suggests that lawsone acts as a potent modulator or inhibitor of COX-2 (1).
Structural analogues of lawsone, a 1, 4-naphthoquinone, further support this enzyme-inhibition hypothesis (12). Synthetic thioglucoside derivatives (U-286 and U-548) demonstrated direct inhibitory activity against the COX-2 enzyme activity in LPS-stimulated RAW 264.7 macrophage cells (5). Specifically, the compound U-286 significantly inhibited COX-2 activity by 82.5% at 1 mM concentrations. This effect is physiologically relevant, as COX-2 activity is increased by the release of the pro-inflammatory cytokine IL-1b, suggesting a complex upstream and downstream regulatory network involving COX-2 and purinergic signaling (5).
Suppression of the NF-κB Pathway and Inflammatory Cytokines
Lawsone exhibits potent anti-inflammatory activity by directly modulating key regulatory pathways in immune cells. In the FCA-induced RA model, lawsone treatment significantly suppressed mRNA expression of the master inflammatory transcription factor, Nuclear Factor-Kappa B (NF-κB) (p < 0.001) (1). Activation of NF-κB is central to inflammation, as it triggers the transcription and expression of primary pro-inflammatory mediators (1).
Downstream effects confirmed that lawsone effectively minimized the expression levels of several NF-κB target genes, including pro-inflammatory cytokines such as Tumor Necrosis Factor-alpha (TNF-a), interleukin-1 beta (IL-1b), and interleukin-6 (IL-6) (1), and matrix metalloproteinases and angiogenesis factors such as matrix metalloproteinase (MMP)-2, MMP-3, and vascular endothelial growth factor (VEGF) were significantly downregulated, inhibiting the processes of joint destruction and synovial angiogenesis characteristic of severe RA (1).
Furthermore, computational modelling provides insight into how lawsone may directly interact with these inflammatory mediators in silico docking studies predicted lawsone’s capacity to bind key cytokines, TNF-a and IL-1a. TNF-a, lawsone demonstrated the ability to bind to TNF-a with a predicted binding affinity of -4.7 kcal/mol. Molecular dynamics simulation suggested that lawsone inhibits TNF-a activity without disrupting the protein’s structural integrity (8). IL-1a, in binding models targeting the skin epidermal layer, lawsone demonstrated a high binding affinity of -5.2 kcal/mol to the pro-inflammatory cytokine IL-1a (10). Critical analysis of the binding pose confirmed interactions with key residues, notably Asp65 and Ile68, validating lawsone’s capacity to interfere with IL-1a signaling (10).
The concurrent reduction of COX-2/PGE₂ output and NF-κB signalling supports a single integrated inflammatory circuit in which NF-κB can reinforce COX-2 expression and downstream PGE₂-driven inflammatory tone. In this context, the proposed P2X7R interaction should be framed strictly as a hypothesis-generating upstream node supported only by scaffold based analogy that may converge on NF-κB and COX-2/PGE₂, pending receptor level validation.
Evidence for P2X7 Receptor (P2X7R) Antagonism through Structural Analogy
The efficacy and multi-target nature of lawsone are strongly supported by pharmacological evidence obtained from its naphthoquinone analogues targeting the P2X7 purinergic receptor (P2X7R), an important therapeutic target in chronic inflammation and neuropathic pain (4, 6, 13). Lawsone is a natural 1, 4-naphthoquinone derivative, a chemical scaffold widely recognized for its potential in developing P2X7R antagonists (4, 6, 7).
Studies on synthetic 1, 4-naphthoquinone analogues demonstrate potent P2X7R inhibitory activities:
(i) Pore and ion channel blockade: Derivatives, such as the thioglucoside conjugates (U-286, U-548, U-556, U-557), significantly blocked ATP-induced calcium influx (Ca2+) in neuroblastoma and macrophage cells (4, 5, 14). These compounds also inhibited ATP-induced macropore formation (as measured by EtBr or YO-PRO-1 dye uptake) (4, 14). Crucially, the analogue U-556 was shown to almost completely suppress dye uptake induced by the specific P2X7R agonist, BzATP, indicating a complete blockade of receptor function under these specific conditions (4).
(ii) In vivo efficacy and mechanisms: The naphthoquinone derivative NSA15, which contains a juglone (5-hydroxy-1, 4-naphthelenedione) moiety, demonstrated anti-inflammatory activity in vivo by reducing ATP-induced paw edema. Pretreatment with NSA15 also inhibited carrageenan-induced peritonitis, suggesting that its anti-inflammatory action is partially mediated through P2X7R inhibition (6).
(iii) Molecular docking confirmation: molecular modelling consistently predicts that naphthoquinones, including NSA15 and U-556, bind to the allosteric binding site of the P2X7R, located in the extracellular domain between subunits (4, 6). This binding mode suggests that they inhibit receptor function without competing directly with ATP, offering a distinct pharmacological advantage (7). The fact that lawsone shares the core 1, 4-naphthoquinone structure strongly supports the hypothesis that it, too, modulates this key receptor in a multi-target fashion.
To improve clarity and address concerns regarding evidence grading, the findings are organized into two summary tables that explicitly distinguish direct, lawsone-specific experimental evidence from indirect, hypothesis-supporting evidence. Table 1 presents the preclinical studies in which lawsone was directly tested and reports outcomes and mechanistic readouts that are experimentally substantiated, particularly along the COX-2/PGE2 and NF-κB/cytokine axes.
| Evidence domain | Model / system | Dose (route) | Direct mechanistic readouts | Main finding (condensed) |
|---|---|---|---|---|
| Acute analgesic effect | Hot plate test (rat) | 80 mg/kg (oral) | Behavioural analgesia | Analgesic effect stronger than aspirin 200 mg/kg (reported p < 0.1)(11) |
| Acute anti-inflammatory effect | Carrageenan-induced paw edema | 80 mg/kg | Paw edema (inflammation outcome) | Anti-inflammatory effect comparable to aspirin (11) |
| Anti-arthritic efficacy | FCA-induced arthritis (rat) | 100–200 mg/kg | Paw edema / arthritis severity outcomes | Reduced paw edema; efficacy comparable to piroxicam (1) |
| Safety / tolerability at effective doses | Hepatic/renal markers (in vivo) | - | ALT/AST + renal indicators | No hepatotoxicity/nephrotoxicity signals; markers improved/maintained at effective doses (1) |
| Prostaglandin axis (COX-2 → PGE2) | FCA-induced RA (in vivo) | - | Serum PGE2 | Significant PGE2 reduction (reported p < 0.001), consistent with COX-2 pathway modulation (1) |
| NF-κB inflammatory signalling | RA rats (gene expression/markers) | - | NF-κB + TNF-α, IL-1β, IL-6 | Suppressed NF-κB signalling with downstream cytokine reductions (1) |
| Joint destruction / angiogenesis markers | FCA model (in vivo) | - | MMP-2, MMP-3, VEGF | Downregulated tissue-destructive and angiogenic markers, supporting disease-modifying potential (1) |
In contrast to the direct evidence summarized in Table 1, Table 2 compiles evidence that is indirect and hypothesis-generating, derived primarily from structurally related 1, 4-naphthoquinone analogues and in silico analyses. This table is included to provide mechanistic plausibility especially for the proposed P2X7R axis while explicitly acknowledging that these data do not constitute direct validation in lawsone and therefore require dedicated receptor-level and protein-level confirmation.
| Evidence source | Representative compound (s) | Model / system | Indirect mechanistic signal (condensed) | Implication (hypothesis-level) |
|---|---|---|---|---|
| In vitro (analogue) | U-286 | RAW 264.7 macrophages (1 µM) | 82.5% COX-2 inhibition | Supports that the 1, 4-naphthoquinone scaffold can engage COX-2–related anti-inflammatory targets (scaffold plausibility; not lawsone-specific)(4) |
| In vitro (analogue; P2X7R function) | U-286, U-548, U-556, U-557 | Macrophages / neuroblastoma | Blocked ATP-induced Ca²⁺ influx; inhibited macropore formation | Provides a functional precedent that related naphthoquinones can antagonise P2X7R (supports plausibility of P2X7R hypothesis for lawsone)(5, 13) |
| In vivo (analogue; P2X7R-mediated inflammation) | NSA15 | ATP-induced edema; peritonitis | Reduced ATP-driven inflammation | Supports that P2X7R pathway modulation can translate to in vivo anti-inflammatory effects (still indirect for lawsone)(6) |
| In silico (analogue; P2X7R binding) | NSA15, U-556 | P2X7 receptor model | Stable allosteric binding at the P2X7R pocket | Strengthens the structural rationale for possible P2X7R antagonism within the scaffold family (requires experimental confirmation for lawsone)(4, 6) |
| In silico (lawsone; cytokine docking) | Lawsone | Molecular docking | TNF-α binding affinity –4.7 kcal/mol | Suggests potential interaction with inflammatory mediators, but remains computational (supportive, not confirmatory)(8) |
| In silico (lawsone; cytokine docking) | Lawsone | Molecular docking | IL-1α binding affinity –5.2 kcal/mol | Same as above hypothesis-supporting only; needs wet-lab validation (10) |
Lawsone’s proposed analgesic mechanism involves coordinated modulation of inflammatory mediators, linking COX-2/PGE₂ suppression with NF-κB-dependent cytokine downregulation. The inclusion of P2X7R antagonism reflects a hypothesis supported by structural and pharmacological data from related naphthoquinones rather than direct experimental validation, as illustrated in Figure 2.
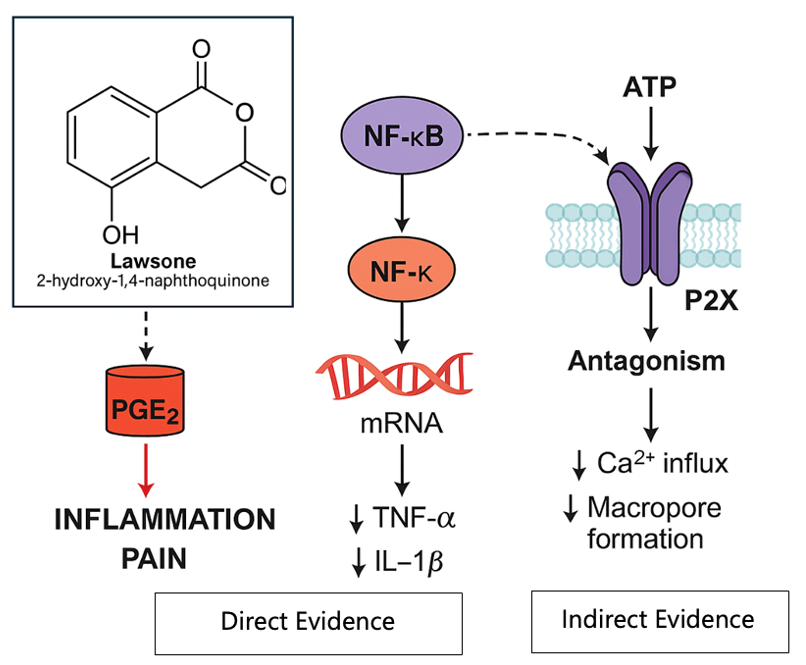
Although the preclinical literature consistently supports modulation of the COX-2/PGE2 axis and NF-κB–dependent cytokine signalling by lawsone, the overall evidence base is derived from a limited number of experimental studies, with heterogeneity in models, dosing, and outcome reporting. Importantly, P2X7R involvement is currently inferred primarily from structural analogy and data on related 1, 4-naphthoquinone derivatives rather than direct functional assays of isolated lawsone. Future work should therefore prioritise (i) direct P2X7R pharmacology for lawsone, (ii) protein-level confirmation of NF-κB/cytokine changes, and (iii) standardised reporting of efficacy and safety outcomes to strengthen translational interpretation.
Conclusion
This narrative review demonstrates that lawsone possesses significant analgesic and anti-inflammatory activity supported by robust in vivo evidence, primarily through modulation of the COX-2/PGE₂ axis and suppression of NF-κB-driven cytokine signaling. Beyond these established mechanisms, the 1, 4-naphthoquinone core of lawsone provides a strong structural rationale for a potential interaction with the P2X7 purinergic receptor, inferred from pharmacological and computational studies on structurally related analogues rather than direct experimental validation. Collectively, these findings position lawsone as a promising preclinical lead and molecular scaffold, highlighting its potential for multi-target analgesic drug development while underscoring the need for direct mechanistic and translational studies.
Future Directions and Perspectives
While the current synthesis strongly supports lawsone’s potent multi-target activity against inflammatory and pain markers, several critical research avenues must be pursued to establish its clinical viability. A fundamental step is to move beyond preclinical efficacy and rigorously validate its overall safety profile. Specifically, future investigations must prioritise well-controlled clinical studies to verify both the systemic safety and therapeutic efficacy of lawsone for chronic usage in humans. Comprehensive safety profiling, including acute, chronic, and cumulative toxicity studies, is necessary. Crucially, research is warranted to assess teratogenicity and potential drug-drug interactions before lawsone can transition safely into formalised clinical trials. Furthermore, extensive pharmacokinetic and pharmacodynamic (PK/PD) studies are required to determine the optimal dose and route of administration accurately, and to enhance understanding of lawsone’s bioavailability. Mechanistically, definitive proof of lawsone’s multi-target activity demands several focused studies. To confirm the suppression cascade inferred from mRNA data, future work must determine the protein expression levels of key inflammatory mediators, such as TNF-a, IL-1b, IL-6, and NF-κB, ideally using techniques such as Western Blotting to provide validation at the translational level. Regarding the central hypothesis concerning P2X7R, the structural analogy requires pharmacological validation using isolated lawsone, not just its derivatives; specifically, further in vitro investigation should precisely evaluate lawsone's selectivity for purinergic receptors using transgenic cell lines that stably express P2X7R. Moreover, targeted studies should investigate the influence of lawsone on critical downstream signaling pathways implicated in chronic inflammatory diseases, including the JAK/STAT, MAPK, and AMPK pathways. Finally, lawsone's inherent hydrophobicity contributes to low water solubility and limited availability, thereby limiting its clinical effectiveness. To overcome this significant pharmaceutical challenge, future research should explore advanced drug delivery designs. Promising strategies include the use of nanotechnology, such as encapsulating lawsone in innovative carriers, such as cell-penetrating peptide-conjugated cationic liposomes. Such formulation strategies could enhance lawsone’s stability, improve targeted delivery to inflamed tissues, and effectively bypass current pharmacokinetic limitations, paving the way for lawsone’s development as a safe and effective multi-target analgesic agent.
Abbreviations
COX-2: cyclooxygenase-2; PGE₂: prostaglandin E₂; NF-κB: nuclear factor kappa B; P2X7R: P2X7 purinergic receptor; TNF-α: tumor necrosis factor-alpha; IL: interleukin; MMP: matrix metalloproteinase; VEGF: vascular endothelial growth factor.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
All data generated or analyzed during this study are included in this published article.
Ethics Statement
Ethical approval was not required for this study.
Funding Information
Not applicable.
References
- Sattar S, Shabbir A, Shahzad M, Akhtar T, Anjum SM, Bourhia M, et al. Evaluation of anti-inflammatory and immunomodulatory potential of Lawsone (2-hydroxy-1,4-naphthoquinone) using pre-clinical rodent model of rheumatoid arthritis. Front. Pharmacol. 2023;14. doi: https://doi.org/10.3389/fphar.2023.1279215
- Hidayat NN, Musfiroh I. Lawsonia inermis: Review of Its Anti-inflammatory Properties and Therapeutic Potential. Ijpst. 2025;12:81-89. doi: https://doi.org/10.24198/ijpst.v12s2.58785
- Kumar K, Kumar P, Ahmed S. Isolation and biological properties of lawsone: a review. Ijpsm. 2024;9(4):13-25. doi: https://doi.org/10.47760/ijpsm.2024.v09i04.002
- Kozlovskiy S, Pislyagin E, Menchinskaya E, Chingizova E, Kaluzhskiy L, Ivanov AS, et al. Tetracyclic 1,4-Naphthoquinone Thioglucoside Conjugate U-556 Blocks the Purinergic P2X7 Receptor in Macrophages and Exhibits Anti-Inflammatory Activity In Vivo. Ijms. 2023;24(15):12370. doi: https://doi.org/10.3390/ijms241512370
- Kozlovskiy SA, Pislyagin EA, Menchinskaya ES, Chingizova EA, Sabutski YE, Polonik SG, et al. Anti-Inflammatory Activity of 1,4-Naphthoquinones Blocking P2X7 Purinergic Receptors in RAW 264.7 Macrophage Cells. Toxins. 2023;15(1):47. doi: https://doi.org/10.3390/toxins15010047
- Pacheco PAF, Faria JV, Silva AC, von Ranke NL, Silva RC, Rodrigues CR, et al. In silico and pharmacological study of N,S-acetal juglone derivatives as inhibitors of the P2X7 receptor-promoted in vitro and in vivo inflammatory response. Biomedicine & Pharmacotherapy. 2023;162114608. doi: https://doi.org/10.1016/j.biopha.2023.114608
- de Luna Martins D, Borges AA, e Silva NADA, Faria JV, Hoelz LVB, de Souza HVCM, et al. P2X7 receptor inhibition by 2-amino-3-aryl-1,4-naphthoquinones. Bioorganic Chemistry. 2020;104:104278. doi: https://doi.org/10.1016/j.bioorg.2020.104278
- Abdullah SS, Mohammad Latif MA, Md Tohid SF. In Vitro And In Silico Studies Of Lawsone On Inflammation-Induced Skin Cells For Development Of Skin Anti-Inflammatory Treatment. Mab. 2024;53(3):197-209. doi: https://doi.org/10.55230/mabjournal.v53i3.2801
- Nair A, Sekar M, Gan SH, Kumarasamy V, Subramaniyan V, Wu YS, et al. Lawsone Unleashed: A Comprehensive Review on Chemistry, Biosynthesis, and Therapeutic Potentials. Dddt. 2024;Volume 18:3295-3313. doi: https://doi.org/10.2147/dddt.s463545
- Zulkefle HS, Abdullah SS, Mohammad Latif MA, Md Tohid SF. Lawsone attenuates acute low dose ethanol-induced skin inflammation in A431 epidermoid carcinoma skin cells and its possible mechanisms in targeting interleukin (IL)-1α in silico. Lsmb. 2024;8(1). doi: https://doi.org/10.28916/lsmb.8.1.2024.162
- Al-Snafi AE, Talab TA, Alfuraiji N. The analgesic and anti-inflammatory effect of lawsone isolated from Lawsonia inermis. Sr: Ps. 2022;(1(35)):77-84. doi: https://doi.org/10.15587/2519-4852.2022.253555
- Kavaliauskas P, Opazo FS, Acevedo W, Petraitiene R, Grybaitė B, Anusevičius K, et al. Synthesis, Biological Activity, and Molecular Modelling Studies of Naphthoquinone Derivatives as Promising Anticancer Candidates Targeting COX-2. Pharmaceuticals. 2022;15(5):541. doi: https://doi.org/10.3390/ph15050541
- Pislyagin E, Kozlovskiy S, Agafonova I, Menchinskaya E, Chingizova E, Gorpenchenko T, et al. Sea Urchin Pigment Ethylspinazarin (U-573): A Novel P2X7 Receptor Antagonist with Neuroprotective and Antiparkinsonian Effects. Ijms. 2025;26(17):8639. doi: https://doi.org/10.3390/ijms26178639
- Pislyagin E, Kozlovskiy S, Menchinskaya E, Chingizova E, Likhatskaya G, Gorpenchenko T, et al. Synthetic 1,4-Naphthoquinones inhibit P2X7 receptors in murine neuroblastoma cells. Bioorganic & Medicinal Chemistry. 2021;31:115975. doi: https://doi.org/10.1016/j.bmc.2020.115975
