RESEARCH ARTICLE
Green-Synthesized Silver Nanoparticles Using Erythrina subumbrans Leaf Extract: Optimization and Antibacterial Activity
Academic Editor: Garnadi Jafar
Sciences of Pharmacy|Vol. 5, Issue 2, pp. 112-122 (2026)
Received
Feb 1, 2026Revised
Mar 1, 2026Accepted
Apr 7, 2026Published
Apr 21, 2026
Abstract
Keywords:
Introduction
Indonesia is rich in medicinal plants with long-standing traditional uses, one of which is Erythrina subumbrans (Hassk. ) Merr., commonly known as dadap serep. This plant grows widely in tropical and subtropical regions, including Indonesia, the Philippines, Thailand, and Malaysia (1). In traditional medicine, the leaves and stem bark of E. subumbrans are frequently utilized. The leaves have been empirically used to treat fever, postpartum fever, inflammation, bleeding, abdominal pain, and to promote lactation, while the stem bark is traditionally used as an expectorant (2). Pharmacological studies have reported that E. subumbrans leaves exhibit antipyretic, anti-inflammatory, antioxidant, and antibacterial activities (3). These biological activities are attributed to the presence of various secondary metabolites, including phenolics, tannins, alkaloids, flavonoids, saponins, and steroids (4). Previous studies demonstrated that extracts of E. subumbrans showed antibacterial activity against g-positive bacteria, with minimum inhibitory concentrations ranging from 2 to 4 μg/mL (5).
Beyond their antibacterial properties, secondary metabolites in E. subumbrans leaves also possess substantial reducing and stabilizing capabilities, making them promising natural bioreductants for the green synthesis of silver nanoparticles. Compounds such as flavonoids, phenolics, alkaloids, and tannins are known to facilitate the reduction of silver ions (Ag⁺) to elemental silver (Ag⁰) while simultaneously stabilizing the formed nanoparticles (6, 7). Several studies have successfully employed plant extracts as bioreductants in silver nanoparticle synthesis, including extracts from Areca catechu L. (8), Musa paradisiaca (9), Imperata cylindrica L. (10), Dendrobium crumenatum (6), and E. subumbrans stem bark (4), supporting the feasibility of plant-mediated nanoparticle synthesis. The stem of E. subumbrans has been previously reported to contain flavonoids, saponins, isoflavonoids, alkaloids, and lectins that function as effective bioreductants in nanosilver biosynthesis. Earlier studies demonstrated that variation in stem extract concentration significantly influenced AgNP formation, yielding SPR peaks at 429–436 nm and particle sizes ranging from 66.13 to 75.96 nm with PDI values below 0.5, indicating uniform nanoparticle distribution and optimal stability at a 1: 1 extract-to-AgNO₃ ratio (3). Despite these findings, existing research has been exclusively limited to the stem part of E. subumbrans, leaving the bioreductive potential of its leaves entirely unexplored.
Nanoparticles are materials with particle sizes ranging from 1 to 100 nm and exhibit unique physicochemical properties (11). Among various metal and metal oxide nanoparticles, silver nanoparticles (AgNPs) have attracted considerable attention due to their superior antibacterial activity, even at low concentrations, compared to zinc oxide and gold nanoparticles (12–20). The strong antibacterial efficacy of AgNPs makes them promising candidates for addressing the growing concern of antibiotic resistance. Silver nanoparticles can be synthesized through physical, chemical, and biological methods. Although physical and chemical processes can produce nanoparticles rapidly, they often require high energy input, toxic chemicals, and generate hazardous by-products. In contrast, green synthesis using biological resources, particularly plant extracts, offers an environmentally friendly, cost-effective, and non-toxic alternative. In this approach, natural bioreductants facilitate the reduction of Ag⁺ ions through electron transfer mechanisms and functional group transformations (21–23).
Synthesis parameters, such as AgNO₃ concentration, temperature, and incubation time, strongly influence the efficiency and characteristics of green-synthesized silver nanoparticles (24). Therefore, optimization of these parameters is essential to obtain nanoparticles with desirable physicochemical properties and enhanced antibacterial activity. It is proposed that the aqueous extract of E. subumbrans leaves functions as a natural bioreductant and capping agent during green silver nanoparticle synthesis, thereby generating nanoparticles with optimized physicochemical attributes and improved stability. This dual role is expected to influence particle size, morphology, and surface characteristics, which are critical factors affecting biological activity. Based on these considerations, this study aimed to optimize the green synthesis of silver nanoparticles using E. subumbrans leaf extract as a natural bioreductant through a Box–Behnken experimental design implemented in Design-Expert® software. The optimization focused on key synthesis parameters to achieve maximum efficiency and reproducibility of nanoparticle formation. Furthermore, the antibacterial activity of the optimized silver nanoparticles against Propionibacterium acnes was systematically evaluated to determine their potential as alternative antimicrobial agents. The results of this study are expected to contribute to the development of eco-friendly and sustainable antibacterial nanomaterials derived from indigenous medicinal plants, as well as provide a scientific basis for their application in pharmaceutical and dermatological formulations.
Methodology
Equipment
The instruments used in this study were an analytical balance (Pioneer PX Series, OHAUS, Germany/USA), a hot plate magnetic stirrer (DLAB MSH280-Pro, China), an autoclave, an incubator (Memmert, Germany), a laminar air flow cabinet (Biobase, China), a UV–visible spectrophotometer (Thermo Scientific Genesys 10S, USA), a Fourier transform infrared (FTIR) spectrometer (Bruker Alpha II, Germany), a particle size analyzer (SZ-100, HORIBA Scientific, Japan), an X-ray diffractometer (Bruker D8 Advance, USA), and a scanning electron microscope equipped with energy-dispersive X-ray spectroscopy (SEM-EDX; Carl Zeiss EVO 10, Germany).
Materials
The materials used in this study were deionized water (OneMed®), E. subumbrans (Hassk. ) Merr. leaves, silver nitrate (AgNO₃; Merck®, Germany), and the test bacterium Propionibacterium acnes. The reagents employed included Mayer’s reagent, Dragendorff’s reagent, hydrochloric acid, sulfuric acid, Liebermann–Burchard reagent, 10% sodium hydroxide solution, chloroform, and ferric chloride (all supplied by PT Kimia Jaya Labora, Indonesia).
Methods
Preparation of Erythrina subumbrans Leaf Extract
Leaves of E. subumbrans (Hassk. ) Merr. were collected from Mendalo Indah Village, Muaro Jambi Regency, Jambi Province, Indonesia, and taxonomically identified at Universitas Padjadjaran, Indonesia, as confirmed by a certificate of determination (No. 56/HB/12/2025). The leaves were cleaned by wet sorting, washed briefly with running water, cut into uniform pieces, and dried at 60 °C for 48 h. The dried material was powdered, sieved through a 60-mesh sieve, and used for extraction. Extraction was performed by the infusion method by heating 0.75 g of leaf powder in 100 mL of deionized water (0.75% w/v) at approximately 90 °C for 15 min with occasional stirring. The mixture was cooled, filtered, and the obtained filtrate was used directly for silver nanoparticle synthesis (4).
Phytochemical screening
Phytochemical screening was conducted to qualitatively determine the presence of flavonoids, saponins, alkaloids, tannins, phenolics, and steroids/triterpenoids in the extract using standard procedures. The presence of flavonoids was confirmed through the Shinoda test, indicated by the formation of a pink to reddish coloration after the addition of magnesium and hydrochloric acid. Saponins were identified based on the formation of stable foam following vigorous shaking and acidification. Alkaloids were detected using Dragendorff’s and Mayer’s reagents, which yielded orange and cream precipitates, respectively. Tannins and phenolic compounds were verified using the ferric chloride test, characterized by the appearance of dark blue, green, or black coloration. Steroids and triterpenoids were assessed using the Liebermann–Burchard reaction, indicated by the development of green or bluish coloration. These qualitative analyses provide preliminary evidence of the presence of bioactive compounds that may contribute to the reduction and stabilization processes during nanoparticle synthesis (2).
Optimization of Silver Nanoparticle Synthesis
Optimization of silver nanoparticle synthesis was conducted using response surface methodology with a Box–Behnken design in Design-Expert® version 13 by varying AgNO₃ concentration, temperature, and incubation time. Each factor was evaluated at two levels (low and high), as shown in Table 1, with the experimental design comprising 17 runs (Table 2). Silver nanoparticles were synthesized by mixing E. subumbrans leaf extract and AgNO₃ solution at a 1: 1 ratio and heating under the specified conditions. Nanoparticle formation was indicated by a color change from yellow to brown. At the same time, optical responses, including absorbance, surface plasmon resonance (SPR) peak, and transmittance, were analyzed using UV–Visible spectroscopy (200–800 nm) to determine the optimal synthesis conditions. Optimum synthesis conditions were determined using response surface methodology with a Box–Behnken design in Design-Expert® version 13, optimizing response goals within defined limits; desirability values approaching 1 indicated optimal silver nanoparticle synthesis.
| Parameter | Coded Level –1 | Coded Level +1 |
|---|---|---|
| AgNO3 Concentration (mM) | 1 | 3 |
| Incubation Time (h) | 1 | 3 |
| Temperature (°C) | 50 | 90 |
| Run | AgNO₃ Concentration (mM) | Incubation Time (h) | Temperature (°C) | Absorbance | SPR (nm) | Transmittance (%) |
|---|---|---|---|---|---|---|
| 1 | 1 | 3 | 70 | 0.95549 | 422 | 79.2 |
| 2 | 3 | 2 | 90 | 2.54913 | 424 | 62.1 |
| 3 | 2 | 1 | 90 | 2.20896 | 424 | 69.7 |
| 4 | 2 | 3 | 90 | 2.50004 | 430 | 65.2 |
| 5 | 2 | 2 | 70 | 1.75821 | 422 | 70.1 |
| 6 | 3 | 1 | 70 | 1.53181 | 422 | 73.3 |
| 7 | 3 | 2 | 50 | 3.18294 | 418 | 54.7 |
| 8 | 2 | 2 | 70 | 2.33894 | 422 | 62.6 |
| 9 | 1 | 2 | 90 | 2.67786 | 420 | 68.6 |
| 10 | 2 | 2 | 70 | 2.02441 | 422 | 69.0 |
| 11 | 2 | 1 | 50 | 3.31106 | 422 | 54.1 |
| 12 | 2 | 2 | 70 | 2.15886 | 422 | 68.0 |
| 13 | 1 | 2 | 50 | 1.88862 | 422 | 64.5 |
| 14 | 1 | 1 | 70 | 1.26003 | 420 | 69.8 |
| 15 | 2 | 3 | 50 | 3.00805 | 418 | 61.0 |
| 16 | 2 | 2 | 70 | 2.74586 | 418 | 48.6 |
| 17 | 3 | 3 | 70 | 2.95405 | 424 | 36.0 |
Characterization of Optimized Silver Nanoparticles
The optimized silver nanoparticles were characterized for their optical properties, size distribution, surface charge, functional groups, morphology, and crystalline structure. Absorbance, surface plasmon resonance (SPR), and transmittance were analyzed by UV–Visible spectroscopy over 200–800 nm, with transmittance measured at 650 nm. Particle size, polydispersity index (PDI), and zeta potential were determined using a particle size analyzer (PSA). Functional groups involved in nanoparticle formation were identified by Fourier transform infrared (FTIR) spectroscopy in the range of 400–4000 cm⁻¹ after freeze-drying. Prior to this step, the nanoparticle suspension was centrifuged at 5000 rpm for 30 min, the supernatant was removed, and the resulting pellet was washed with deionized water to eliminate residual impurities before analysis. Particle morphology was examined using scanning electron microscopy (SEM), while crystalline structure was analyzed by X-ray diffraction (XRD) using Cu-Kα radiation.
Antibacterial Activity Assay
Antibacterial activity against Propionibacterium acnes was evaluated using the Kirby-Bauer disc diffusion method. A bacterial suspension was precisely adjusted to a 0.5 McFarland standard and uniformly inoculated onto sterile nutrient agar plates to create a confluent growth. Sterile 6 mm paper discs were then impregnated with 20 µL of the synthesized silver nanoparticles. For a comprehensive comparative analysis, AgNO₃ solution and Mediklin® Gel 1% were employed as negative and positive controls, respectively, while the pure leaf extract was tested separately to determine its baseline activity. After incubation at 37 °C for 24 h under appropriate conditions, the resulting inhibition zones were measured using a digital caliper. All assays were conducted in triplicate to ensure statistical reliability and results were expressed as mean diameters.
Results and Discussion
Phytochemical screening
Phytochemical screening was conducted to identify secondary metabolites with potential roles in bioreduction during silver nanoparticle synthesis. As shown in Table 3, the aqueous extract of E. subumbrans leaves tested positive for flavonoids, tannins, and phenolic compounds, while saponins, alkaloids, and steroids/triterpenoids were not detected. An orange coloration in the Shinoda test indicated the presence of flavonoids. In contrast, positive reactions for tannins and phenolics were confirmed by the formation of dark brown to black color with ferric chloride, suggesting complex formation between Fe³⁺ ions and phenolic hydroxyl groups. The absence of stable foam, precipitate formation, or characteristic color changes evidenced negative results for saponins, alkaloids, and steroids/triterpenoids. These findings are partially consistent with previous studies (1), with variations likely attributed to differences in extraction methods, solvents, and extract concentrations, which may influence metabolite detectability. The phytochemical analysis was limited to qualitative identification without quantitative measurement of metabolite levels. This restricts detailed mechanistic interpretation of their role in Ag-NP synthesis, highlighting the need for quantitative profiling in future studies.
| Phytochemical Test | Reagent | Result | Observation |
|---|---|---|---|
| Flavonoids | Concentrated HCl + Mg powder | + | Formation of orange coloration |
| Saponins | 2 N HCl | − | No stable foam formation |
| Alkaloids | Mayer’s reagent | − | No yellowish-white precipitate formation |
| Tannins | 3% FeCl₃ | + | Formation of dark brown coloration |
| Phenolics | 5% FeCl₃ | + | Formation of dark brown coloration |
| Steroids/Triterpenoids | Liebermann–Burchard reagent | − | No red, orange, or blue coloration |
Optimization of Silver Nanoparticle Synthesis
Silver nanoparticle formation was indicated by a color change from pale yellow to reddish-brown, confirming the reduction of Ag⁺ to Ag⁰. Increasing AgNO₃ concentration, heating temperature, and heating time resulted in a darker solution color, indicating greater nanoparticle formation. Synthesis optimization was performed using response surface methodology with a Box–Behnken design, evaluating absorbance, surface plasmon resonance (SPR) wavelength, and transmittance. The absorbance values ranged from 0.96 to 3.31, with SPR peaks observed at 418–430 nm, consistent with the characteristic SPR region of silver nanoparticles (Table 2). Transmittance values (36–81.7%) showed an inverse relationship with absorbance, in accordance with the Beer–Lambert law, indicating increased nanoparticle formation at higher absorbance levels.
The absorbance response was statistically analyzed using ANOVA for the polynomial response surface model (Table 4). The model was statistically significant (F = 3.36; p = 0.0209), and the non-significant lack-of-fit indicates good agreement between the predicted and experimental values, confirming an adequate model fit. The effect of the independent variables on absorbance was described by a quadratic model, indicating that AgNO₃ concentration (A) had the most significant positive impact, with higher concentrations increasing absorbance (Supplemental Equation 1 and Figure 1).
| Source | Sum of Squares | df | Mean Square | F-value | p-value | Significance |
|---|---|---|---|---|---|---|
| Model | 709.67 | 14 | 50.69 | 3.36 | 0.0209 | significant |
| A-Tem. (C) | 280.33 | 1 | 280.33 | 18.59 | 0.0010 | |
| B-Incubation Period | 0.3333 | 1 | 0.3333 | 0.0221 | 0.8843 | |
| C-AgNO3 Conc. (mM) | 48.00 | 1 | 48.00 | 3.18 | 0.0997 | |
| D-Extract Volume (mL) | 5.33 | 1 | 5.33 | 0.3536 | 0.5631 | |
| AB | 1.00 | 1 | 1.00 | 0.0663 | 0.8012 | |
| AC | 169.00 | 1 | 169.00 | 11.20 | 0.0058 | |
| AD | 1.0000 | 1 | 1.0000 | 0.0663 | 0.8012 | |
| BC | 1.0000 | 1 | 1.0000 | 0.0663 | 0.8012 | |
| BD | 121.00 | 1 | 121.00 | 8.02 | 0.0151 | |
| CD | 16.00 | 1 | 16.00 | 1.06 | 0.3234 | |
| A² | 33.33 | 1 | 33.33 | 2.21 | 0.1629 | |
| B² | 5.33 | 1 | 5.33 | 0.3536 | 0.5631 | |
| C² | 0.3333 | 1 | 0.3333 | 0.0221 | 0.8843 | |
| D² | 16.33 | 1 | 16.33 | 1.08 | 0.3186 | |
| Residual | 181.00 | 12 | 15.08 | |||
| Lack of Fit | 149.00 | 10 | 14.90 | 0.9313 | 0.6220 | not significant |
| Pure Error | 32.00 | 2 | 16.00 | - | - | |
| Cor Total | 890.67 | 26 | - | - | - |
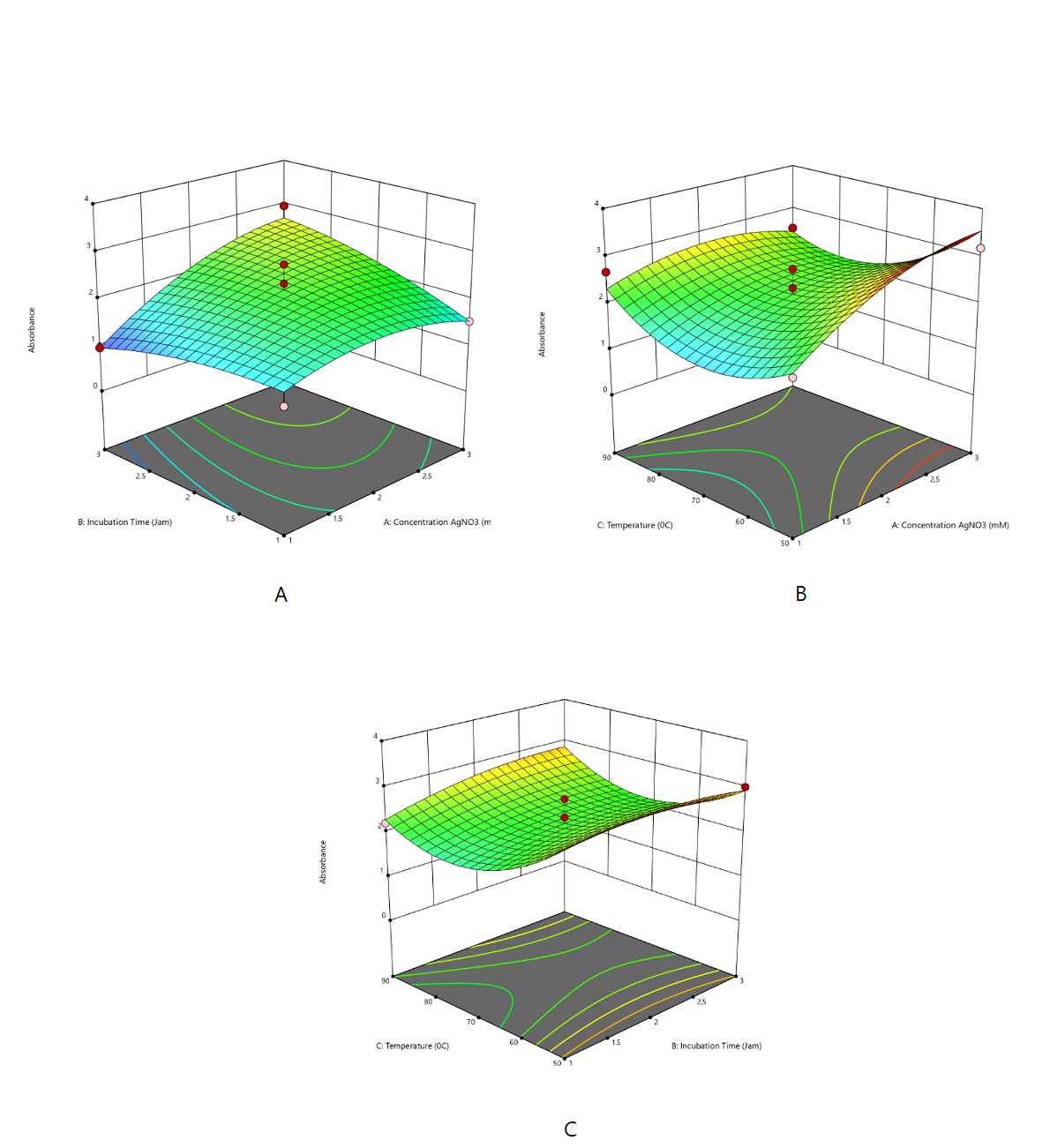
A quadratic model well described the absorbance response. The model indicated that heating temperature (°C) exerted a significant positive effect on the SPR response, shifting the SPR peak toward longer wavelengths (Supplemental Equation 2 and Figure 2).
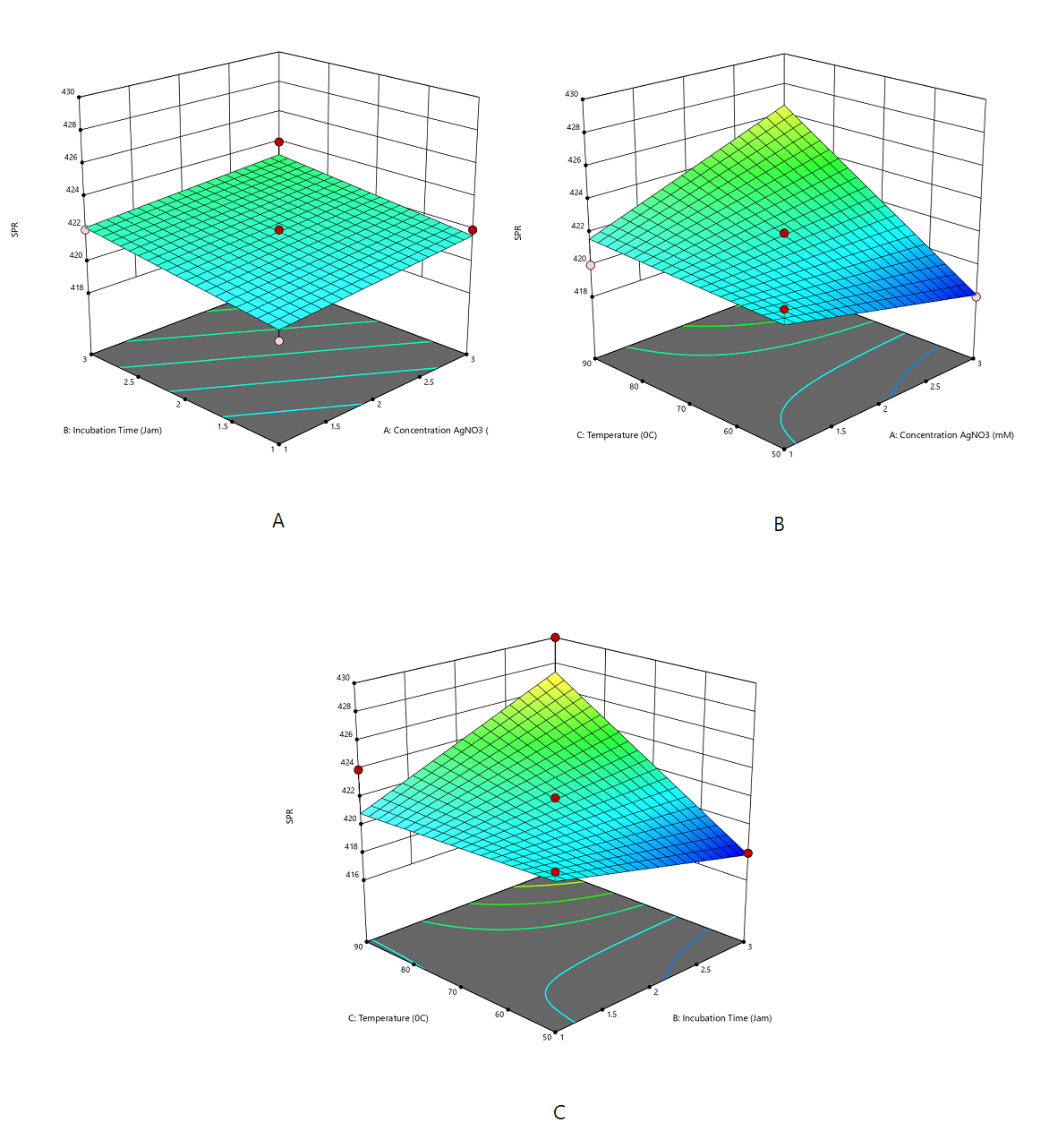
The transmittance response was adequately described by a two-factor interaction (2FI) model. The negative coefficient of AgNO₃ concentration (A) indicated that increasing AgNO₃ concentration decreased transmittance due to enhanced nanoparticle formation and greater light absorption. The negative interaction between AgNO₃ concentration and incubation time (AB) further reduced transmittance, whereas heating temperature (C) exerted a positive effect under specific conditions (Supplemental Equation 3 and Figure 3). Overall, transmittance showed an inverse relationship with absorbance, as expected by the Beer–Lambert law.
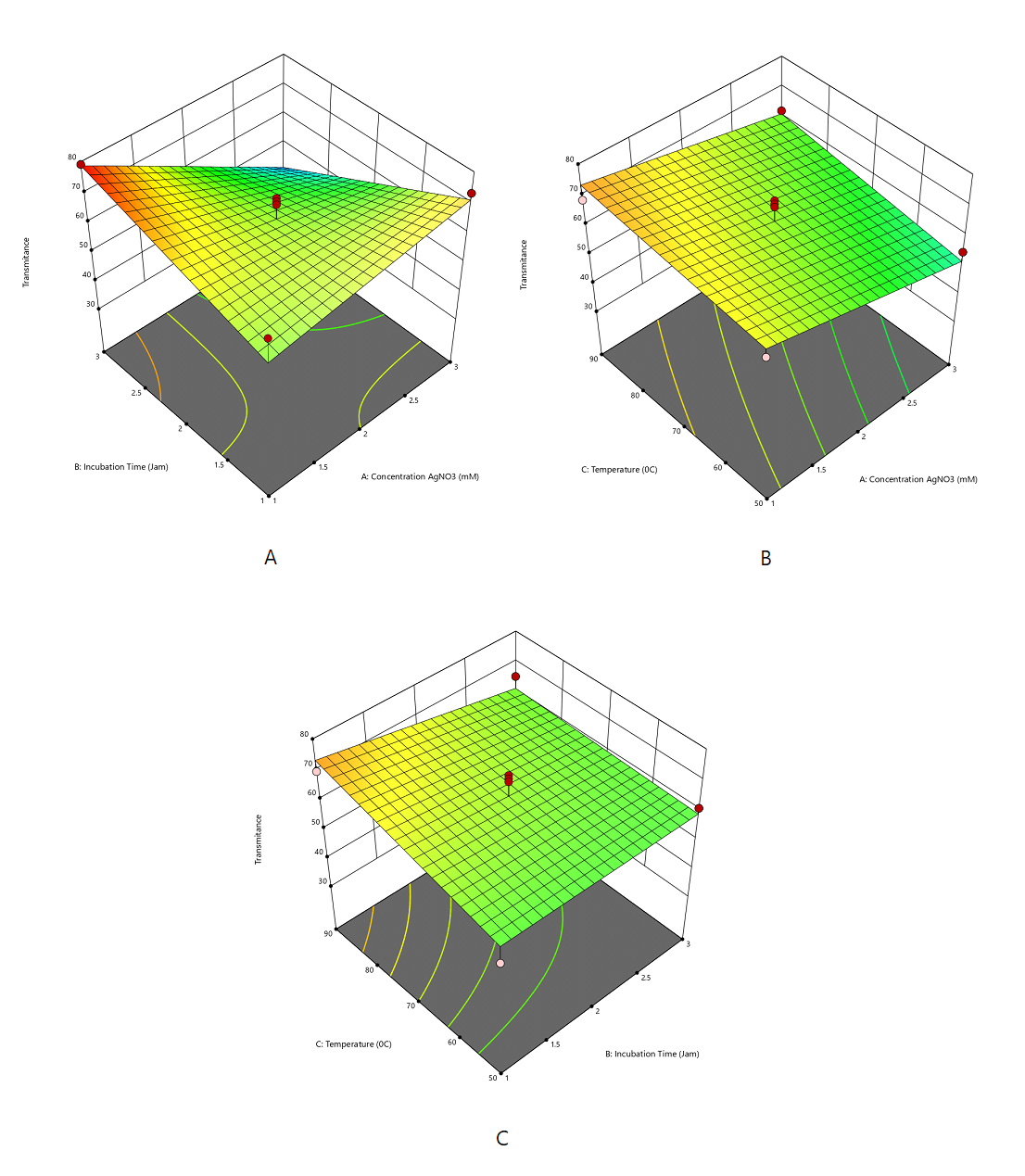
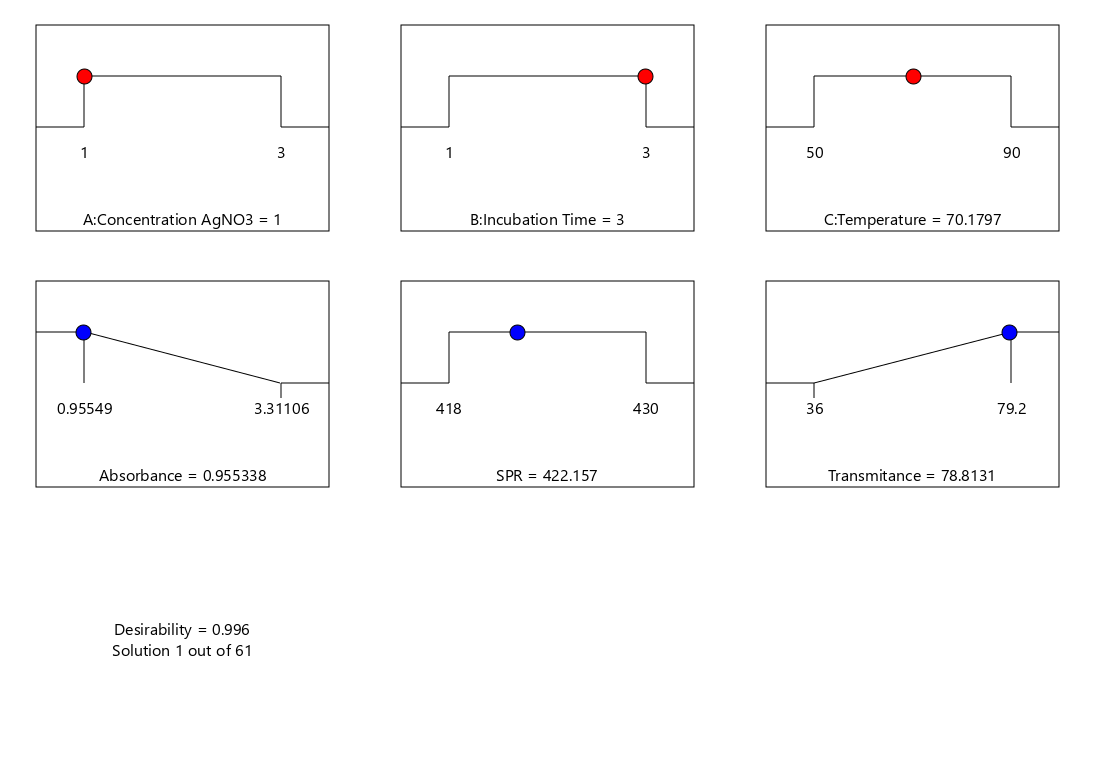
The Design-Expert optimization identified the optimal conditions for Ag-NP synthesis as an AgNO₃ concentration of 1 mM, an incubation time of 3 h, and a temperature of approximately 70 °C, yielding a high desirability value of 0.996 (Figure 4). Under these conditions, the predicted responses were an absorbance of 0.955, an SPR peak at 422.16 nm, and a transmittance of 78.81%, confirming efficient silver nanoparticle formation with favorable optical properties. The experimental validation results showed good agreement with the predicted optimum responses, with all experimental values falling within the 95% confidence interval (CI) of the model predictions. The observed absorbance, SPR peak, and transmittance values were comparable to the predicted results and remained within the characteristic range of silver nanoparticles. These findings confirm the adequacy and predictive reliability of the optimization model at a 95% confidence level.
Characterization of Optimized Silver Nanoparticles
The optimized silver nanoparticles were characterized for particle size, polydispersity index (PDI), and zeta potential using a dynamic light scattering (DLS) particle size analyzer. As shown in Figure 5 and Table 5, the nanoparticles exhibited an average particle size of 53.6 ± 2.3 nm, indicating successful nanoscale formation. The PDI value (0.269 ± 0.058) reflected a narrow and homogeneous size distribution, while the zeta potential (−41.1 ± 0.7 mV) indicated high colloidal stability due to strong electrostatic repulsion. (4) Overall, these results confirm that the biosynthesized silver nanoparticles were stable, well-dispersed, and consistent with standard nanoparticle characteristics.
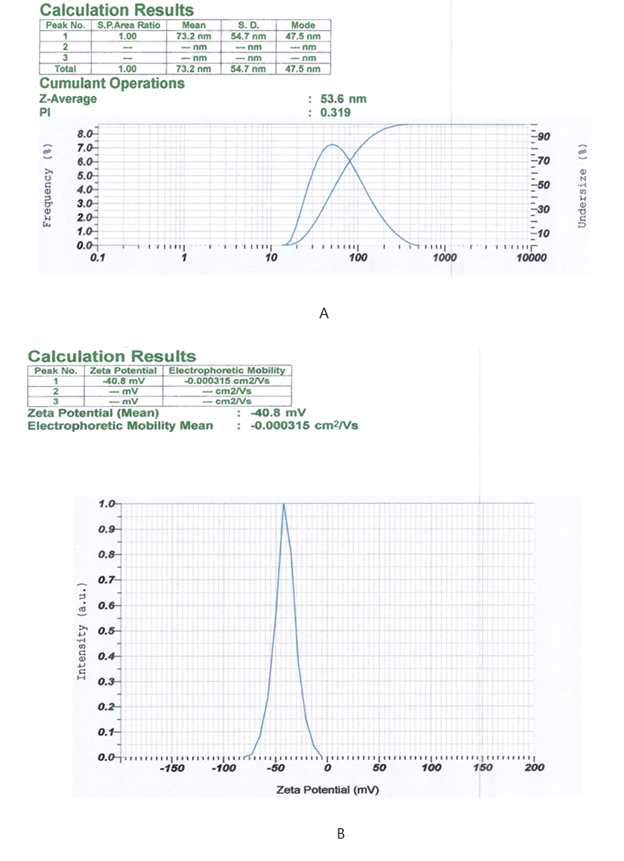
| Replication | Particle size (nm) | PDI | Zeta potential (mV) |
|---|---|---|---|
| 1 | 51.3 | 0.283 | - 40.6 |
| 2 | 55.9 | 0.205 | - 41.9 |
| 3 | 53.6 | 0.319 | - 40.8 |
| Avarege ±SD | 53.6 ± 2.3 | 0.269 ± 0.058 | - 41.1 ± 0.7 |
FTIR analysis was conducted to identify functional groups involved in the synthesis and stabilization of silver nanoparticles. E. subumbrans leaf extract spectrum exhibited a broad absorption band at 3311.68 cm⁻¹, corresponding to –OH group vibrations, typically associated with phenolic and flavonoid compounds (Figure 6A). Additionally, the band at 1634.71 cm⁻¹ indicates the presence of aromatic C=C and/or conjugated C=O groups, which also act as reducing agents in nanoparticle synthesis. After the formation of AgNPs, a shift in the absorption band to 3266.78 cm⁻¹ for the –OH group was observed, indicating interaction between hydroxyl groups and the nanoparticle surface. This shift suggests the involvement of bioactive compounds in the reduction of Ag⁺ to Ag⁰ and in its stabilization. Furthermore, the appearance of a new band at 2917.42 cm⁻¹, corresponding to C–H vibrations, indicates the contribution of organic compounds in the capping of the nanoparticles (25, 26). SEM images revealed irregularly shaped nanoparticles with relatively rough surfaces and a tendency toward agglomeration (Figure 6B), a common feature of plant-mediated green synthesis. The SEM images show nanoparticle agglomeration, which the drying process may influence during sample preparation. Solvent evaporation can induce capillary forces and particle clustering, leading to aggregation that may not reflect the true colloidal dispersion state of the nanoparticles (27, 28). XRD analysis exhibited distinct diffraction peaks corresponding to the (111), (200), (220), (311), and (222) planes (Figure 6C), confirming a face-centered cubic (FCC) crystalline structure consistent with standard silver nanoparticles (JCPDS No. 04-0783). The crystallite size of AgNPs, calculated from the (111), (200), (220), (311), and (222) peaks using the Scherrer equation, is 11.37 nm, confirming nanoscale crystalline silver consistent with green-synthesized AgNPs.
Antibacterial Activity Assay
The antibacterial activity of the optimized silver nanoparticle formulation against Propionibacterium acnes was evaluated using the disc diffusion method. The silver nanoparticles produced a mean inhibition zone diameter of 19.97 ± 0.83 mm, comparable to the positive control, Mediklin® Gel 1% (21.84 ± 1.67 mm), and higher than that of AgNO₃ solution (11.28 ± 5.96 mm) (Table 6). At the same time, the E. subumbrans leaf extract showed no inhibition zone. Statistical analysis revealed significant differences among treatments (p < 0.001), and Duncan’s post hoc test placed the silver nanoparticles in the same subset as the positive control. These results indicate that silver nanoparticle formation significantly enhances antibacterial activity, likely due to nanoscale particle size and increased surface area that promote stronger interactions with bacterial cells (29, 30).
| Specimen | Inhibition Zone Diameter (mm) | Mean ± SD | ||
|---|---|---|---|---|
| Replicate 1 | Replicate 2 | Replicate 3 | ||
| Positive control (Mediklin® Gel 1%) | 20.43 | 21.42 | 23.69 | 21.84 ± 1.67 |
| Negative control (AgNO₃ solution) | 14.56 | 14.90 | 4.41 | 11.28 ± 5.96 |
| Leaf extract | 0 | 0 | 0 | 0 |
| Silver nanoparticles | 19.66 | 20.92 | 19.34 | 19.97 ± 0.83 |
Conclusion
This study demonstrates the successful green synthesis of silver nanoparticles from E. subumbrans leaf extract under optimized conditions, yielding stable, bioactive AgNPs. The synthesized nanoparticles exhibited enhanced antibacterial activity against Propionibacterium acnes, indicating their potential as alternative antimicrobial agents. However, this study is limited to preliminary antibacterial evaluation using the disc diffusion method. Further studies, including MIC/MBC determination, cytotoxicity assessment, and formulation development, are necessary to support their application, particularly in topical treatments such as acne therapy.
Abbreviations
E. subumbrans= Erythrina subumbrans; AgNO3 = Silver Nitrate; AgNPs = Silver Nanoparticle; SEM = scanning electron microscopy; FTIR = Fourier transform infrared spectroscopy; XRD= X-ray diffraction; PSA= Particle Size Analyzer; PDI= Polydispersity index; SPR= surface plasmon resonance;
Declarations
Acknowledgment
The authors gratefully acknowledge the Directorate of Downstreaming and Partnerships, Directorate General of Research and Development, Ministry of Higher Education, Science, and Technology, Republic of Indonesia, for financial support and research facilitation under Contract No. 53/SPK/C.C4/PPK.DHK/VII/2025. The authors also thank all parties who contributed to the completion of this study.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
All data generated or analyzed during this study are included in this published article
Ethics Statement
Ethical approval was not required for this study.
Funding Information
This work was supported by the Directorate of Downstreaming and Partnerships, Directorate General of Research and Development, Ministry of Higher Education, Science, and Technology, Republic of Indonesia, under Grant Number 53/SPK/C.C4/PPK.DHK/VII/2025.
Supplemental Material
This article contains <a class="cursor-pointer" href="https://etflin.com/file/document/20260421025444_856536_abd0c378.docx">supplementary material</a>, including <b>Supplemental Equations 1</b>, <b>2</b>, and <b>3</b>, which are available online.
References
- Susilawati E, Levita J, Susilawati Y, Sumiwi S. Erythrina subumbrans (Hassk) Merr. (Fabaceae) Inhibits Insulin Resistance in the Adipose Tissue of High Fructose-Induced Wistar Rats. Dddt. 2024;Volume 18:3825-3839. doi: https://doi.org/10.2147/dddt.s472660
- Rangkuti SN, Aprilliani A, Sulestari F. Farmagazine. 2023; 10 (2): 17–27.
- Ayu Putu Chika Iswari Anjani S, Ratna Sari Dewi K, Gede Bayu Surya Ekayana I, Chandra Yowani S. Aktivitas Lotion Tabir Surya Ekstrak Daun Dadap Serep (Erythrina subumbrans) dan Daun Kelor (Moringa oleifera L.) Secara In-Vitro. Jurnal Global Ilmiah. 2025;2(4):1-10. doi: https://doi.org/10.55324/jgi.v2i4.179
- Maharini I, Fitrianingsih F, Utami DT. Green synthesis nanoperak menggunakan ekstrak batang dadap serep (Erythrina Subumbrans (Hassk.) Merr). Jps. 2023;6(5):255-259. doi: https://doi.org/10.36490/journal-jps.com.v6i5-si.410
- Phukhatmuen P, Meesakul P, Suthiphasilp V, Charoensup R, Maneerat T, Cheenpracha S, et al. Antidiabetic and antimicrobial flavonoids from the twigs and roots of Erythrina subumbrans (Hassk.) Merr. Heliyon. 2021;7(4):e06904. doi: https://doi.org/10.1016/j.heliyon.2021.e06904
- Panjaitan KN, Syamsurizal S, Maharini I, Putra DOY, Septiana P. Optimasi dan Uji Antibakteri Nanopartikel Perak dengan Bioreduktor Ekstrak Batang Anggrek Merpati (Dendrobium crumenatum Sw.). sinteza. 2025;5(1):18-31. doi: https://doi.org/10.29408/sinteza.v5i1.29574
- Maharini I, Havizur R, Utami DT. The effect of concentration liquid extract gaharu (Aquilaria malaccensis) leaves on green synthesis nanosilver. In: Asian Federation for Pharmaceutical Sciences (AFPS); 2019.
- Bemis R, Deswardani F, Heriyanti H, Puspitasari RD, Azizah N. Green Synthesis of Silver Nanoparticles Using Areca catechu L Peel Bioreductor as an Antibacterial Escherichia coli and Staphylococcus aureus. Ind. J. Chem. Anal. 2023;6(2):176-186. doi: https://doi.org/10.20885/ijca.vol6.iss2.art9
- Fajri N, Putri LFA, Prasetio MR, Azizah N, Pratama Y, Susanto NCA. Potensi Batang Pisang (Musa paradisiaca l) sebagai bioreduktor dalam Green Sintesis Ag nanopartikel. Jps. 2022;24(1):33. doi: https://doi.org/10.56064/jps.v24i1.668
- Zulaicha AS, Saputra IS, Sari IP, Ghifari MA, Yulizar Y, Permana YN, et al. Green Synthesis Nanopartikel Perak (AgNPs) Menggunakan Bioreduktor Alami Ekstrak Daun Ilalang (Imperata cylindrica L). rjna. 2021;1(1):11-19. doi: https://doi.org/10.33369/rjna.v1i1.15588
- Joseph T, Kar Mahapatra D, Esmaeili A, Piszczyk Ł, Hasanin M, Kattali M, et al. Nanoparticles: Taking a Unique Position in Medicine. Nanomaterials. 2023;13(3):574. doi: https://doi.org/10.3390/nano13030574
- Duffy LL, Osmond-McLeod MJ, Judy J, King T. Investigation into the antibacterial activity of silver, zinc oxide and copper oxide nanoparticles against poultry-relevant isolates of Salmonella and Campylobacter. Food Control. 2018;92:293-300. doi: https://doi.org/10.1016/j.foodcont.2018.05.008
- Al-Momani H, Massadeh MI, Almasri M, Al Balawi D, Aolymat I, Hamed S, et al. Anti-Bacterial Activity of Green Synthesised Silver and Zinc Oxide Nanoparticles against Propionibacterium acnes. Pharmaceuticals. 2024;17(2):255. doi: https://doi.org/10.3390/ph17020255
- Joshi V, Bachhar V, Mishra SS, Shukla RK, Duseja M. Hexagonal ZnO nanoparticles synthesised using methanolic extract of Piper chaba aerial parts for catalytic oxidation of morin, 4-nitrophenol reduction, antioxidant and antibacterial applications. Journal of Molecular Structure. 2025;1348:143550. doi: https://doi.org/10.1016/j.molstruc.2025.143550
- Bachhar V, Joshi V, Pant P, Duseja M, Chitme H, Sharma N. Green synthesis of TiO2/Ag/Cu2O nanoparticles: Antioxidant potential and in-vitro/in-vivo antidiabetic activity in STZ-induced diabetic albino mice. Inorganic Chemistry Communications. 2026;183:115900. doi: https://doi.org/10.1016/j.inoche.2025.115900
- Bachhar V, Joshi V, Singh A, Mir MA, Bhardwaj A. Antibacterial, antioxidant, and antidiabetic activities of TiO₂ nanoparticles synthesized from Euphorbia hirta. 2025; 14 (1): 1–13.
- Tabasum H, Bhat BA, Sheikh BA, Mehta VN, Rohit JV. Emerging perspectives of plant-derived nanoparticles as effective antimicrobial agents. Inorganic Chemistry Communications. 2022;145:110015. doi: https://doi.org/10.1016/j.inoche.2022.110015
- Kailasa SK, Park TJ, Rohit JV, Koduru JR. Antimicrobial activity of silver nanoparticles. Elsevier; 2019. doi: https://doi.org/10.1016/b978-0-12-816504-1.00009-0
- Mushtaq M, Rohit JV. Enhancing the medicinal potency of Colchicum luteum by forming biogenic silver nanoparticles: Antimicrobial, anticancer and antioxidant evaluation. Microbial Pathogenesis. 2026;210:108181. doi: https://doi.org/10.1016/j.micpath.2025.108181
- Reshi MA, Ara T, Rohit JV. Biogenic metal nanoparticles based visual sensor for the monitoring of environmental pollutants. Microchemical Journal. 2025;219:115839. doi: https://doi.org/10.1016/j.microc.2025.115839
- Eker F, Akdaşçi E, Duman H, Bechelany M, Karav S. Green Synthesis of Silver Nanoparticles Using Plant Extracts: A Comprehensive Review of Physicochemical Properties and Multifunctional Applications. Ijms. 2025;26(13):6222. doi: https://doi.org/10.3390/ijms26136222
- Vanlalveni C, Lallianrawna S, Biswas A, Selvaraj M, Changmai B, Rokhum SL. Green synthesis of silver nanoparticles using plant extracts and their antimicrobial activities: a review of recent literature. RSC Adv. 2021;11(5):2804-2837. doi: https://doi.org/10.1039/d0ra09941d
- Almatroudi A. Silver nanoparticles: synthesis, characterisation and biomedical applications. Open Life Sciences. 2020;15(1):819-839. doi: https://doi.org/10.1515/biol-2020-0094
- Siregar NC, Yanti DD, Ashari A, Aji A. Application of silver nanoparticles synthesised using Moringa leaf extract for detection of mercury ions. Stannum J Sains Terap Kim. 2025; 7 (1): 37–45.
- Esther Arland S, Kumar J. Green and chemical syntheses of silver nanoparticles: Comparative and comprehensive study on characterization, therapeutic potential, and cytotoxicity. European Journal of Medicinal Chemistry Reports. 2024;11:100168. doi: https://doi.org/10.1016/j.ejmcr.2024.100168
- Satria D, Siregar NA, Waruwu SB, Putra EDL, Sinaga SM, Septama AW, et al. Eco-friendly one-pot synthesis of silver nanoparticles via microwave irradiation using Artocarpus camansi extract: Characterization, antioxidant, and antibacterial applications. J King Saud Univ Sci. 2026;38:10112025. doi: https://doi.org/10.25259/jksus_1011_2025
- Furxhi I, Faccani L, Zanoni I, Brigliadori A, Vespignani M, Costa AL. Design rules applied to silver nanoparticles synthesis: A practical example of machine learning application. Computational and Structural Biotechnology Journal. 2024;25:20-33. doi: https://doi.org/10.1016/j.csbj.2024.02.010
- Kumarasamy RV, Natarajan PM, Mathivanan I, Balasubramaniam M, Sn S, Prabhu D, et al. Sustainable biogenic synthesis of silver nanoparticles for oral cancer: a comprehensive review. Front. Nanotechnol. 2026;7. doi: https://doi.org/10.3389/fnano.2025.1648900
- Nalawati AN, Suyatma NE, Wardhana DI. Sintesis Nanopartikel Perak (NPAg) dengan Bioreduktor Ekstrak Biji Jarak Pagar dan Kajian Aktivitas Antibakterinya. jtip. 2021;32(1):98-106. doi: https://doi.org/10.6066/jtip.2021.32.2.98
- Saini P, Mushtaq M, Sofi TA, Anjum S, Bhat BA, Rohit JV. Sustainable management of plant fungus using nanobiopesticide of Delphinium cashmirianum derived bio silver nanoparticles. Sustainable Chemistry One World. 2026;9:100174. doi: https://doi.org/10.1016/j.scowo.2025.100174