REVIEW
Leveraging Artificial Intelligence to Strengthen Vaccine and Drug Development Capacity in Low-resource African Settings
Academic Editor: Adeleye Ademola Olutayo
Sciences of Pharmacy|Vol. 5, Issue 2, pp. 145-161 (2026)
Received
Jan 8, 2026Revised
Feb 17, 2026Accepted
Feb 24, 2026Published
Apr 28, 2026
Abstract
Introduction
Global inequities in vaccine and drug innovation are increasingly driven by disparities in access, delivery, and manufacturing capacity rather than scientific capability alone (1). High‑income countries benefit from advanced infrastructures that enable rapid responses to emerging health threats, while low‑resource regions, particularly in Africa, remain dependent on external supply chains and imports, as highlighted during the COVID‑19 pandemic (2). This dependency reinforces vulnerability and delays equitable access to life‑saving interventions. Africa carries a disproportionate burden of infectious and neglected diseases. Sub‑Saharan Africa accounts for approximately 67% of the global HIV burden, with an estimated 25.6 million people living with HIV in 2023, despite a 57% decline in new infections in Eastern and Southern Africa since 2010 (3). Malaria remains a leading cause of morbidity and mortality, with the continent responsible for 96% of global malaria cases and 97% of malaria deaths in 2023, and progress toward elimination targets has plateaued (4). Tuberculosis (TB) continues to pose a major challenge, with 2.5 million new TB cases reported in Africa in 2021, alongside rising multi-drug‑resistant strains that threaten World Health Organization’s (WHO’s) 2035 elimination goals (5). Viral hepatitis is also a growing concern, with over 80 million people chronically infected with hepatitis B or C in Africa, contributing significantly to liver‑related morbidity and mortality (6). Together, these statistics underscore the urgent need for innovative approaches to strengthen biomedical research and development capacity, since these diseases necessitate effective therapeutics for treatment and vaccines for prevention. Artificial intelligence (AI) and machine learning (ML) have emerged as transformative tools in biomedical innovation. AI refers to computational systems capable of performing tasks that normally require human intelligence, including reasoning, learning, and decision‑making (7, 8). ML, a subset of AI, employs algorithms that learn from large datasets to improve predictions and optimize outcomes without explicit programming (7, 9). Applications in drug discovery and vaccinology include antigen design, protein structure prediction, epitope mapping, molecular docking, and adaptive clinical trial design (10). Tools such as AlphaFold have revolutionized protein modelling, while generative chemistry platforms have accelerated compound identification (11). Despite these advances, the applicability of AI in Africa’s resource‑limited contexts remains under-explored. Existing literature highlights global advances in AI for biomedical innovation (10-15) but rarely contextualizes these within Africa’s unique infrastructural and governance constraints (16, 17). Some reviews critically examine how readiness factors such as infrastructure, skills transfer, and ethical frameworks intersect with AI adoption in resource‑limited settings (18-20). This review addresses that gap by synthesizing evidence into a framework for sustainable, locally relevant AI integration in vaccine and drug development. It introduces a conceptual framework that situates AI within the broader innovation ecosystem, linking computational capabilities to infrastructural, regulatory, and human capital dimensions. By critically analyzing both opportunities and persistent challenges, the review aims to provide a roadmap for building resilient, locally owned biomedical innovation systems.
Methodology
This study was conducted as a narrative review, designed to integrate diverse forms of evidence on the role of AI in vaccine and drug development in Africa. The narrative review was chosen to allow flexibility in synthesizing empirical studies, institutional reports, and case‑based accounts, while situating global advances within African‑specific contexts.
Search Approach
Evidence was identified through targeted searches of PubMed, Scopus, Web of Science, and Google Scholar, complemented by repositories maintained by the World Health Organization, Africa Centers for Disease Control and Prevention. Boolean operators were applied to refine searches, using combinations such as (“Artificial Intelligence” OR “Machine Learning”) AND (“Vaccine” OR “Drug Discovery”) AND (“Africa”). The time window of 2015-2025 was selected to capture the decade in which deep learning and computational modelling became widely applied in biomedical research, including landmark developments such as AlphaFold.
Selection and Eligibility
Sources were included if they: 1) Explicitly addressed AI applications in vaccine development, drug discovery, or biomedical innovation relevant to Africa, 2) Reported empirical findings or case studies with transferable insights to African contexts, 3) Examined infrastructural, policy, or ethical frameworks enabling or constraining AI integration.
Exclusion criteria applied to speculative accounts lacking empirical grounding, opinion pieces without methodological transparency, non-English sources and studies focused exclusively on non‑African contexts without relevance.
Data Handling and Analysis
Data was extracted on study objectives, AI techniques employed, disease focus, geographic scope, and reported outcomes. Extracted material was organized thematically to highlight recurring patterns across infrastructural enablers and barriers, human capital requirements, and translational pathways for AI integration.
Ethical Integration
Ethical dimensions were operationalized by assessing whether studies addressed data privacy, consent frameworks, and equitable access. These considerations were integrated into the thematic synthesis to ensure contextual relevance for African biomedical ecosystems.
Credibility and Triangulation
Findings were triangulated across peer‑reviewed literature, institutional reports, and case‑based evidence. This approach ensured that global exemplars (e. g., AlphaFold, generative chemistry platforms) were critically examined alongside African initiatives (e. g., H3D Centre, BioStruct‑Africa, Ersilia Open Source Initiative), thereby enhancing both credibility and contextual depth.
Results and Discussion
Limitations in Africa’s vaccine and drug research and development ecosystem
Africa’s biomedical research ecosystem continues to face major hurdles. Infrastructure remains one of the most significant challenges. Few facilities meet international standards for manufacturing or preclinical testing, limiting the continent’s ability to independently develop vaccines and medicines (21). This infrastructural deficit reduces the generation of standardized datasets, weakening AI model training and slowing translation into clinical products. Biomanufacturing capacity is also highly uneven worldwide, with low‑resource regions such as Africa, lacking the enabling infrastructure required to support vaccine and drug development (22). Preclinical research is further constrained by shortages of specialized facilities, equipment, and trained personnel, slowing the translation of laboratory discoveries into viable biomedical products (22). Without coordinated investment in shared infrastructure and research platforms, innovation pipelines risk fragmentation and inaccessibility, reinforcing global disparities in biopharma development (22). Data systems present another weakness. Health datasets and biobanks are often fragmented, with limited interoperability between countries or institutions. Africa hosts less than 1% of global high‑performance computing capacity (23), making it difficult to run large‑scale AI models that rely on integrated, high‑quality data. In many regions, scientific data remains siloed across institutions, hindering the development of cohesive research ecosystems (24). Weak research networks slow knowledge exchange and reduce the impact of scientific outputs (24). Human capital is also scarce, there are too few bioinformaticians, computational biologists, and data scientists to meet the growing demand for AI‑driven research (25). While training programs exist, they remain limited in scale and scope, leaving many institutions without the skilled personnel needed to harness modern tools. This shortage undermines Africa’s capacity to fully participate in global innovation networks and weakens preparedness for emerging health threats (25). The COVID-19 pandemic further exposed Africa’s heavy dependency on external vaccine development and supply, with most doses secured through imports rather than local production (26). This dependency reinforced inequities, as wealthier nations monopolized early supplies, leaving low‑income regions vulnerable and highlighting the urgent need for stronger local innovation ecosystems (26). Table 1 summarizes the systemic bottlenecks that continue to constrain vaccine and drug research and development across Africa. It highlights infrastructural, data, human capital, and regulatory challenges, as well as the continent’s dependency on external supply chains. Together, these limitations illustrate the structural barriers that AI integration must address to strengthen biomedical innovation.
| S/N | Bottleneck Area | Key Limitations | References |
|---|---|---|---|
| 1 | Biomanufacturing and preclinical infrastructure | Limited GMP‑compliant facilities, shortages of specialized equipment, and inadequate enabling research platforms slow translation of discoveries into products. | (22) |
| 2 | Data ecosystems and research networks | Fragmented datasets, poor interoperability, and weak collaborative frameworks restrict large‑scale analysis and coordinated innovation. | (24) |
| 3 | Human capital | Severe shortage of skilled bioinformaticians, computational scientists, and trained biomedical researchers undermines capacity for advanced R& D. | (25) |
| 4 | Structural barriers in biopharma | Weak regulatory systems, financing gaps, and reliance on imports constrain adoption and scaling of biotherapeutics. | (22) |
| 5 | Dependency in vaccine development | Heavy reliance on external R& D and supply chains, exposed during COVID‑19, left Africa vulnerable to inequitable access. | (26) |
Role of AI in biomedical innovation
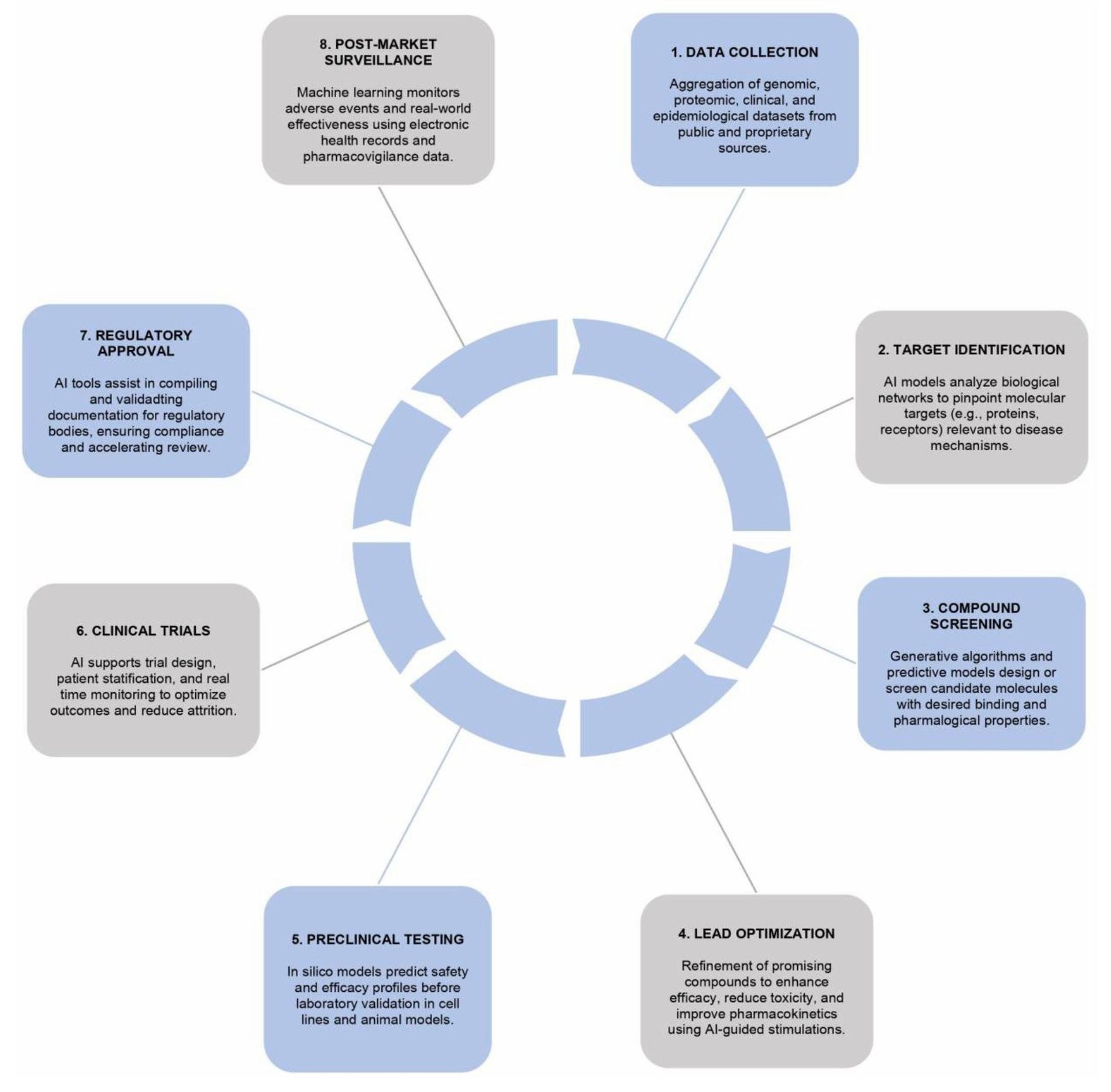
AI brings together a range of computational techniques such as machine learning, deep learning, and natural language processing that can reshape biomedical research. It is important to distinguish conventional computational biology methods (e.g. , docking, cryo‑EM) from AI‑driven approaches (e.g. , machine learning‑based epitope prediction). While both are computational, only AI systems learn from large datasets to generate predictive insights. AI and ML principles in biomedicine enable predictive modelling, pattern recognition, and optimization of therapeutic strategies, thereby accelerating drug discovery and improving clinical decision‑making (27, 28). AI technologies are increasingly applied in molecular modelling, compound screening, and antigen discovery, where they accelerate the identification of drug candidates and optimize antibody design by predicting structural interactions and therapeutic potential (29). In drug discovery, AI speeds up molecular modeling, compound screening, and toxicity prediction. In vaccine design, it supports antigen discovery, epitope prediction, and immune response modelling (30). AI also integrates well with omics data, making it possible to design treatments that are more precise and tailored to specific populations. For example, AI‑driven genomic surveillance has been used to track COVID‑19 variants in real time, showing its value for public health (27). This also supports precision medicine by revealing predictive biomarkers and disease patterns (31). Global examples highlight the scale of AI’s impact. AlphaFold's breakthrough in protein structure prediction opened new doors for rational drug and vaccine design (32). AI‑powered screening platforms have also identified promising compounds for neglected tropical diseases, offering hope for conditions that have long been overlooked (33). For Africa, the challenge is to translate these advances into local contexts. This requires investment in cloud computing, regional data centers, and centers of excellence that can support AI research. Programs like ‘Holistic Drug Discovery and Development’ (H3D) Africa demonstrate that building bioinformatics capacity is possible, and collaborations with international AI hubs can accelerate skills transfer (34). AI should be seen not as a replacement for traditional methods, but as a complementary tool that can democratize biomedical innovation. By cutting costs and timelines, it allows resource‑limited settings to participate more fully in global research and development, strengthening both local resilience and global health security. Figure 1 outlines the stages of an AI‑driven vaccine and drug discovery pipeline, illustrating how computational tools can accelerate candidate identification, optimize molecular design, and support adaptive clinical trial strategies. The schematic emphasizes the complementary role of AI in reducing costs and timelines in resource‑limited settings.
AI in vaccine design and development
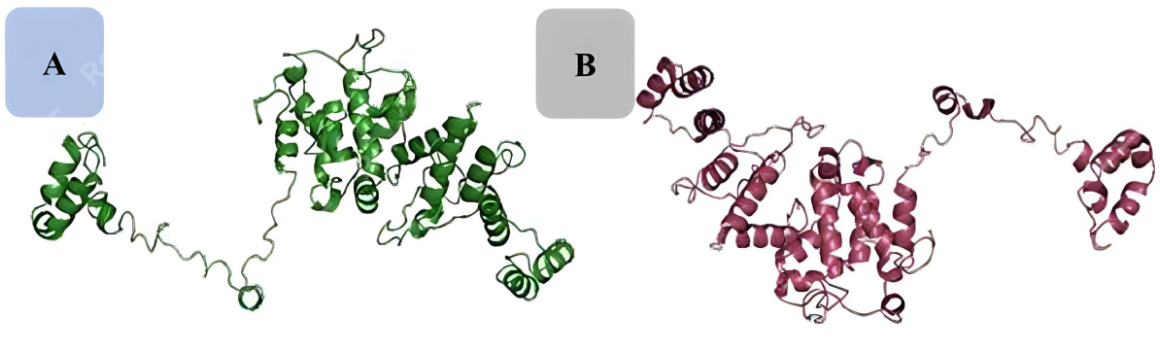
The few African vaccine manufacturers on the continent are located in eight countries: Egypt, Tunisia, Algeria, Morocco, South Africa, Senegal, Nigeria, and Ethiopia (35). The application of AI in vaccine development is accelerating the deployment of vaccines. Researchers can utilize the interactions between bio-molecules with the help of AI (36). Vaccine candidates are produced by fusing peptide epitopes with linkers to simplify vaccine design. Molecular docking can help develop multi-epitope vaccines and can also be used to study the immune receptor binding affinity of putative vaccine candidates (37). Studies employing algorithms have shown immunogenicity improvement in proof-of-concept experiments notably of SARS-CoV-2 mRNA vaccines. Beyond COVID-19, epitope-focused ML pipelines have shown promise in designing peptide-based vaccines for HIV and malaria. Current advancements have integrated AI-driven molecular simulations to further validate and optimize predicted epitopes (38). In malaria, two sexual-stage surface proteins essential for Plasmodium transmission, Pfs230 5 and Pfs48/45, 6 are expressed in both gametocytes and gametes. The Pfs230: Pfs48/45 complex forms the basis for leading malaria transmission-blocking vaccine (TBV) candidates. A recent study used cryo-electron (cryo-EM) microscopy to elucidate the structure of the endogenous Pfs230: Pfs48/45 complex bound to six transmission-blocking antibodies. This remains at the proof‑of‑concept stage, with no clinical validation yet achieved. Figure 2 presents the cryo‑EM structure of the endogenous Pfs230: Pfs48/45 complex bound to six antibodies. This visualization improves molecular understanding of malaria transmission‑blocking activity and informs the development of next‑generation Plasmodium falciparum TBV candidates (39).
Rapid epitope mapping to adaptive clinical trial designs enabled by AI/ML have helped shorten certain phases from years to months, exhibiting a potential paradigm shift in how vaccines are conceived, tested, and produced (38). Such acceleration has been observed primarily in pandemic contexts (e.g. , COVID‑19 mRNA vaccines), whereas routine vaccine pipelines remain slower and resource‑dependent. In another study, a multi-epitope sub-unit vaccine was designed against adenylate kinase 2 protein and evaluated for its potential to elicit protective immunity against three Schistosoma species. CTL, HTL, and B-cell epitopes were identified using immunoinformatics tools and linked using AAY and KK linkers, respectively. The 50S ribosomal protein L7/L12, a known TLR4 agonist, was incorporated as an adjuvant to enhance immune activation in the vaccine. The three-dimensional (3D) model was predicted using the Iterative Threading Assembly Refinement (I-TASSER) server (https: //zhanggroup. org/I-TASSER/) and refined using the Galaxy Web server. Molecular docking and molecular dynamics (MD) simulations demonstrated a strong binding affinity between the vaccine and human TLR4, supported by low RMSD and Rg values, indicating structural stability. The negative binding energy further validated the vaccine’s potential for engaging TLR4. The immunogenic profile predicted robust activation of CD4+ and CD8+ T cells, as well as neutralizing antibody responses (40). Figure 3 depicts the three‑dimensional structure of a multi‑epitope vaccine designed against S. species. The refined model demonstrates strong binding affinity with human TLR4, supporting its potential immunogenic profile and translational relevance.
Over the past 15 years, numerous HIV-1 vaccine trials have failed to produce significant evidence of efficacy (41). Generally, AI/ML solutions have been deployed in relatively few African countries, primarily Kenya, Nigeria, Ghana, Ethiopia, and South Africa. Expanding AI/ML capabilities across Africa is now underway, supported by both the private sector, government, and non-governmental organizations (42). Table 2 presents selected AI applications in vaccine development relevant to Africa. It documents case studies of malaria and schistosomiasis vaccine candidates, detailing the computational techniques employed and their geographic relevance. These examples illustrate how AI and ML are being applied to neglected tropical diseases.
| S/N | Vaccine candidate: target species | Medical geography of disease | AI/ML (and other key techniques) used | References |
|---|---|---|---|---|
| 1 | Malaria vaccine (Plasmodium falciparum) | Tropics and Subtropics especially Sub-Saharan Africa | Molecular docking, (cryoelectronic microscopy) | (39) |
| 2 | Schistosoma vaccine (Schistosoma haematobium and Schistosoma mansoni and Schistosoma japonicum) | Tropics and Subtropics especially Sub-Saharan Africa | Docking, homology modelling: I-TASSER, Galaxy Webserver | (40) |
AI in drug discovery, re-purposing, design and development
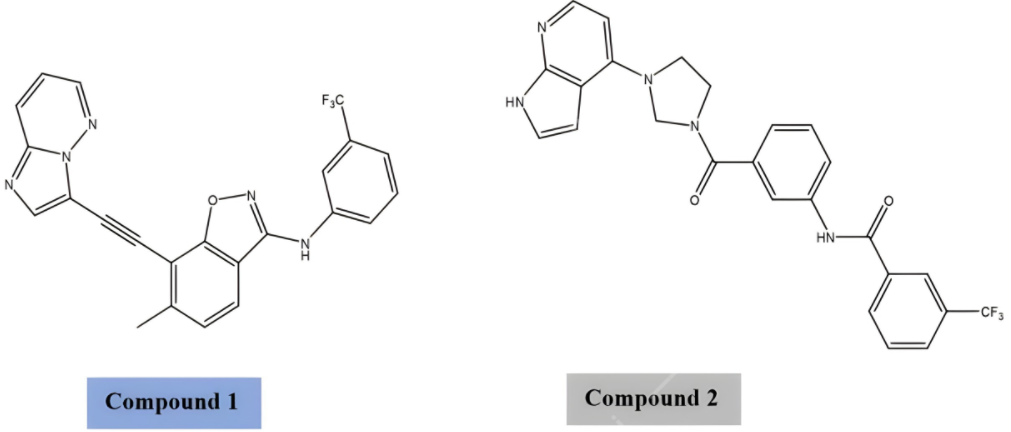
Quantitative structure activity relationship (QSAR) remains a foundational method in computer-aided drug design (CADD). Traditionally, QSAR models describe the relationship between chemical structure and biological activity, encompassing properties from target bioactivity to ADMET (absorption, distribution, metabolism, excretion, and toxicity) parameters such as solubility, permeability, and physicochemical traits. Recent advances in deep learning have significantly enhanced QSAR, enabling more accurate predictions and broader applicability across drug discovery pipelines (43, 44). Multi-parametric optimization (MPO) platforms now integrate QSAR outputs with encoded target/off-target bio-activities and ADMET features, allowing simultaneous optimization of multiple drug properties. This holistic approach reduces attrition rates in preclinical development and accelerates candidate prioritization (43). Beyond classical QSAR, AI-driven generative models such as Generative Tensorial Reinforcement Learning (GENTRL) have demonstrated the ability to design novel compounds, including DDR1 and DDR2 inhibitors, within weeks rather than years (45). These generative approaches exemplify how AI can introduce predictive novelty, though validation against established screening methods remains essential to ensure translational reliability. Recent work on deep QSAR highlights the synergy between traditional CADD and modern AI. By leveraging large molecular datasets, improved computational power, and neural architectures, deep QSAR models outperform conventional methods in predicting bio-activity and ADMET properties, offering a scalable framework for drug discovery in resource-limited settings (44, 47). Together, QSAR, MPO, and generative AI approaches represent complementary strategies - classical CADD provides interpretability and mechanistic grounding, while AI introduces speed, scalability, and novel predictive capacity. Their integration is reshaping drug discovery globally and holds particular promise for regions like Africa, where pharmaceutical innovation is increasingly tied to digital transformation and AI adoption (46, 47). Figure 4 shows the chemical structures of two epithelial DDR inhibitors developed using generative tensorial reinforcement learning. This example demonstrates the application of AI‑driven generative chemistry in drug discovery. It highlights how computational design can accelerate the identification of novel therapeutic compounds.
Upon the recent landmark discovery of the praziquantel (PZQ) target in Africa-endemic S. mansoni (Sm. TRPMPZQ), molecular docking was used to elucidate the drug-target interactions (48-52). Figure 5 illustrates the docking of praziquantel onto its S. mansoni TRPMPZQ target. The visualization highlights key hydrogen bonding and hydrophobic interactions that underpin drug‑target affinity. This structural insight advances understanding of praziquantel’s mechanism of action in endemic schistosomiasis.
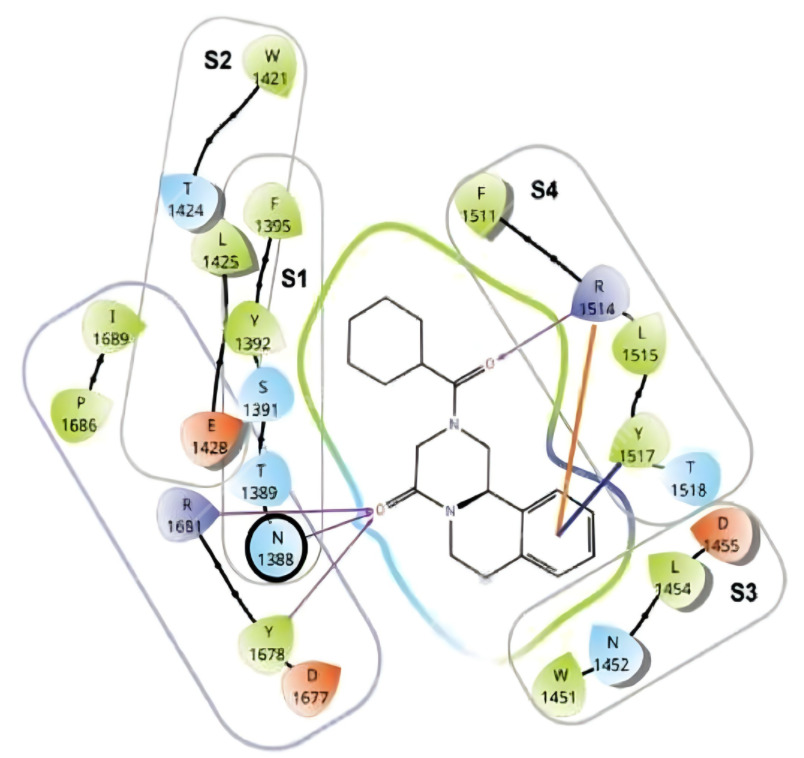
| KEY: TRP: transient receptor potential; S1 to S4: transmembrane helices; Amino acid single letter symbols: D: aspartate; E: glutamate; F: phenylalanine; I: isoleucine; L: leucine; N: asparagine; P: proline; R: arginine; S: serine; T: threonine; W: tryptophan; Y: tyrosine. |
Note the two carbonyl oxygens of PZQ act as hydrogen bond acceptors (HBAs) to amide, phenoxyl and guanidino hydrogens of arginine (R), tyrosine (Y) and asparagine (N) amino acid residue side chains (hydrogen bond donors i.e. HBDs). Also notable are hydrophobic interactions between the phenyl group of PZQ to both Y and R. Recently, drug repurposing has witnessed the development of numerous computational resources dedicated to supporting these endeavors. Notable examples include DrugRepo, Drug Repurposing Hub, repoDB, and RepurposeDB, along with an array of other web-based databases, collectively contributing directly or indirectly to the drug repurposing landscape (53). However, performance metrics such as predictive accuracy, reproducibility, and external validation remain limited, reducing confidence in scalability. Hundreds of computational resources such as databases and predictive platforms have been developed that can be applied for drug re-purposing, making it challenging to select the right resource for a specific drug re-purposing project (54). Public availability of diverse compound libraries, well annotated in terms of chemical structures and scaffolds, modes of action, and bio-activities are cardinal to ensure the participation of academic laboratories in these screening efforts, especially in resource-limited settings (55). A study conducted in Egypt used numerous cheminformatics tools to investigate the chemical and biological properties of the bark of Eucalyptus globulus grown in Egypt. Sirius software was used to process liquid chromatography - mass spectrometry (LC-MS) chemical profiling (56).
Capacity building and infrastructure for AI-driven research and development in Africa
One of the earliest efforts in genomics research expansion in Africa is the Human Heredity and Health in Africa (H3Africa) initiative. The ultimate goal of the initiative is to facilitate a contemporary research approach to the study of genomics and environmental determinants of common diseases with the aim of improving the health of African populations (57). The African Center of Excellence in Bioinformatics and Data-Intensive Science (ACE) Mali centre to address critical gaps in research infrastructure and training capacity in bioinformatics and data science in Mali and West Africa (58). To advance its mission of discovering and developing novel, life-saving medicines for infectious diseases that predominantly affect African populations, the Holistic Drug Discovery and Development (H3D) Centre of the University of Cape Town works closely with Ersilia Open Source Initiative (EOSI) aimed at disseminating AI/ML methodologies applied to urgent biomedical needs in low-resource settings. In 2020 EOSI, a non-profit organization was created with the primary aim of attracting code developers into contributing to the discovery of new anti-infectives. Ersilia was conceived as an in silico drug discovery endeavor, with a focus on leveraging AI/ML techniques to support experimentation in the Global South, notably in Sub-Saharan Africa, where many infectious diseases are endemic. By 2024, Ersilia has become a thriving community of developers (59). Its brainchild ZairaChem is an AI/ML-based tool for QSAR modelling. ZairaChem is fully automated and has been implemented at the H3D Centre. By leveraging in-house data collected over a decade, a virtual screening cascade for malaria and tuberculosis drug discovery was developed (60). BioStruct-Africa provides training in cutting-edge AI tools, such as AlphaFold, ChimeraX, and PyMOL the goal of nurturing a skilled community of structural biologists committed to addressing emerging diseases in Africa. Its workshops foster networks among 100 young scientists and early-career scientists from 20 different African countries (61). For instance BioStruct‑Africa has trained over 100 young scientists, but retention rates and sustained funding models remain unclear. Without addressing energy infrastructure and maintenance capacity, training alone cannot resolve systemic bottlenecks. Bioinformatics heavily relies on robust computational tools, expensive software, and strong digital infrastructure. Notably, Africa has about 1% of the global computing capacity, inadvertently limiting its capacity to effectively harness computational technology for biomedical research (23, 62). High-performance computing is essential for quantum, mechanics and molecular dynamics simulations, protein structure modelling, etc. However, universities in many African countries generally lack the financial capacity to invest in such infrastructure. In situations where the hardware has been donated, it becomes challenging to maintain it in terms of powering and upgrading software (63). Table 3 lists African institutions currently advancing AI in biomedical research. It provides details on their establishment, services, and international partnerships. The table highlights the growing network of centers of excellence that are building computational and bioinformatics capacity across the continent.
| S/N | Institution/Organization, Mother Institution, Location | Year of Establishment | Biomedical AI Services | Partner institution | References |
|---|---|---|---|---|---|
| 1 | African Center of Excellence in Bioinformatics and Data-Intensive Science (ACE), University of Sciences, Techniques and Technologies of Bamako (USTTB), Bamako, Mali. | 2015 | Provide advanced computing and storage resources to researchers and bioinformatics | US National Institute of Allergy and Infectious Diseases (NIAID), US National Institutes of Health (NIH) | (58) |
| 2 | African Center of Excellence in Bioinformatics and Data Intensive Sciences (ACE), Makerere University, Kampala, Uganda. | 2019 | Provide advanced computing and storage resources to researchers and bioinformatics | NIAID, NIH | (64) |
| 3 | Human Heredity and Health in Africa (H3Africa), Addis Ababa, Ethiopia. | 2010 | Genomics and environmental determinants of common diseases | NIH, Wellcome Trust, African Society of Human Genetics (AfSHG) | (52) |
| 4 | African Pathogen Genomics Initiative (PGI), South Africa, Senegal, Ghana, Nigeria, Kenya, DR Congo and Uganda | 2020 | Manage endemic infections such as malaria, HIV, TB and cholera. integrating pathogen bioinformatics and genomics in disease surveillance | Africa Centres for Disease Control (CDC), World Health Organization (WHO), Illumina, Oxford Nanopore, Gates Foundation, US CDC Microsoft | (52) |
| 5 | BioStruct-Africa, Kumasi, Ghana. | 2019 | Empowering researchers through structural biology knowledge. | Google DeepMind, The Company of Biologists, Swedish Research Council | (61) |
| 6 | Holistic Drug Discovery and Development (H3D) Centre, University of Cape Town (UCT), Cape Town, South Africa. | 2023 | ZairaChem AI/ML QSAR model for infectious disease small molecule drug discovery | Ersilia, Schmidt Sciences | (60) |
| 7 | Center for Drug Discovery (CeDD), University of Buea, Cameroon. | 2022 | Developing computational screening of the African Natural Products Database | Nature-inspired Discovery of Novel Antivirals (NiDNA) network | (65) |
Ethical, regulatory, and equity considerations
The integration of AI within the vaccine R& D continuum faces ongoing barriers -heterogeneity of immunological datasets, and interpretability issues in advanced AI models and regulatory uncertainties. These challenges have been highlighted in global AI ethics frameworks, underscoring the need for region‑specific adaptation in Africa. Regulators such as the US Food and Drug Administration (FDA), Europeans Medicines Agency (EMA) are formulating guidelines for safe and effective AI adoption in healthcare contexts, though standardized frameworks for AI in vaccinology are still emerging (66). Recent guidance documents outline pathways for AI/ML in medical devices, but equivalent frameworks for vaccinology remain underdeveloped. In parallel, issues of intellectual property, data privacy, and consent frameworks necessitate careful navigation to maintain public trust in AI solutions. Close collaboration among regulatory agencies e.g. , WHO, FDA, EMA, etc is vital for clear guidelines on AI-driven clinical trials, data validation, and safety oversight. Transparency requirements regarding model architecture, performance metrics, and interpretability should be standardized before approval. Emerging regulatory proposals, such as the EU AI Act, emphasize the importance of standardized disclosure protocols (67). Concretely, implementing robust bias auditing frameworks and ensuring diverse, representative datasets will enhance AI fairness. Operational models for bias auditing in healthcare AI demonstrate how fairness can be measured and enforced. Interdisciplinary ethics committees provide additional oversight, maintaining public trust through accountability and transparent communication (68). Regional ethics committees in Africa could adapt these models to local governance structures, ensuring culturally appropriate oversight. This integration also faces ethical uncertainties. By enabling adaptive clinical trial designs, AI has been shown to cut the conventional years R& D cycle down to months in certain pandemic scenarios. Evidence from COVID‑19 vaccine development illustrates that acceleration was context‑specific, with routine pipelines remaining slower. Enhanced AI algorithms have recently enabled even finer predictive modelling for clinical trial outcomes. Ethical review boards can oversee algorithmic explainability and data governance (38). Ethical issues surrounding human genome research in Africa have been a serious concern that need to be holistically addressed. Initiatives such as H3Africa have documented these challenges, particularly around consent and community engagement. Some of these have raised ethical questions on several issues such as the proper model of consent for archiving samples, privacy, data sharing and confidentiality, appropriate mode of community engagement in the research process and the best method of communicating laboratory results to participants (57). AI is poised to transform society and scientific activities such as anti-infectives drug discovery. It is important to consider how AI will be deployed fairly and at scale in practice, asking who will benefit the most from this technology and who is at risk of being left behind. Aware of this imbalance, the EOSI was launched with the determined aim of supporting infectious disease research in Africa (68). This initiative exemplifies how open‑source AI can address equity gaps, though sustainability and scalability remain ongoing challenges.
Policy and strategic frameworks
African countries mostly rank low on the AI Readiness Index, with sub‑Saharan Africa identified as the worst‑scoring world region in terms of government preparedness to implement AI in public services (69). This low ranking underscores structural barriers such as limited infrastructure, fragmented data systems, and regulatory weaknesses, which directly constrain the integration of AI into biomedical innovation. Notable policy and strategic frameworks have been proposed, including investment in infrastructure and equipment and the recognition of patents on AI-generated inventions (70). However, the feasibility of these frameworks depends on coordinated implementation and sustained funding models. The application of AI technologies for improving health interventions via drug discovery and vaccine design is expanding across Africa, particularly in South Africa, Kenya, Nigeria, and Ghana (71). For example, AI is improving pharmaceutical product quality and efficiency in Nigeria, while South African pharmaceutical plants employ robotics for automation in filling, packaging, and quality inspection. Kenya’s pharmaceutical industry is exploring AI for process optimization and predictive maintenance, and Ghana is increasingly harnessing AI to strengthen product quality and supply chain integrity (72-74). These examples demonstrate early‑stage successes but remain proof‑of‑concept rather than evidence of scalable transformation. At the continental level, the Africa Centres for Disease Control and Prevention (Africa CDC) and the African Union Development Agency-New Partnership for Africa’s Development (AUDA‑NEPAD) have begun promoting the incorporation of AI in local diagnostic manufacturing (75, 76). To translate these initiatives into sustainable outcomes, strong coordination between international funders, local governments, and the Africa CDC is required to ensure that funding commitments materialize (70). Without such structural alignment, policy frameworks risk remaining aspirational. Capacity building is essential, but outcome‑based assessment must accompany training programs. Metrics such as workforce retention, research outputs, and translational success rates are needed to evaluate impact. With successful capacity building, Africa could benefit from safer and more effective treatments, reducing healthcare costs and supporting disease‑eradication goals outlined by the World Health Organization (70). However, structural challenges such as energy infrastructure, maintenance capacity, and regulatory harmonization must be addressed to ensure sustainability. Looking ahead, the use of AI in vaccine development is expected to grow, with quantum computing offering potential to accelerate candidate discovery. The prediction accuracy of AI models utilized in vaccine research is also anticipated to increase (75-80). Yet, these advances will only translate into meaningful impact if embedded within grounded policy frameworks that address infrastructural deficits, regulatory pathways, and institutional heterogeneity. Table 4 outlines regional policy initiatives supporting AI and health innovation in Africa. It describes the committees, objectives, and strategic frameworks guiding implementation. The table demonstrates how political will and coordinated governance are shaping the continent’s readiness for AI‑driven biomedical advances.
| S/N | AI and health innovation | Regional Committee | Initiative Description | Policy Implementation Objectives |
|---|---|---|---|---|
| 1 | Political will. | African Research and Development (R& D) Ministerial Advisory Committee. | Ministers of Health; Ministers of Higher Education and Science and Technology or Innovation; Director General of Africa CDC; and Chief Executive Officer of AUDA, NEPAD. | Lead the advocacy for R& D with their respective Heads of State and take responsibility for administrative and budgetary matters. |
| 2 | Policy and governance. | Steering Committee. | Develop legal and ethical frameworks that safeguard health data sovereignty. | Establish regional and national AI health policies. Form oversight bodies for ethical governance. Coordinate the strategic direction of the governance and coordinating framework to ensure its independence, professionalism, and credibility; the Steering Committee is responsible for appointing members of the Scientific Advisory Committee. |
| 3 | Ethical AI design | Ethics and regulatory policies, procedures, and guidelines subcommittee. | Develop inclusive, culturally-sensitive AI systems grounded in African data realities. | Build representative, diverse datasets. Co-design solutions with communities. Play technical and scientific roles in developing and harmonizing ethics and regulatory policies, procedures, and guidelines in the continent. |
| 4 | Local system integration. | Health research subcommittee. | Ensure alignment between AI tools and national health policies and workflows. | Embed AI in primary healthcare systems. Train front-line clinicians in AI supported practices. Play technical and scientific roles in continental and regional health research including the development and review of clinical trial protocols. |
| 5 | Dynamic learning systems. | Digital technologies subcommittee. | Enable continuous learning and adaptation based on local health data. | Establish real-time feedback loops between clinicians and AI systems. Play technical and scientific roles in the digitalization of R& D activities continentally and regionally. |
| 6 | Open science infrastructure. | Networking subcommittee. | Promote transparency and accessibility through shared platforms and open repositories. | Encourage open data initiatives. Support local innovation labs and research hubs. Play technical and scientific roles in establishing R& D networks for knowledge and skill exchanges, joint protocols, and working together. |
Future directions and innovation roadmap
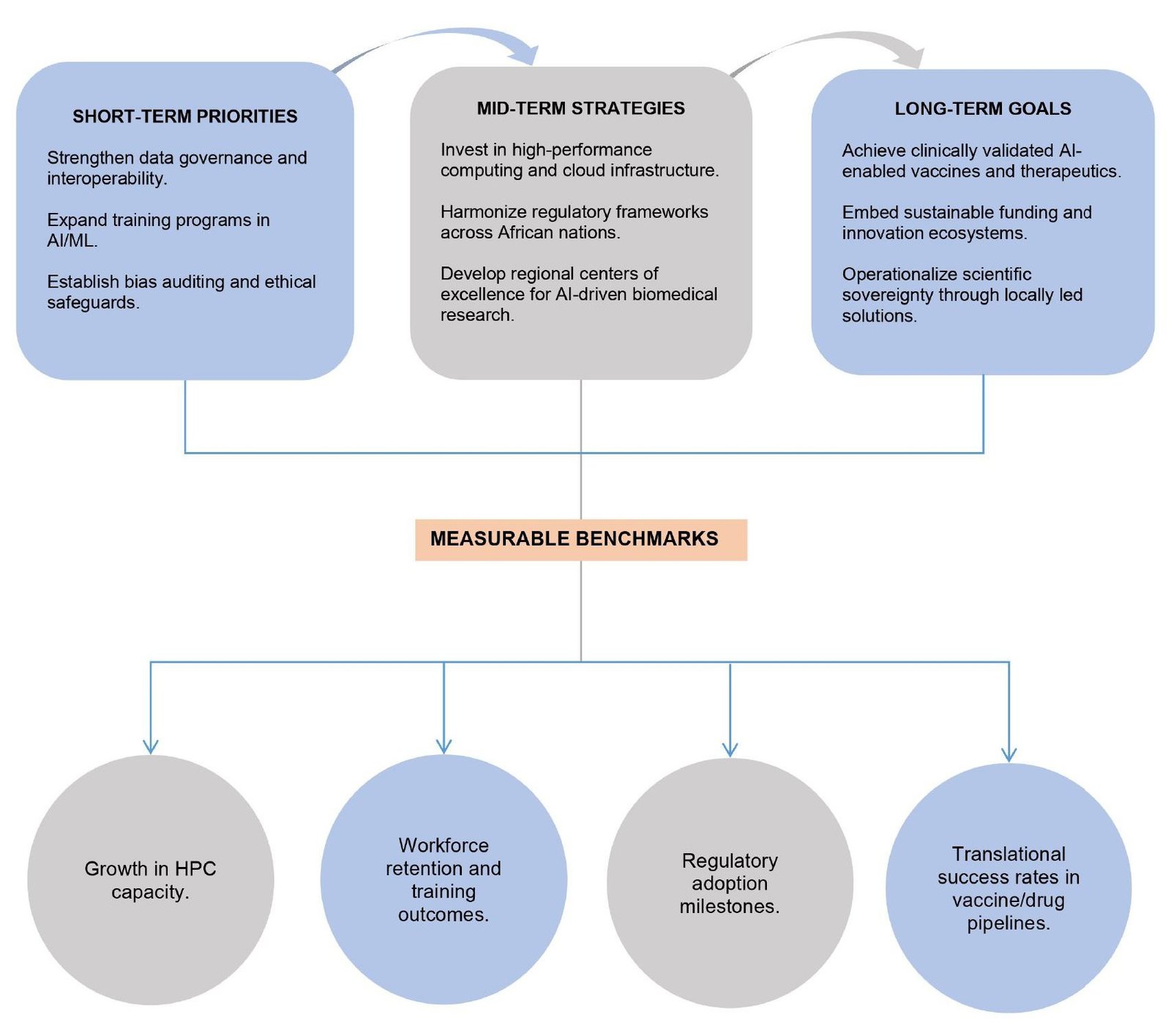
AI is poised to transform anti‑infectives drug discovery and healthcare delivery in resource‑limited settings (RLS). However, persistent challenges such as shortages of healthcare professionals, data scarcity, and inadequate regulatory frameworks leave RLS particularly vulnerable to AI’s potential risks and ethical violations (81, 82). These structural constraints highlight the need for policies that balance innovation with safeguards to ensure equitable deployment. Examples of capacity‑building initiatives illustrate how data sharing and training can stimulate world‑class research. The Ersilia Open Source Initiative has organized workshops across Africa to strengthen local expertise (83). Similarly, H3ABioNet manages data storage and network infrastructure for the H3Africa consortium, providing a backbone for genomic and biomedical research (70). The BioStruct‑Africa workshops further contribute by training scientists in cutting‑edge AI tools, cultivating a cadre of structural biologists committed to addressing emerging diseases in Africa (61, 84, 85). These initiatives demonstrate early‑stage computational successes but remain dependent on sustained funding, infrastructure, and workforce retention to achieve long‑term impact. In summary, AI’s role in Africa remains at three levels: (i) theoretical potential, (ii) early-stage computational successes, and (iii) clinically validated implementation with most current examples situated at stages (i) and (ii). This distinction underscores the need for sustained investment to achieve stage (iii). Figure 6 illustrates a progressive roadmap for strengthening AI‑enabled vaccine and drug research capacity in Africa. It highlights short‑term priorities such as data governance and training, mid‑term strategies including infrastructure investment and regulatory harmonization, and long‑term goals focused on clinically validated innovations and scientific sovereignty. Measurable benchmarks are integrated to ensure accountability and sustainable progress.
Recommendations
The successful integration of artificial intelligence (AI) and machine learning (ML) into vaccine and drug development in low-resource African settings will depend on coordinated collaboration across multiple stakeholders. Governments, pan-African research institutions, NGOs, and private-sector partners must work together to establish a shared understanding of existing gaps in the biomedical R& D ecosystem and to design a strategic roadmap for AI adoption. Public-private partnerships are particularly important, as they can mobilize resources and accelerate technology transfer, ensuring that AI innovations are embedded within vaccine and drug pipelines (86, 87). A critical priority is the development of regional centers of excellence dedicated to AI-enabled biomedicine. While previous consortia such as the Crick Africa Network and SANTHE have advanced vaccine R& D, they have not consolidated expertise into specialized hubs. Establishing centers strategically located near global transport and data exchange hubs would facilitate collaboration and ensure accessibility. These centers should host interdisciplinary teams focused on different stages of the R& D life-cycle, from target discovery to clinical trial optimization. In addition to conducting research, they must serve as training hubs to cultivate future generations of African scientists and healthcare professionals, thereby ensuring long-term sustainability of expertise (88-90). Strengthening data infrastructure and governance is equally essential. Secure, interoperable repositories are needed to enable real-time sharing of genomic, clinical, and epidemiological datasets. Open-source frameworks should be promoted to enhance transparency and reproducibility, but these must be accompanied by robust ethical and regulatory safeguards at both national and continental levels. African-led data governance models will be vital to ensure sovereignty and equitable benefit-sharing, preventing dependency on external actors (37, 91). Human capital development must be prioritized through targeted training programs. Fellowship and mentorship initiatives in AI/ML applications for biomedical sciences should be tailored to African contexts, while postgraduate curricula in medicine, veterinary science, and public health should integrate AI/ML modules. Retention of expertise can be encouraged by providing career pathways within regional centers of excellence, thereby reducing brain drain and ensuring continuity of innovation (92, 93). Finally, continuous knowledge exchange and global collaboration will be critical. Regular virtual workshops, in-person symposium, and cross-disciplinary collaborations should be institutionalized to share advances in AI-driven vaccine and drug development. Partnerships with international organizations will ensure that Africa remains integrated into global innovation ecosystems. Importantly, AI/ML integration must align with One Health and equity principles, addressing zoonotic diseases and antimicrobial resistance while ensuring that low-resource settings benefit equitably from accelerated biomedical pipelines (94, 95).
Conclusion
AI holds significant promise for strengthening vaccine and drug development capacity across Africa, but its current role remains largely at the level of theoretical potential and early computational successes. Evidence from case studies demonstrates progress in data sharing, predictive modeling, and training initiatives, yet few examples have reached clinically validated implementation. This underscores the importance of grounding expectations in the realities of infrastructure, workforce, and regulatory constraints. The concept of scientific sovereignty defined here as the ability of African nations to generate, regulate, and apply AI‑enabled biomedical innovations independently emerges as a critical long‑term goal. Achieving this requires coordinated investment in infrastructure, harmonized regulatory frameworks, and sustained workforce development to reduce dependency on external imports and ensure locally relevant solutions. However, this review is limited by the uneven availability of region‑specific empirical studies and the scarcity of longitudinal evidence on AI adoption in Africa. In addition, the reliance on published literature may overlook emerging initiatives or unpublished data that could further enrich the analysis. Looking forward, actionable strategies can be prioritized across different time horizons. In the short term, strengthening data governance, bias auditing, and training programs will build foundational capacity. In the mid‑term, investments in high‑performance computing, regulatory harmonization, and regional centers of excellence will enable more robust translational pipelines. In the long term, clinically validated AI‑enabled vaccines and therapeutics, supported by sustainable funding and governance models, will operationalize scientific sovereignty and deliver equitable innovation. Uncertainties remain, particularly regarding scalability in resource‑limited settings, sustainability of funding, and feasibility of regulatory harmonization across diverse institutions. Assertions about accelerated discovery must therefore be contextualized: while pandemic‑specific examples illustrate shortened timelines, routine pipelines continue to face structural bottlenecks. To track progress, measurable benchmarks such as computing capacity growth, workforce retention rates, regulatory adoption milestones, and translational success rates should be established. These benchmarks provide a roadmap for moving from aspiration to implementation, ensuring that AI contributes meaningfully to Africa’s biomedical innovation ecosystem.
Abbreviations
AAY =Alanine-Alanine-Tyrosine linker; ACE = African Center of Excellence; ADMET = Absorption, Distribution, Metabolism, Excretion, and Toxicity; AfSHG = African Society of Human Genetics; AI =Artificial Intelligence; AIDRIVEN = Artificial Intelligence Driven; AUDA = African Union Development Agency; B-Cell =B Lymphocyte; CADD =Computer-Aided Drug Design; CD4+ = Cluster of Differentiation 4 Positive T Cells; CD8+ = Cluster of Differentiation 8 Positive T Cells; CDC = Centers for Disease Control; CeDD =Center for Drug Discovery; CGI = Computer-Generated Imagery; CRYO-EM = Cryogenic Electron Microscopy; CTL =Cytotoxic T Lymphocyte; DAC = Diagnostic Advisory Committee; DDR1/DDR2 = Discoidin Domain Receptor 1/2; DNA =Deoxyribonucleic Acid; EMA =European Medicines Agency; EOSI =Ersilia Open Source Initiative; FDA = Food and Drug Administration; GENTRL =Generative Tensorial Reinforcement Learning; GMP = Good Manufacturing Practices; GPU =Graphics Processing Unit; H3ABioNet =Human Heredity and Health in Africa Bioinformatics Network; H3Africa =Human Heredity and Health in Africa; H3D =Holistic Drug Discovery and Development Centre; HBAs =Hydrogen Bond Acceptors; HBDs =Hydrogen Bond Donors; HIV =Human Immunodeficiency Virus; HPC = High-Performance Computing; HTL = Helper T Lymphocyte; IP = Intellectual Property; I-TASSER =Iterative Threading ASSEmbly Refinement; IVDs = In Vitro Diagnostics; KK linkers =Lysine-Lysine linkers; LC-MS =Liquid Chromatography-Mass Spectrometry; LLM = Large Language Model; MD =Molecular Dynamics; ML =Machine Learning; MPO =Multiparametric Optimization; mRNA = Messenger Ribonucleic Acid; NCD =Non-Communicable Disease; NEPAD = New Partnership for Africa’s Development; NGO = Non-Governmental Organization; NIAID =National Institute of Allergy and Infectious Diseases; NIH = National Institutes of Health; PGI = Pathogen Genomics Initiative; PZQ =Praziquantel; QSAR =Quantitative Structure-Activity Relationship; R& D =Research and Development; Rg = Radius of Gyration; RLS =Resource-Limited Settings; RMSD = Root Mean Square Deviation; RNA =Ribonucleic Acid; SARS-CoV-2 =Severe Acute Respiratory Syndrome Coronavirus 2; Sm. =Schistosoma mansoni ;STI =Sexually Transmitted Infection; TB =Tuberculosis; TBV = Transmission-Blocking Vaccine; TLR4 = Toll-Like Receptor 4; TRP = Transient Receptor Potential; WHO =World Health Organization
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
All data generated or analyzed during this study are included in this published article.
Ethics Statement
Ethical approval was not required for this study.
Funding Information
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
References
- Jensen N, Barry A, Kelly AH. More-than-national and less-than-global: The biochemical infrastructure of vaccine manufacturing. Economy and Society. 2022;52(1):9-36. doi: https://doi.org/10.1080/03085147.2022.2087899
- Strange R. The 2020 Covid-19 pandemic and global value chains. Journal of Industrial and Business Economics [Internet]. 2020 Jun 8;47.
- Endeshaw D, Dessie G, Kasa AS, Endalamaw A, Tesfaye TD, Birhanu S, et al. Consistent condom utilization among sexually active HIV positive individuals in Sub-Saharan Africa: systematic review and meta-analysis. Sci Rep. 2024;14(1). doi: https://doi.org/10.1038/s41598-024-56574-5
- Sibomana O, Bukuru J, Saka SA, Uwizeyimana MG, Kihunyu AM, Obianke A, et al. Routine malaria vaccination in Africa: a step toward malaria eradication?. Malar J. 2025;24(1). doi: https://doi.org/10.1186/s12936-024-05235-z
- Li S, Mensah E, Liu M, Pan L, Lu W, Zhou S, et al. The burden of tuberculosis and drug resistance in 22 Sub-Saharan African countries, 1990–2021: a GBD 2021 analysis and progress towards WHO 2035 targets with projections to 2050. Front. Microbiol. 2025;16. doi: https://doi.org/10.3389/fmicb.2025.1695592
- Schweitzer A, Horn J, Mikolajczyk RT, Krause G, Ott JJ. Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review of data published between 1965 and 2013. The Lancet. 2015;386(10003):1546-1555. doi: https://doi.org/10.1016/s0140-6736(15)61412-x
- Elfatimi E, Lekbach Y, Prakash S, BenMohamed L. Artificial intelligence and machine learning in the development of vaccines and immunotherapeutics—yesterday, today, and tomorrow. Front. Artif. Intell. 2025;8. doi: https://doi.org/10.3389/frai.2025.1620572
- Black S, Madhi SA, Rappuoli R. Artificial intelligence in the development of vaccines for infectious diseases. The Lancet Infectious Diseases. 2026;26(3):221-222. doi: https://doi.org/10.1016/s1473-3099(25)00497-9
- Thalange AV, Patil AR, Athavale VA. A Review of Artificial Intelligence and Machine Learning for Vaccine Research. Lecture notes in electrical engineering. 2024 Jan 1;85-101.
- Chen H, Engkvist O, Wang Y, Olivecrona M, Blaschke T. The rise of deep learning in drug discovery. Drug Discovery Today. 2018;23(6):1241-1250. doi: https://doi.org/10.1016/j.drudis.2018.01.039
- Zhavoronkov A, Ivanenkov YA, Aliper A, Veselov MS, Aladinskiy VA, Aladinskaya AV, et al. Deep learning enables rapid identification of potent DDR1 kinase inhibitors. Nat Biotechnol. 2019;37(9):1038-1040. doi: https://doi.org/10.1038/s41587-019-0224-x
- Jumper J, Evans R, Pritzel A, Green T, Figurnov M, Ronneberger O, et al. Highly Accurate Protein Structure Prediction with Alphafold. Nature [Internet]. 2021 Jul 15;596(7873):583–9.
- da Silva RGL. The advancement of artificial intelligence in biomedical research and health innovation: challenges and opportunities in emerging economies. Global Health. 2024;20(1). doi: https://doi.org/10.1186/s12992-024-01049-5
- Qimin Z. Science and Technology Innovation and International Biomedical Development. Singapore: Springer Nature Singapore; 2023. doi: https://doi.org/10.1007/978-981-99-1199-8_11
- Dzobo K, Adotey S, Thomford NE, Dzobo W. Integrating Artificial and Human Intelligence: A Partnership for Responsible Innovation in Biomedical Engineering and Medicine. OMICS: A Journal of Integrative Biology. 2020;24(5):247-263. doi: https://doi.org/10.1089/omi.2019.0038
- Shah R, Dey D, Pietzonka T, Obeng P, Ashiru B, Schiestl M, et al. Determinants of Use of Biotherapeutics in sub-Saharan Africa. Trends in Pharmacological Sciences. 2021;42(2):75-84. doi: https://doi.org/10.1016/j.tips.2020.11.012
- Adebisi YA, Nwogu IB, Alaran AJ, Badmos AO, Bamgboye AO, Rufai BO, et al. Revisiting the issue of access to medicines in Africa: Challenges and recommendations. Public Health Challenges. 2022;1(2). doi: https://doi.org/10.1002/puh2.9
- Adebayo Olusegun Aderibigbe, Peter Efosa Ohenhen, Nwabueze Kelvin Nwaobia, Joachim Osheyor Gidiagba, Emmanuel Chigozie Ani. Artificial intelligence in developing countries: bridging the gap between potential and implementation. Comput. sci. IT res. j. 2023;4(3):185-199. doi: https://doi.org/10.51594/csitrj.v4i3.629
- Onunga J. From Readiness to Maturity: Institutional Pathways for Responsible AI Transformation in Africa. The Journal of Digital Transformation in Africa. 2025 Dec 29;1(1):28-39.
- Kyambade M, Namatovu A. Health-care leaders’ perspectives on AI implementation in Uganda: overcoming barriers, driving innovation and strategic considerations. Leadership in Health Services. 2025;38(3):442-463. doi: https://doi.org/10.1108/lhs-02-2025-0025
- Mayoka G, Cheuka PM. Medicinal chemistry capacity in Sub‑Saharan Africa. J Med Chem. 2025.
- Asin-Garcia E, Fawcett JD, Batianis C, Martins dos Santos VA. A snapshot of biomanufacturing and the need for enabling research infrastructure. Trends in Biotechnology. 2025;43(5):1000-1014. doi: https://doi.org/10.1016/j.tibtech.2024.10.014
- Kissami I, Basmadjian R, Chakir O, Abid MR. TOUBKAL: a high-performance supercomputer powering scientific research in Africa. J Supercomput. 2025;81(15). doi: https://doi.org/10.1007/s11227-025-07858-8
- Ramalli E, Pernici B. Challenges of a Data Ecosystem for scientific data. Data & Knowledge Engineering. 2023;148:102236. doi: https://doi.org/10.1016/j.datak.2023.102236
- Aron S, Chauke PA, Ras V, Panji S, Johnston K, Mulder N. The Development of a Sustainable Bioinformatics Training Environment Within the H3Africa Bioinformatics Network (H3ABioNet). Front. Educ. 2021;6. doi: https://doi.org/10.3389/feduc.2021.725702
- Van De Pas R, Widdowson MA, Ravinetto R, N Srinivas P, Ochoa TJ, Fofana TO, et al. COVID-19 vaccine equity: a health systems and policy perspective. Expert Review of Vaccines. 2021;21(1):25-36. doi: https://doi.org/10.1080/14760584.2022.2004125
- Doke-Bagade PE, Bagade O. Applications of AI-Based Models in the Field of Biomedicine. Singapore: Springer Nature Singapore; 2024. doi: https://doi.org/10.1007/978-981-97-2596-0_12
- Dara S, Dhamercherla S, Jadav SS, Babu CM, Ahsan MJ. Machine Learning in Drug Discovery: A Review. Artif Intell Rev. 2021;55(3):1947-1999. doi: https://doi.org/10.1007/s10462-021-10058-4
- Cheng J, Liang T, Xie XQ, Feng Z, Meng L. A new era of antibody discovery: an in-depth review of AI-driven approaches. Drug Discovery Today. 2024;29(6):103984. doi: https://doi.org/10.1016/j.drudis.2024.103984
- Namba-Nzanguim CT, Turon G, Simoben CV, Tietjen I, Montaner LJ, Efange SMN, et al. Artificial intelligence for antiviral drug discovery in low resourced settings: A perspective. Front. Drug. Discov. 2022;2. doi: https://doi.org/10.3389/fddsv.2022.1013285
- Savitha S, Keerthana R, Logeswaran K, Keerthika P, Sharmila V, Sangeetha M. Integration of Multi-Omics Data. : IGI Global; 2025. doi: https://doi.org/10.4018/979-8-3693-9521-9.ch006
- Rennie ML, Oliver MR. Emerging frontiers in protein structure prediction following the AlphaFold revolution. J. R. Soc. Interface. 2025;22(225). doi: https://doi.org/10.1098/rsif.2024.0886
- Kwofie SK, Adams J, Broni E, Enninful KS, Agoni C, Soliman MES, et al. Artificial Intelligence, Machine Learning, and Big Data for Ebola Virus Drug Discovery. Pharmaceuticals. 2023;16(3):332. doi: https://doi.org/10.3390/ph16030332
- Turon G, Njoroge M, Mulubwa M, et al. AI for pharmacogenetic research in Africa. Nature. 2024.
- Saied AA, Metwally AA, Dhawan M, Choudhary OP, Aiash H. Strengthening vaccines and medicines manufacturing capabilities in Africa: challenges and perspectives. EMBO Mol Med. 2022;14(8). doi: https://doi.org/10.15252/emmm.202216287
- Kannan S, Subbaram K, Faiyazuddin M. Artificial intelligence in vaccine development: Significance and challenges ahead.: Elsevier; 2023. doi: https://doi.org/10.1016/b978-0-323-89925-3.00017-4
- Ghosh A, Larrondo-Petrie MM, Pavlovic M. Revolutionizing Vaccine Development for COVID-19: A Review of AI-Based Approaches. Information. 2023;14(12):665. doi: https://doi.org/10.3390/info14120665
- El Arab RA, Alkhunaizi M, Alhashem YN, Al Khatib A, Bubsheet M, Hassanein S. Artificial intelligence in vaccine research and development: an umbrella review. Front. Immunol. 2025;16. doi: https://doi.org/10.3389/fimmu.2025.1567116
- Bekkering ET, Yoo R, Hailemariam S, Heide F, Ivanochko D, Jackman M, et al. Cryo-EM structure of endogenous Pfs230:Pfs48/45 complex with six antibodies reveals mechanisms of malaria transmission-blocking activity. Immunity. 2025;58(11):2899-2916.e10. doi: https://doi.org/10.1016/j.immuni.2025.09.014
- Gandvi D, Jyotishi C, Patel M, Gupta R. In silico design of a multi-epitope pan vaccine targeting Schistosoma species. Genom. Inform. 2025;23(1). doi: https://doi.org/10.1186/s44342-025-00053-4
- Jin R, Zhang L. AI applications in HIV research: advances and future directions. Frontiers in Microbiology. 2025 Feb 20;16.
- Hare J, Nielsen M, Kiragga A, Ochiel D. Sustainable integration of artificial intelligence and machine learning approaches within the African infectious disease vaccine research and development ecosystem. Front. Pharmacol. 2024;15. doi: https://doi.org/10.3389/fphar.2024.1499079
- Koirala M, Yan L, Mohamed Z, DiPaola M. AI-Integrated QSAR Modeling for Enhanced Drug Discovery: From Classical Approaches to Deep Learning and Structural Insight. Ijms. 2025;26(19):9384. doi: https://doi.org/10.3390/ijms26199384
- Tropsha A, Isayev O, Varnek A, Schneider G, Cherkasov A. Integrating QSAR modelling and deep learning in drug discovery: the emergence of deep QSAR. Nat Rev Drug Discov. 2023;23(2):141-155. doi: https://doi.org/10.1038/s41573-023-00832-0
- Gangwal A, Ansari A, Ahmad I, Azad AK, Kumarasamy V, Subramaniyan V, et al. Generative artificial intelligence in drug discovery: basic framework, recent advances, challenges, and opportunities. Front. Pharmacol. 2024;15. doi: https://doi.org/10.3389/fphar.2024.1331062
- Ferreira FJN, Carneiro AS. AI-Driven Drug Discovery: A Comprehensive Review. ACS Omega. 2025;10(23):23889-23903. doi: https://doi.org/10.1021/acsomega.5c00549
- Uddin MR, Sovon MSI, Mondal S, Ahmed S, Al-Mizan MA, Aktar F, et al. Artificial Intelligence and Machine Learning in Pharmaceutical Sciences: Unpacking Regulatory Guidance, Opportunities, and Challenges for Safe and Effective Drug Development. 2025;. doi: https://doi.org/10.2139/ssrn.5505018
- Spangenberg T, editor. Praziquantel: Discovery and Development of an Anthelmintic Drug [Internet]. Cham: Springer Nature Switzerland; 2026 [cited 2025 Oct 8].
- Chevalier FD, Le Clec’h W, Berriman M, Anderson TJ. A single locus determines praziquantel response in Schistosoma mansoni. Antimicrob Agents Chemother. 2024;68(3). doi: https://doi.org/10.1128/aac.01432-23
- Park SK, Friedrich L, Yahya NA, Rohr CM, Chulkov EG, Maillard D, et al. Mechanism of praziquantel action at a parasitic flatworm ion channel. Sci. Transl. Med. 2021;13(625). doi: https://doi.org/10.1126/scitranslmed.abj5832
- Le Clec’h W, Chevalier FD, Mattos ACA, Strickland A, Diaz R, McDew-White M, et al. Genetic analysis of praziquantel response in schistosome parasites implicates a transient receptor potential channel. Sci. Transl. Med. 2021;13(625). doi: https://doi.org/10.1126/scitranslmed.abj9114
- Park SK, Gunaratne GS, Chulkov EG, Moehring F, McCusker P, Dosa PI, et al. The anthelmintic drug praziquantel activates a schistosome transient receptor potential channel. Journal of Biological Chemistry. 2019;294(49):18873-18880. doi: https://doi.org/10.1074/jbc.ac119.011093
- Anokian E, Bernett J, Freeman A, List M, Santamaría LP, Tanoli Z, et al. Machine Learning and Artificial Intelligence in Drug Repurposing—Challenges and Perspectives. Drug Repurposing. 2024;1(1). doi: https://doi.org/10.58647/drugrepo.24.1.0004
- Tanoli Z, Fernández-Torras A, Özcan UO, Kushnir A, Nader KM, Gadiya Y, et al. Computational drug repurposing: approaches, evaluation of in silico resources and case studies. Nat Rev Drug Discov [Internet]. 2025 July [cited 2025 Oct 28];24(7):521–42.
- Betow JY, Turon G, Metuge CS, Akame S, Shu VA, Ebob OT, et al. The Chemical Space Spanned by Manually Curated Datasets of Natural and Synthetic Compounds with Activities against SARS‐CoV‐2. Molecular Informatics. 2024;44(1). doi: https://doi.org/10.1002/minf.202400293
- Raslan MA, Raslan SA, Shehata EM, Mahmoud AS, Sabri NA. Chemoinformatics Advances from Egypt: A Review Article. . 2023. doi: https://doi.org/10.20944/preprints202304.1095.v1
- Ibe C, Otu AA, Mnyambwa NP. Advancing disease genomics beyond COVID-19 and reducing health disparities: what does the future hold for Africa?. Briefings in Functional Genomics. 2022;22(3):241-249. doi: https://doi.org/10.1093/bfgp/elac040
- Cisse C, Wele M, Shaffer JG, Li J, Whalen C, Giovanni MY, et al. Building Sustainable Bioinformatics Training Programs in Africa: Insights from ACE-Mali. Curr. J. Appl. Sci. Technol. 2025;44(8):186-198. doi: https://doi.org/10.9734/cjast/2025/v44i84599
- Turon G, Tse E, Qiu X, Todd M, Duran-Frigola M. Open Source Code Contributions to Global Health: The Case of Antimalarial Drug Discovery. ACS Med. Chem. Lett. 2024;15(9):1645-1650. doi: https://doi.org/10.1021/acsmedchemlett.4c00131
- Turon G, Hlozek J, Woodland JG, Kumar A, Chibale K, Duran-Frigola M. First fully-automated AI/ML virtual screening cascade implemented at a drug discovery centre in Africa. Nat Commun. 2023;14(1). doi: https://doi.org/10.1038/s41467-023-41512-2
- Sankhe S, Fofana FG, Heiba W, Ogunyemi OM, Masisi K, Muiruri I, et al. BioStruct-Africa’s capacity building workshops as a model for advancing the emerging community of structural biologists in Africa. Commun Chem. 2025;8(1). doi: https://doi.org/10.1038/s42004-025-01519-w
- Sovereign AI Capacity in Africa: Beyond Technology Transfer Through Lessons from India [Internet]. Arrey; [cited 2025 Dec 9].
- Bansal R, Singh R. Steroidal Pyrazolines As a Promising Scaffold in Drug Discovery. Future Med. Chem. 2020;12(10):949-959. doi: https://doi.org/10.4155/fmc-2019-0325
- Galiwango R, Whalen CJ, Kebirungi G, Atwine MT, Kimera R, Ssekagiri A, et al. Ten simple rules for building and maintaining sustainable high-performance computing infrastructure for research in resource-limited settings. PLoS Comput Biol. 2025;21(9):e1013481. doi: https://doi.org/10.1371/journal.pcbi.1013481
- Namba-Nzanguim CT, Turon G, Simoben CV, Tietjen I, Montaner LJ, Efange SMN, et al. Artificial intelligence for antiviral drug discovery in low resourced settings: A perspective. Front. Drug. Discov. 2022;2. doi: https://doi.org/10.3389/fddsv.2022.1013285
- Thakkar S, Slikker W, Yiannas F, Silva P, Blais B, Chng KR, et al. Artificial intelligence and real-world data for drug and food safety – A regulatory science perspective. Regulatory Toxicology and Pharmacology. 2023;140:105388. doi: https://doi.org/10.1016/j.yrtph.2023.105388
- Levin-Epstein M. FDA Issues Draft Guidance of AI-Enabled Devices. J Clin Eng. 2025;50(2):50-50. doi: https://doi.org/10.1097/jce.0000000000000694
- Turon G, Arora D, Duran-Frigola M. Infectious Disease Research Laboratories in Africa Are Not Using AI Yet─Large Language Models May Facilitate Adoption. ACS Infect. Dis. 2024;10(9):3083-3085. doi: https://doi.org/10.1021/acsinfecdis.4c00585
- Shonhe L, Min Q, Phuti R. Government AI readiness in the ESARBICA community: findings from the Oxford Insights AI Readiness Index 2022. ESARBICA. 2024;43.
- Turon G, Njoroge M, Mulubwa M, Duran-Frigola M, Chibale K. AI can help to tailor drugs for Africa — but Africans should lead the way. Nature. 2024;628(8007):265-267. doi: https://doi.org/10.1038/d41586-024-01001-y
- Hare J, Nielsen M, Kiragga A, Ochiel D. Sustainable integration of artificial intelligence and machine learning approaches within the African infectious disease vaccine research and development ecosystem. Front. Pharmacol. 2024;15. doi: https://doi.org/10.3389/fphar.2024.1499079
- Ogunye RO, Egwuatu D, Anene PC, Azubuike EO, Asenuga OO, Sargwak JP, et al. The Impact of Emerging Technologies on Pharmaceutical Process Design and Optimization in Africa: A Review. J. Pharm. Res. Int. 2024;36(9):46-60. doi: https://doi.org/10.9734/jpri/2024/v36i97576
- Amewu RK, Amoateng P, Arthur PK, Asare P, Asiamah I, Boamah D, et al. Drug discovery research in Ghana, challenges, current efforts, and the way forward. PLoS Negl Trop Dis. 2022;16(9):e0010645. doi: https://doi.org/10.1371/journal.pntd.0010645
- Fallah MP, Dereje N, Temfack E, Byonanebye DM, Weyer J, Mutombo PN, et al. A call for health R&D prioritisation and governance mechanisms in Africa. The Lancet. 2025;405(10482):889-890. doi: https://doi.org/10.1016/s0140-6736(25)00242-9
- Africa CDC. Africa CDC and AUDA- NEPAD established diagnostic advisory committee (DAC) to Harmonize regulatory processes for Invitro diagnostics (Ivds) and to accelerate access to diagnostics for the health security of the continent [Internet]. Africa CDC; [cited 2025 Oct 11].
- Yimer SA, Booij BB, Tobert G, Hebbeler A, Oloo P, Brangel P, et al. Rapid diagnostic test: a critical need for outbreak preparedness and response for high priority pathogens. BMJ Glob Health. 2024;9(4):e014386. doi: https://doi.org/10.1136/bmjgh-2023-014386
- Vora LK, Gholap AD, Jetha K, Thakur RRS, Solanki HK, Chavda VP. Artificial Intelligence in Pharmaceutical Technology and Drug Delivery Design. Pharmaceutics. 2023;15(7):1916. doi: https://doi.org/10.3390/pharmaceutics15071916
- Thomas S, Abraham A, Baldwin J, Piplani S, Petrovsky N. Artificial Intelligence in Vaccine and Drug Design. New York, NY: Springer US; 2021. doi: https://doi.org/10.1007/978-1-0716-1884-4_6
- Blanco-González A, Cabezón A, Seco-González A, Conde-Torres D, Antelo-Riveiro P, Piñeiro Á, et al. The Role of AI in Drug Discovery: Challenges, Opportunities, and Strategies. Pharmaceuticals. 2023;16(6):891. doi: https://doi.org/10.3390/ph16060891
- Alowais SA, Alghamdi SS, Alsuhebany N, Alqahtani T, Alshaya AI, Almohareb SN, et al. Revolutionizing healthcare: the role of artificial intelligence in clinical practice. BMC Med Educ. 2023;23(1). doi: https://doi.org/10.1186/s12909-023-04698-z
- Odone A, Barbati C, Amadasi S, Schultz T, Resnik DB. Artificial intelligence and infectious diseases: an evidence-driven conceptual framework for research, public health, and clinical practice. The Lancet Infectious Diseases. 2026;26(3):e152-e167. doi: https://doi.org/10.1016/s1473-3099(25)00412-8
- Zhao RC, Yuan X. AI in Healthcare for Resource Limited Settings: An Exploration and Ethical Evaluation. Companion Proceedings of the ACM on Web Conference 2025. 2025;:1953-1960. doi: https://doi.org/10.1145/3701716.3717747
- Hlozek J, Chibale K, Woodland JG. Ongoing Implementation and Prospective Validation of Artificial Intelligence/Machine Learning Tools at an African Drug Discovery Center. ACS Med. Chem. Lett. 2024;15(7):989-993. doi: https://doi.org/10.1021/acsmedchemlett.4c00243
- Nji E, Traore DAK, Ndi M, Joko CA, Doyle DA. BioStruct-Africa: empowering Africa-based scientists through structural biology knowledge transfer and mentoring – recent advances and future perspectives. J Synchrotron Rad. 2019;26(5):1843-1850. doi: https://doi.org/10.1107/s1600577519008981
- Ouologuem DT, Maiga FO, Dara A, Djimdé A, Traore DAK, Nji E. Hands-on training in structural biology, a tool for sustainable development in Africa series 4. Biology Open. 2022;11(8). doi: https://doi.org/10.1242/bio.059487
- Ade-Ibijola A, Okonkwo C. Artificial Intelligence in Africa: Emerging Challenges. Cham: Springer International Publishing; 2023. doi: https://doi.org/10.1007/978-3-031-08215-3_5
- Silcox C, Zimlichmann E, Huber K, Rowen N, Saunders R, McClellan M, et al. The potential for artificial intelligence to transform healthcare: perspectives from international health leaders. npj Digit. Med. 2024;7(1). doi: https://doi.org/10.1038/s41746-024-01097-6
- Crick Africa Network [Internet]. Crick. 2025 [cited 2026 Feb 19]. Available from: https://www.crick.ac.uk/research/research-partnerships/research-partnerships/crick-africa-network
- SANTHE (Sub-Saharan African Network for TB/HIV Research Excellence) [Internet]. SANTHE (Sub-Saharan African Network for TB/HIV Research Excellence). 2025 [cited 2026 Feb 19].
- Kamali A, Price MA, Lakhi S, Karita E, Inambao M, Sanders EJ, et al. Creating an African HIV Clinical Research and Prevention Trials Network: HIV Prevalence, Incidence and Transmission. PLoS ONE. 2015;10(1):e0116100. doi: https://doi.org/10.1371/journal.pone.0116100
- ElZarrad MK, Lee AY, Purcell R, Steele SJ. Advancing an agile regulatory ecosystem to respond to the rapid development of innovative technologies. Clinical Translational Sci. 2022;15(6):1332-1339. doi: https://doi.org/10.1111/cts.13267
- Kaushik R, Kant R, Christodoulides M. Artificial intelligence in accelerating vaccine development - current and future perspectives. Front. Bacteriol. 2023;2. doi: https://doi.org/10.3389/fbrio.2023.1258159
- Olawade DB, Teke J, Fapohunda O, Weerasinghe K, Usman SO, Ige AO, et al. Leveraging artificial intelligence in vaccine development: A narrative review. Journal of Microbiological Methods. 2024;224:106998. doi: https://doi.org/10.1016/j.mimet.2024.106998
- Khalid K, Poh CL. The Promising Potential of Reverse Vaccinology-Based Next-Generation Vaccine Development over Conventional Vaccines against Antibiotic-Resistant Bacteria. Vaccines. 2023;11(7):1264. doi: https://doi.org/10.3390/vaccines11071264
- Mellado B, Wu J, Kong JD, Bragazzi NL, Asgary A, Kawonga M, et al. Leveraging Artificial Intelligence and Big Data to Optimize COVID-19 Clinical Public Health and Vaccination Roll-Out Strategies in Africa. Ijerph. 2021;18(15):7890. doi: https://doi.org/10.3390/ijerph18157890
