RESEARCH ARTICLE
Physico-Chemical Characterisation and Determination of Luteolin Content of Red Pidada (Sonneratia Caseolaris L.) Leaf Extract
Academic Editor: Garnadi Jafar
Sciences of Pharmacy|Vol. 5, Issue 2, pp. 138-144 (2026)
Received
Jan 31, 2026Revised
Mar 1, 2026Accepted
Apr 13, 2026Published
Apr 28, 2026
Abstract
Red Pidada (Sonneratia caseolaris L.) leaves are widely used in traditional medicine, particularly for skin care and treatment of minor ailments. This study aimed to characterise the powdered leaf material and determine luteolin content of the ethanolic leaf extract. Dried leaf powder was extracted by maceration using ethanol. The powdered material was characterised through macroscopic and microscopic evaluations, as well as determining physicochemical parameters including water-soluble extractive value, ethanol-soluble extractive value, moisture content, total ash, and acid-insoluble ash. Furthermore, metabolite profiling of the extract was conducted using high-resolution liquid chromatography–mass spectrometry (HR-LCMS) to identify constituents. Luteolin content was quantified using thin-layer chromatography (TLC) densitometry on silica gel 60 F254 plates with a mobile phase of n-hexane:ethyl acetate (2:3). Macroscopically, the leaves were oval to elongated, with rounded to blunt apices, entire margins, pinnate venation, and dark-green colour. Microscopic examination revealed upper and lower epidermal tissues, stomata, and vascular bundles. The powdered leaves exhibited a bitter taste. The water-soluble and ethanol-soluble extractive values were 11% and 16%, respectively, while moisture content, total ash, and acid-insoluble ash were 8.8%, 8.12%, and 0.47%. HR-LCMS analysis produced 38 tentative compound matches, including luteolin. Under specified conditions, TLC-densitometry showed an Rf value of 0.54 for luteolin, with a mean amount of 3.849 µg. These findings provide baseline physicochemical and phytochemical data for standardisation of S. caseolaris L. leaf extract for future research applications and quality control purposes in pharmaceutical and herbal product development to ensure consistency safety and efficacy across batches.
Introduction
Red Pidada (Sonneratia caseolaris L.) is a mangrove species traditionally used in Indonesia for food, household use, and ethnomedicine. The fruit and leaves are used in traditional preparations, including cold powder and scar remedies, and various parts of Sonneratia species have been used to treat conditions such as fever, sprains, haemorrhoids, and skin disorders (1, 2). These uses indicate ethnopharmacological relevance, but traditional use alone is not sufficient to support scientific standardisation of the plant material.
Among Dayak communities in Borneo, S. caseolaris L is also used in a traditional cosmetic preparation known as "bedak dingin," which is sold in local markets in East Kalimantan. This practice has encouraged interest in the phytochemical constituents of the species. Previous studies have shown that S. caseolaris L leaves contain phenolic and flavonoid compounds and exhibit antioxidant and other bioactivities, while isolated constituents from this species include luteolin and luteolin 7-O-β-glucoside (4-6). However, previous reports have mainly emphasised biological activity screening or metabolite detection. Fewer studies have focused on integrating pharmacognostic and physicochemical characterisation of the leaf raw material with a simple marker-based assay that may support preliminary laboratory standardisation.
This gap is important because the development of herbal raw materials requires not only evidence of biological relevance but also reproducible physicochemical and analytical data. In particular, studies that combine pharmacognostic and physicochemical characterisation of S. caseolaris L leaves with quantitative determination of a defined marker compound in the extract remain limited. The present study, therefore, differs from previous reports by integrating leaf characterisation, extract profiling, and targeted marker quantification within one analytical framework.
Luteolin was selected as the marker compound because it has previously been reported in S. caseolaris L, belongs to a pharmacologically relevant flavonoid class, and possesses a conjugated chromophore that facilitates densitometric detection (5, 11-13). Other flavonoids, including quercetin derivatives, may also be relevant, but luteolin was prioritised here because it was directly traceable in prior reports on S. caseolaris L and was considered analytically suitable as a preliminary marker for a low-cost TLC-based assay. In the context of herbal standardisation, a marker compound should be chemically identifiable, analytically measurable, and relevant to the phytochemical profile of the species.
Although HPLC-DAD and LC-MS offer higher instrumental sensitivity, TLC-densitometry remains a practical option for routine herbal analysis because it is simpler, more economical, requires relatively small solvent volumes, and enables direct comparison of sample and reference standard on the same plate (8, 14-18). We hypothesised that powdered S. caseolaris L leaves would show measurable physicochemical characteristics and that luteolin could be detected and quantified in the ethanolic leaf extract using TLC-densitometry. Therefore, this study aimed to characterise powdered S. caseolaris L leaves through physicochemical analysis and to determine the luteolin content of the ethanolic leaf extract using TLC-densitometry.
Materials and Methods
Materials
The materials used in this study included S. caseolaris L leaves, 95% ethanol, distilled water, magnesium powder, iron (III) chloride, amyl alcohol, n-hexane, and ethyl acetate. The equipment included standard glassware, filter paper, a glass funnel, vials, cuvettes, a watch glass, gloves, a porcelain dish, an analytical balance, a blender, aluminium foil, a 60-mesh sieve, a stir bar, a micropipette, silica gel 60 F254 TLC plates, a chamber, a densitometer, and a UV-Vis spectrophotometer (Shimadzu UV-1800).
Plant Collection, Identification, and Preparation
Leaves of S. caseolaris L were collected in January 2022 from Sanga-sanga, Kutai Kartanegara, East Kalimantan, Indonesia. The plant material was identified by a botanist in the Biology Department, Faculty of Mathematics and Natural Sciences, Mulawarman University, Indonesia. A voucher specimen number was not reported in the available study record, which limits traceability and should be corrected in future studies.
The collected leaves were cleaned under running water, cut into smaller pieces, and dried in the shade for approximately one week. The dried leaves were then weighed, ground into powder, and passed through a 60-mesh sieve.
Macroscopic and Microscopic Characterisation of Sonneratia caseolaris L Leaves
Macroscopic examination was carried out as part of the pharmacognostic evaluation of the leaf material. Microscopic examination of leaf sections was performed at 400x magnification to observe the upper epidermis, lower epidermis with stomata, and vascular bundles.
Physicochemical Evaluation of the Powdered Leaves
Physicochemical evaluation comprised determination of the water-soluble extractive value, ethanol-soluble extractive value, moisture content, total ash, and acid-insoluble ash by routine gravimetric procedures commonly used in herbal quality control (9). Because the available manuscript record did not preserve full step-by-step procedural conditions, the present description should be regarded as a descriptive summary rather than a complete pharmacopeial validation protocol.
Extraction of the Plant Material
The powdered plant material was extracted by maceration in 95% ethanol for 72 h and filtered through Whatman No. 1 filter paper. The filtrate was concentrated under reduced pressure at 40 °C and stored at 4 °C until analysis. The study was conducted on one laboratory extract batch. Exact sample mass, solvent-to-material ratio, extraction yield, and the number of independent extraction batches were not documented in sufficient detail for full reporting, which limits reproducibility and should be addressed in future confirmatory work.
HR-LCMS Metabolite Profiling
Tentative metabolite profiling was performed by high-resolution liquid chromatography-mass spectrometry (HR-LCMS). However, the instrument model, column specification, ionisation mode, mobile phase composition, gradient programme, scan range, resolution settings, calibration procedure, and blank-run data were not retained in the available manuscript record. Consequently, the HR-LCMS output is interpreted here only as preliminary database-assisted annotation and not as definitive compound identification. Compound assignments based on mzCloud similarity scores require confirmation by MS/MS fragmentation analysis, retention behaviour, authentic reference standards, and contamination controls.
Determination of Luteolin Content by TLC-Densitometry
Luteolin content in the S. caseolaris L extract was determined by TLC-densitometry (8). This method was selected as a pragmatic preliminary approach for herbal standardisation because it permits direct comparison of the sample and reference compound on the same plate, requires relatively simple instrumentation, and is more accessible for routine laboratory screening than HPLC-DAD or LC-MS. The mobile phase n-hexane: ethyl acetate (2: 3) was selected empirically because it produced an acceptable luteolin band under the present laboratory conditions; however, systematic optimisation of the mobile phase was not performed. Method evaluation in this study was limited to analytical repeatability. Specificity, peak purity, limit of detection, limit of quantification, robustness, recovery, and inter-day precision were not established and should be addressed in future validation studies. The calibration range of 1-5 mg/mL was used as the working range adopted in this preliminary assay and should not be interpreted as a fully optimised, validated range.
Preparation of the Extract Solution
An appropriate amount of the extract was dissolved in ethanol to obtain a concentration of 10 mg/mL.
Preparation of Standard Luteolin Solutions
Standard luteolin solutions were prepared at concentrations of 1, 2, 3, 4, and 5 mg/mL to construct a preliminary calibration curve for the working assay range.
Spotting and Development: Each luteolin standard solution was applied to a silica gel 60 F254 TLC plate. The plate was developed using n-hexane: ethyl acetate (2:3) as the mobile phase.
Densitometric Analysis: After development, the TLC plate was analysed using a densitometer at 254 nm to quantify the luteolin spots.
Study Design and Statistical Analysis
This study was descriptive and exploratory. All quantitative values are presented as mean, standard deviation (SD), and coefficient of variation (CV) from four analytical replicate measurements of one prepared extract solution. No inferential statistical analysis was applied because the study did not include group comparisons or biological replicate batches.
Results and Discussion
Physicochemical Characteristics
Macroscopic examination showed that the leaves of S. caseolaris L were oval to elongated, with rounded to blunt apices, entire margins, pinnate venation, and a dark-green colour. The average leaf size was 8.5 cm in length and 4 cm in width. Microscopic examination (Figure 1) showed the upper and lower epidermal tissues, stomata, and vascular bundles.
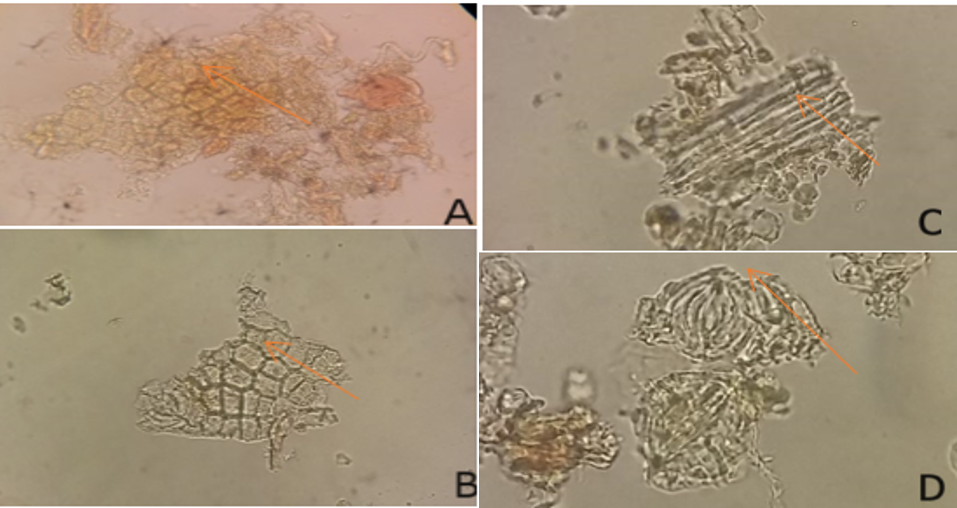
Table 1 presents the water-soluble and ethanol-soluble extractive values of the powdered S. caseolaris L leaves. The water-soluble extractive value was 11%, whereas the ethanol-soluble extractive value was 16%. Under the conditions used in this study, the higher ethanol-soluble extractive value suggests that ethanol recovered a greater mass of soluble material than water in this preparation. However, because extraction yield, replicate batch comparisons, and comparative phytochemical profiling were not available, this result should not be interpreted as definitive evidence that ethanol is universally the better solvent for this species. The physicochemical values in Table 1 were reported from the available manuscript record as single determinations, so no replicate SD values were available for these parameters.
The non-specific physicochemical parameters are also shown in Table 1. The moisture content was 9%, which is within the commonly accepted limit of less than 10% for herbal raw materials. Adequate control of moisture content is important because excessive water can promote microbial growth and accelerate the degradation of bioactive constituents.
| Parameter | Content (%) |
|---|---|
| Specific parameters | |
| Water-soluble extractive value | 11 |
| Ethanol-soluble extractive value | 16 |
| Non-specific parameters | |
| Moisture content | 9 |
| Total ash | 8.17 |
| Acid-insoluble ash | 0.48 |
| No | Name | mzCloud | No | Name | mzCloud |
|---|---|---|---|---|---|
| 1 | Perfluorooctanoic acid (PFOA) | 99.6 | 20 | Galaxolidone | 93.3 |
| 2 | Bis (2-ethylhexyl) phthalate | 99.8 | 21 | 9 (Z), 11 (E)-Conjugated linoleic acid | 91.9 |
| 3 | Dodecyl sulfate | 96.7 | 22 | 4-(2, 3-dihydro-1, 4-benzodioxin-6-yl)butanoic acid | 92.6 |
| 4 | Erucamide | 81.8 | 23 | Perfluoroheptanoic acid | 83.9 |
| 5 | Perfluorooctanoic acid (PFOA) | 99.5 | 24 | Palmitic Acid | 84 |
| 6 | Bis (2-ethylhexyl) phthalate | 99.8 | 25 | 1-Stearoylglycerol | 96 |
| 7 | 4-Dodecylbenzenesulfonic acid | 99.4 | 26 | 4-Ethoxy ethylbenzoate | 94.1 |
| 8 | 2, 2, 3-Trifluoro-3-(1, 1, 2, 2, 3, 3-hexafluoro-3-trifluoromethoxy-propoxy)-propionic acid | 85.1 | 27 | NP-021797 | 71.1 |
| 9 | Myristyl sulfate | 96 | 28 | AL 8810 | 89.6 |
| 10 | Ostruthin | 94.8 | 29 | Hexadecanamide | 94.2 |
| 11 | Dibutyl phthalate | 99.7 | 30 | Docosahexaenoic acid | 98.6 |
| 12 | cis-7-Hexadecenoic acid | 95.6 | 31 | NP-001596 | 93.7 |
| 13 | Oleic acid | 95 | 32 | Oleamide | 77.7 |
| 14 | Quercetin 3-rutinoside | 98.3 | 33 | Luteolin | 99.7 |
| 15 | Dodecanedioic acid | 89.6 | 34 | NP-018716 | 84.9 |
| 16 | 1-[(1S, 4S)-4-(1-Benzyl-3, 5-dimethyl-1H-pyrazol-4-yl)-2-cyclopenten-1-yl]-3-isopropylurea | 75.4 | 35 | Ostruthin | 93.4 |
| 17 | Lidocaine | 99.3 | 36 | 2, 2, 6, 6-Tetramethyl-1-piperidinol | 91.8 |
| 18 | Resolvin E1 | 85.8 | 37 | Betaine | 96.7 |
| 19 | Ethyl myristate | 95 | 38 | Choline | 97.6 |
Total ash and acid-insoluble ash reflect the overall inorganic residue and siliceous matter in the powdered sample and do not provide element-specific information on heavy metals. In this study, the total ash content was 8.17%, and the acid-insoluble ash content was 0.48%, indicating the presence of mineral and non-volatile inorganic residues in the leaf powder. Tentative HR-LCMS annotation generated 38 database matches (Table 2); however, these assignments must be interpreted cautiously. Several matches corresponded to compounds such as phthalates, perfluorinated substances, and lidocaine, which are more plausibly associated with contamination, carry-over, plasticware, solvents, or imperfect database matching than with authentic plant metabolites. Moreover, the mzCloud score represents spectral similarity rather than abundance. Therefore, the present LC-MS output cannot be used to conclude that luteolin was the most abundant phytoconstituent. At most, it indicates that luteolin was one of the compounds tentatively matched in the extract, consistent with previous reports that S. caseolaris L contains flavonoid constituents, including luteolin-related compounds (4, 6). Definitive confirmation would require complete chromatographic conditions, blank runs, MS/MS fragmentation analysis, and authentic standards.
TLC-Densitometric Analysis
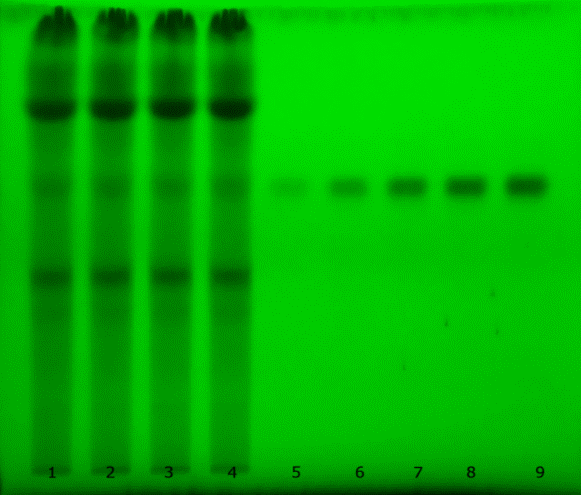
TLC analysis showed that the extract produced a band that migrated in parallel with the luteolin standard. The observed Rf value for luteolin in the extract was 0.54, indicating presumptive correspondence under the selected chromatographic conditions. Figure 2 shows that the chosen mobile phase produced a compact band at the same Rf across replicate sample lanes, supporting the practical suitability of the system for preliminary screening. However, co-migration on TLC alone does not prove chemical identity, and confirmation by orthogonal methods remains necessary.
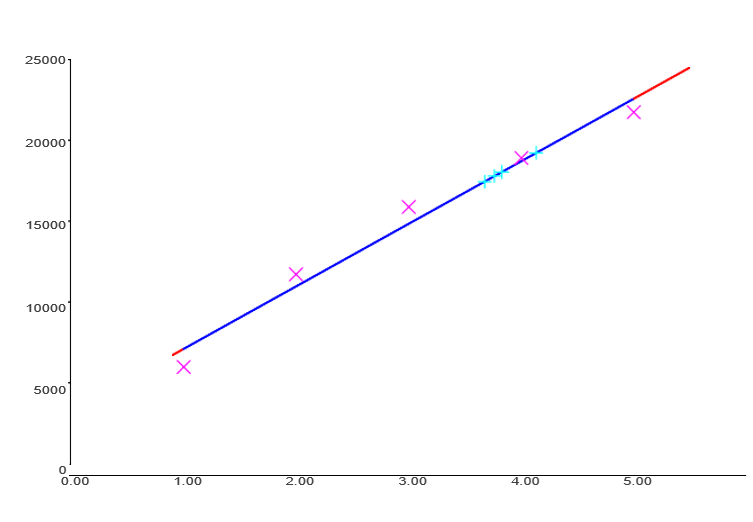
Quantification was carried out by TLC-densitometry using five concentrations of luteolin standard (16-18). Linear regression of peak area against concentration produced the equation Y = 3496 + 3791X with a correlation coefficient (r) of 0.98803, corresponding to R² ≈ 0.976 as presented in Figure 3. This indicates a reasonably linear response within the tested working range. Nevertheless, the linearity assessment remains limited because residual analysis, lack-of-fit testing, and evaluation of LOD and LOQ were not performed.
Only analytical repeatability was evaluated in this study (19, 20). Precision was expressed as the standard deviation (SD) and coefficient of variation (CV) obtained from repeated measurements of the same prepared sample solution. Accuracy, recovery, robustness, and specificity were not examined and therefore cannot be claimed for the present method.
| Replication | Luteolin content (µg) |
|---|---|
| 1 | 3.873 |
| 2 | 3.859 |
| 3 | 3.791 |
| 4 | 3.872 |
| Mean | 3.849 |
| SD | 0.039 |
| Coefficient of variation (CV) % | 1.014 |
Table 3 presents four analytical replicate measurements from the same prepared sample solution. The mean measured luteolin amount was 3.849 µg with an SD of 0.039 and a CV of 1.014%, indicating acceptable repeatability at the measurement level. However, the result should be interpreted as a preliminary assay outcome because the basis of reporting 3.849 µg and its conversion to 5.171% was not fully elaborated in the available analytical record. Accordingly, the value describes the analysed preparation under the present method conditions rather than a fully validated absolute concentration in the plant material. Direct comparison with previous studies is also limited because earlier reports on S. caseolaris L. mainly described compound isolation, qualitative profiling, or biological activity rather than validated quantitative TLC-densitometric determination of luteolin.
Study Limitations
This study has several limitations. The extraction section lacks complete batch-level documentation, such as exact sample mass, solvent-to-material ratio, extraction yield, and independent extraction replicates. A voucher specimen number and full proximate analysis procedures were not reported. The HR-LCMS results are tentative because instrumental conditions, blank runs, and fragmentation-based confirmation were unavailable, and some signals may reflect contamination or database misassignment. The TLC assay was evaluated only for analytical repeatability and not for specificity, recovery, robustness, LOD, or LOQ. No antioxidant or anti-ageing assay data were generated in the final results, so no biological efficacy claim can be made from the present dataset. In addition, raw chromatograms and LC-MS spectra were not deposited in a public repository, which limits independent re-evaluation.
Conclusion
This study provides preliminary physicochemical and analytical data for S. caseolaris L leaves and their ethanolic extract. The powdered leaves showed a water-soluble extractive value of 11%, an ethanol-soluble extractive value of 16%, a moisture content of 9%, a total ash content of 8.17%, and an acid-insoluble ash content of 0.48%. Under the TLC-densitometric conditions used, a band consistent with luteolin was observed at Rf 0.54, and the mean measured luteolin amount was 3.849 ± 0.039 µg in the analysed extract preparation. These findings support the use of luteolin as a tentative analytical marker for preliminary quality assessment, not as definitive evidence of pharmacological efficacy. Further work should include authenticated voucher specimens, complete extraction records, contamination controls, validated quantitative analysis, and direct biological assays if antioxidant or anti-ageing claims are to be advanced.
Data Availability Statement
All tabulated data supporting the findings of this study are included in the manuscript. Original raw chromatograms, LC-MS spectra, and detailed laboratory worksheets were not deposited in a public repository and are therefore unavailable for independent re-analysis.
Abbreviations
TLC = Thin-Layer Chromatography; HR-LCMS = High-Resolution Liquid Chromatography-Mass Spectrometry; AUC = Area under the curve; SD = Standard deviation; CV = Coefficient of variation; Rf = Retardation factor.
Declarations
Acknowledgment
The Research Team would like to thank the Directorate of Research and Community Dedication, Ministry of Research and Technology, the National Research and Innovation Agency, Indonesia for funding this research on the Applied Research Scheme with SPPK contract number: 191/SP2H/AMD/LT/DRPM/2019 November 12, 2019.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
Data sharing is available upon reasonable request to the corresponding author.
Ethics Statement
Ethical approval was not required for this study.
Funding Information
The Research Team would like to thank the Directorate of Research and Community Dedication, Ministry of Research and Technology, the National Research and Innovation Agency, Indonesia for funding this research on the Applied Research Scheme with SPPK contract number: 191/SP2H/AMD/LT/DRPM/2019 November 12, 2019.
References
- Shadu SK, Ahmed F, Ohtsuki T, Ishibashi M. Flavonoids from Sonneratia caseolaris. J Nat Med. 2006;60(3):264–265.
- Syamsul ES, Umar FS, Wahyuni RM, Lestari D, Hamidi D. HR-LCMS-based metabolite profiling and anti-collagenase properties of ethanolic extract of Sonneratia caseolaris: computational and in vitro study. Int J Appl Pharm. 2023;15(1):34–38.
- Jadoon S, Karim S, Bin Asad MH, Akram MR, Khan AK, Malik A, et al. Anti-aging potential of phytoextract-loaded pharmaceutical creams for human skin cell longevity. Oxid Med Cell Longev. 2015;2015:709628.
- Syamsul ES, Umar S, Wahyuni FS, Martien R, Hamidi D. Anti-aging activity, in silico modelling and molecular docking from Sonneratia caseolaris. Open Access Maced J Med Sci. 2022;10(A):1471–1477.
- Syamsul ES, Supomo JS, Wijaya H, Lestari D, Poddar S. Antioxidant activity test of Sonneratia caseolaris leaves using the ABTS method. Res J Pharm Technol. 2022;15(9):3957–3961.
- Arung ET, Kuspradini H, Kusuma IW, Bang TH, Yamashita S, Katakura Y, et al. Effects of isolated compounds from Sonneratia caseolaris leaves: validation of traditional utilisation by melanin biosynthesis and antioxidant assays. J Appl Pharm Sci. 2015;5(10):39–43.
- Avenido P, Serrano AE. Effects of the apple mangrove (Sonneratia caseolaris) on growth, nutrient utilisation, and digestive enzyme activities of Penaeus monodon postlarvae. Eur J Exp Biol. 2012;2(5):1603–1608.
- Hahn ED. Applied thin-layer chromatography. Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA; 2018.
- Balekundri A, Mannur V. Quality control of traditional herbs and herbal products: a review. Future J Pharm Sci. 2020;6:67.
- Van Thuoc D, Mai NT, Le Thi Viet Ha L, Hung DH, Hung NK, Hung NP. Evaluation of antibacterial, antioxidant, and anti-obese activities of fruit juice of crabapple mangrove Sonneratia caseolaris. Int J Agric Sci. 2018;5(2).
- Wang Z, Zeng M, Wang Z, Qin F, Chen J, He Z. Dietary luteolin: a narrative review focusing on its pharmacokinetic properties and effects on glycolipid metabolism. J Agric Food Chem. 2021;69(5):1441–1454.
- Wu B, Song H, Fan M, You F, Zhang L, Luo J, et al. Luteolin attenuates sepsis-induced myocardial injury by enhancing autophagy in mice. Int J Mol Med. 2020;45(5):1477–1487.
- Li B, Du P, Du Y, Zhao D, Cai Y, Yang Q, et al. Luteolin alleviates inflammation and modulates gut microbiota in ulcerative colitis rats. Life Sci. 2021;269:119008.
- Ege M. The hidden danger in phytopharmaceuticals: adulteration. In: Phytopharmaceuticals: potential therapeutic applications. 2021. p. 77–98.
- European Medicines Agency. Validation of analytical procedures: ICH guidelines Q2(R2). Farm Glas. 2022;2:1–34.
- Fichou D, Morlock GE. QuanTLC, an online open-source solution for videodensitometric quantification. J Chromatogr A. 2018;1560:78–81.
- Syamsul ES, Lestari D. Research methodology and pharmaceutical statistics: with the SPSS application. Samarinda: RV Pustaka Horizon; 2020.
- Gumieniczek A, Inglot T, Kończak A. Classical densitometry and videoscanning in a validated method for analysis of candesartan and losartan in pharmaceuticals. J Planar Chromatogr. 2011;24:99–104.
- Pratiwi A, Sari R, Widyawaruyanti A. TLC-densitometry analysis method of asiaticoside assay and antioxidant activity of Centella asiatica (L.) Urban extract. Trop J Pharm Res. 2023;22(9):4003–4009.
- Harmita. Petunjuk pelaksanaan validasi metode dan cara perhitungannya. Jakarta: Departemen Farmasi FMIPA UI; 2004.