RESEARCH ARTICLE
Anti-Inflammatory Properties of Acetone Extract of Green Coffee Beans: An In Vitro Assay
Academic Editor: Sanchaita Rajkhowa
Sciences of Phytochemistry|Vol. 5, Issue 1, pp. 81-87 (2026)
Received
Feb 10, 2026Revised
Mar 13, 2026Accepted
Apr 13, 2026Published
Apr 21, 2026
Abstract
Keywords:
Introduction
Bioactive plants and their extracts possess tremendous therapeutic potential for the treatment of various diseases and for drug discovery, although synthetic drugs currently dominate and are widely used in the pharmaceutical industry (1, 2). Historically, humans have depended on natural products as a primary source of healthcare (3, 4). In developing countries, the active compounds found in plants, which are both affordable and accessible, have shown effectiveness in treating several diseases with minimal or no significant side effects. This has sparked increasing scientific interest in the use of natural products for the development of new drugs (5, 6).
The human body activates a process called inflammation when it is invaded by harmful substances such as bacteria, viruses, pollutants, chemicals, or even damaged cells (7, 8). While this response is essential for protection and healing, excessive or prolonged inflammation can result in tissue damage (9, 10). Tissue and cellular damage occur due to changes in the structure and function of key molecules such as proteins, lipids, deoxyribonucleic acid (DNA), and ribonucleic acid (RNA), as well as cellular membranes, caused by chronic inflammation. Protein denaturation can lead to the formation of abnormal structures that promote inflammatory reactions, while membrane destabilization results in the release of intracellular components that aggravate tissue injury (11, 12).
The stabilization of proteins and biological membranes represents an important strategy in the management of inflammation. In vitro assays such as inhibition of protein denaturation and membrane stabilization using red blood cell models are widely employed as simple, reliable, and cost-effective methods for evaluating potential anti-inflammatory agents (13, 14). These approaches provide insight into the ability of compounds to preserve structural integrity and modulate inflammatory processes under stress conditions.
Non-steroidal anti-inflammatory drugs (NSAIDs) are widely used for the treatment of inflammation. However, irritation of the stomach lining, which can result in gastric ulcers, is one of the major side effects associated with NSAIDs (15, 16). This has increased interest in identifying safer, plant-derived alternatives with comparable therapeutic potential (13).
Coffee is a dicotyledonous plant belonging to the family Rubiaceae. More than 100 species of the coffee plant have been identified (17). However, Coffea arabica, Coffea canephora (commonly called Robusta), and Coffea liberica are the most common species (18).
Studies have shown that green coffee beans (the raw, unroasted seeds of the coffee plant) contain a wide variety of polyphenols, known for their health benefits and their contribution to flavor and aroma. Among these compounds, chlorogenic acid is the most abundant, making up about 12% of the phenolic compounds in green coffee beans. Other polyphenols, such as tannins, lignins, and anthocyanins, are also present but in significantly smaller concentrations (19).
The pharmacological properties of coffee, including its potential antioxidant, anti-inflammatory, antimicrobial, and neuroprotective effects, have been reported in previous studies, supporting its potential as a therapeutic agent (20–22). However, many of these investigations have focused on isolated compounds or advanced molecular and cell-based models rather than crude coffee bean extracts evaluated using simple in vitro assays.
Hence, there is limited information on the anti-inflammatory activity of crude green coffee bean extracts assessed using models such as protein denaturation and membrane stabilization. Furthermore, the use of less-studied coffee varieties, such as Coffe excelsa, and the influence of extraction solvents remain underexplored. Addressing these gaps is important for expanding the understanding of plant-based anti-inflammatory agents using accessible experimental approaches.
Therefore, this study was undertaken to evaluate the anti-inflammatory potential of the acetone extract of green coffee beans by assessing its ability to inhibit protein denaturation and prevent heat-induced hemolysis in vitro. The concentration-dependent effects and the IC₅₀ values of the extract in these assays were determined, and its activity was compared with that of a standard non-steroidal anti-inflammatory drug (acetylsalicylic acid). It is hypothesized that the acetone extract of green coffee beans may exhibit anti-inflammatory activity by preventing protein denaturation and enhancing membrane stability.
Materials and Methods
Sourcing and Identification
Green coffee beans were procured from Bokkos Market, a major agricultural trading center in Bokkos Local Government Area, Plateau State, Nigeria, known for its supply of locally produced agricultural commodities. The botanical identity of the beans was confirmed by Professor I. I. Ibeawuchi of the Department of Crop Science Technology, Federal University of Technology Owerri, using detailed morphological assessment. A voucher specimen of the plant material was deposited in the department herbarium for future reference (Voucher No: FUTO/SAAT/CST/001/2025), ensuring traceability of the study.
Chemicals and Reagents
All chemicals used were of analytical grade. Acetone, hydrogen peroxide, sodium carbonate, trichloroacetic acid, phosphoric acid, sodium hydroxide, and ascorbic acid were obtained from a certified local supplier.
Equipment
The study employed standard laboratory equipment, including a UV–Vis spectrophotometer, water bath, incubator, centrifuge, analytical balance, and standard glassware such as beakers, conical flasks, test tubes, and filter paper.
Plant Extraction
The maceration method was used for extraction. Pulverized green coffee beans (290 g) were soaked in 1200 mL of acetone in a conical flask and left at room temperature for 48 h with occasional stirring. The mixture was then filtered using Whatman No. 1 filter paper, and the filtrate was concentrated in a water bath at 40–45 °C to remove the solvent, yielding 29.8 g of semi-solid extract. The % yield of the extract was calculated as 10.3% based on the dry weight of the plant material. The resulting extract was stored in an airtight container at 4 °C until use.
Preparation of Extract Solution (Stock Solution)
A stock solution of the acetone extract was prepared by dissolving 0.5 g of the crude extract in 10 mL dimethyl sulfoxide (DMSO), yielding a concentration of 50 mg/mL. For each assay, an intermediate solution (5 mg/mL) was prepared by diluting 1 mL of the stock solution with 9 mL of distilled water. Working concentrations of 100, 200, 300, 500, and 1000 µg/mL were then prepared from the intermediate solution immediately before use. The final concentration of DMSO in all assay mixtures did not exceed 1%, ensuring that the solvent did not interfere with protein denaturation or red blood cell membrane stability.
Ethical Considerations
The study utilized anonymized discarded human blood samples obtained from Federal Medical Center, Owerri, and complied with institutional guidelines for minimal-risk in vitro studies. No identifiable donor information was used.
Inhibition of Protein Denaturation
The ability of the extract to inhibit protein denaturation was evaluated using a modified procedure described by Mizushima and Kobayashi (23) and Sakat et al. (24). Briefly, 500 μL of 1% bovine serum albumin (BSA) solution was mixed with 100 μL of the extract at various concentrations. The mixture was incubated at room temperature for 10 min, followed by heating at 51 °C for 20 min to induce protein denaturation. After cooling, absorbance was measured at 660 nm. Acetylsalicylic acid was used as positive control. The % inhibition of protein denaturation was calculated using Eq. 1.
Where AcAc is the absorbance of the control, and AsAs is the absorbance of the sample, IC₅₀ values were determined by nonlinear regression analysis using GraphPad Prism.
Stabilization of Membrane
Preparation of Red Blood Cell (RBC)
Anticoagulated human blood was centrifuged at 3000 rpm for 10 min to separate its components. Following three saline washes of the resultant sample, the red blood cell fraction was separated. The RBC fraction was then diluted with 1x phosphate-buffered saline (PBS) to obtain a final concentration of 10% (v/v), following the method of Sadique et al. (25) and Sakat et al. (24).
Heat-Induced Hemolysis
A 100 μL volume of 10% red blood cell (RBC) suspension was mixed with 100 μL of the plant extract. The mixture was then incubated at 56 °C for 30 min to induce hemolysis, followed by centrifugation at 2500 rpm for 10 min at room temperature. The supernatant was collected, and its absorbance was measured at 560 nm to determine the extent of hemolysis. Acetylsalicylic acid (aspirin) was used as a positive control. The % of membrane stabilization was calculated according to the method described by Shinde et al. (26) and Sakat et al. (24) using Eq. 1. This assay effectively evaluates the potential of the extract to provide protection against thermal-induced protein denaturation and cellular damage.
Statistical Analysis
Data were expressed as mean ± standard deviation (SD) from three independent biological replicates, each measured in triplicate. Statistical comparisons were performed using one-way analysis of variance (ANOVA) followed by Dunnett’s post hoc test. Differences were considered statistically significant at p < 0.0001. All analyses were carried out using GraphPad Prism version 11.0. Concentration values were log₁₀-transformed prior to statistical analysis and graphical representation to improve the linearity of the dose–response relationship.
Results and Discussion
Inhibition of Protein Denaturation
Protein denaturation involves the disruption of secondary, tertiary, and quaternary structures, leading to loss of biological function and contributing to inflammatory processes (14, 27). In this study, the acetone extract of green coffee beans (C. excelsa) exhibited inhibitory activity against protein denaturation across all tested concentrations (Table 1).
| Concentrations (µg/mL) | Log₁₀ (Concentration) | Inhibition of protein denaturation (%) | Inhibition of heat-induced hemolysis (%) |
|---|---|---|---|
| 100 | 2.00 | 13.10 ± 1.85 | 21.16 ± 1.21 |
| 200 | 2.30 | 28.00 ± 1.75 | 29.11 ± 1.22 |
| 300 | 2.48 | 51.19 ± 0.89 | 35.05 ± 1.89 |
| 500 | 2.70 | 63.74 ± 4.99 | 50.47 ± 0.75 |
| 1000 | 3.00 | 70.45 ± 0.84 | 62.06 ± 1.97 |
| Standard | — | 75.79 ± 1.51 | 67.39 ± 2.51 |
| Note: Values are expressed as mean ± standard deviation (n = 3). Statistical significance was set at p < 0.0001. Log₁₀ values were calculated from concentration (µg/mL) and used for statistical analysis and graphical representation. | |||
These findings align with the objective of evaluating the anti-inflammatory potential of the extract using in vitro models.
The extract exhibited an overall increase in inhibitory activity with increasing concentration, although minor variations were observed at intermediate concentrations, with % inhibition ranging from 13.10% at 100 µg/mL to 70.45% at 1000 µg/mL. Nonlinear regression analysis yielded an IC₅₀ value of 249.8 µg/mL, indicating moderate anti-inflammatory potential.
This trend is further illustrated in Figure 1, confirming the nonlinear dose–response pattern observed in the inhibition of protein denaturation. The curve shows a gradual increase in inhibitory activity with concentration, followed by a plateau at higher concentrations, suggesting possible saturation of protein-binding interactions.
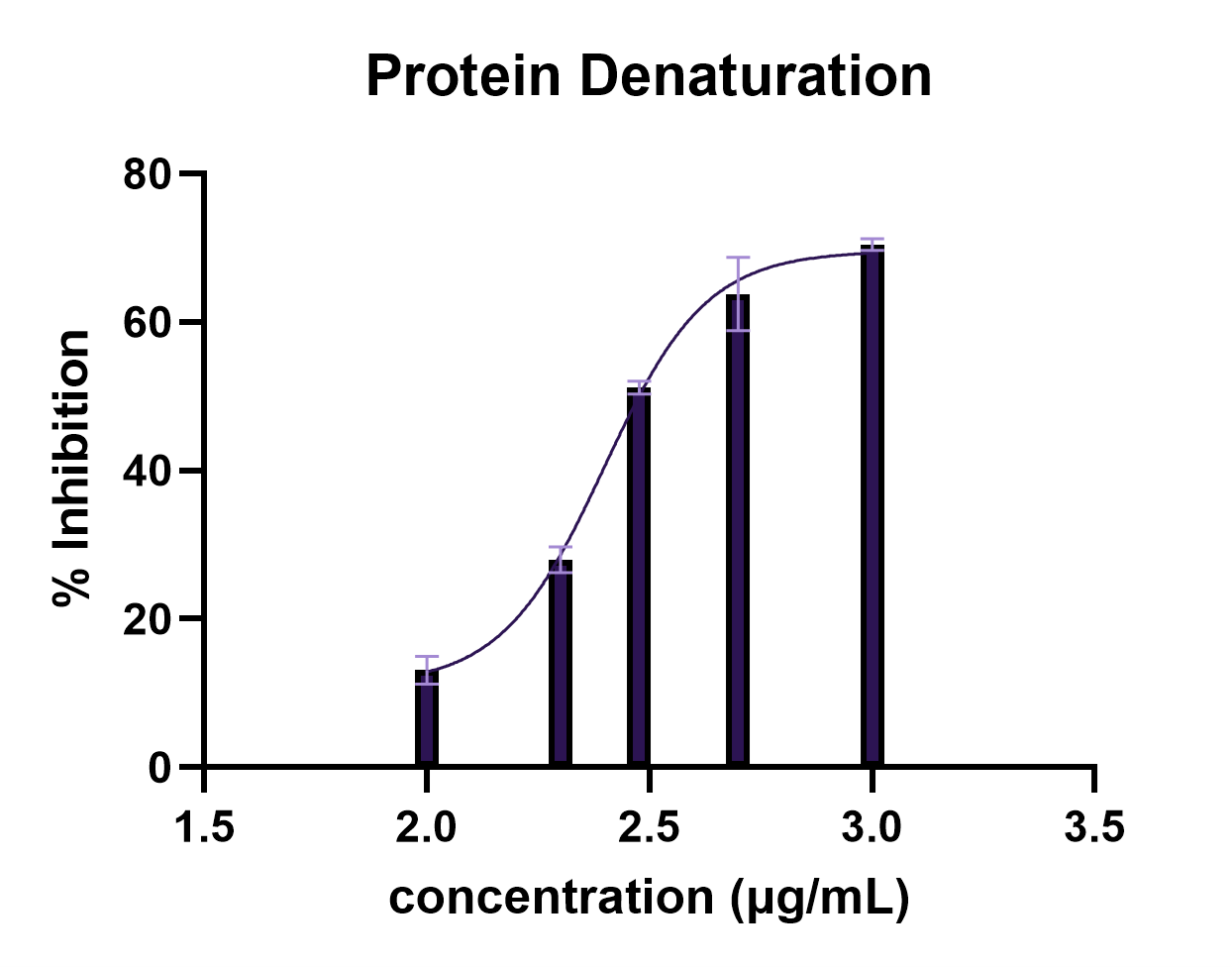
Statistical analysis using one-way ANOVA revealed a significant difference among treatment groups (F = 528.3, p < 0.0001). Dunnett’s post hoc test showed that all concentrations produced significant inhibition compared to the control (p < 0.0001).
The observed increase in inhibitory activity with concentration, together with slight deviations at the intermediate concentrations, suggests a nonlinear dose–response relationship, which may reflect complex interactions between extract constituents and protein molecules.
Compared with previously reported plant extracts, the IC₅₀ value obtained in this study suggests moderate activity, as some plant-derived extracts exhibit lower IC₅₀ values and thus higher potency. For instance, an anti-inflammatory study using Solanum obtusifolium Dunal fruit reported an IC₅₀ value of 197.31 ± 6.89 µg/mL (28), indicating slightly higher inhibitory activity than the present extract. In contrast, extracts of Ephedra alata Decne used in a protein denaturation study exhibited higher IC₅₀ values ranging from 309.45 to 504 µg/mL based on the extraction solvents used (29), suggesting lower activity compared to the current findings. Similarly, extracts of Ajuga integrifolia Buch. -Ham. leaves used in a protein denaturation study reported an IC₅₀ value of 532 µg/mL (30). These comparisons place the activity of the present extract within the moderate range relative to other plant-derived anti-inflammatory agents.
Furthermore, the extract showed lower inhibitory activity than the standard drug acetylsalicylic acid (aspirin), indicating comparatively reduced efficacy. At 1000 µg/mL, the extract showed 70.45% inhibition compared with 75.79% for acetylsalicylic acid, indicating lower but comparable activity.
The observed activity may be attributed to bioactive compounds such as phenolics and flavonoids, which are known to possess anti-inflammatory properties. However, no phytochemical analysis was conducted in this study; therefore, this attribution remains speculative.
Heat-Induced Hemolysis
Membrane stabilization is an important mechanism in the management of inflammation, as destabilization of lysosomal membranes leads to the release of enzymes that exacerbate tissue damage (31, 32). In this study, the acetone extract of green coffee beans demonstrated protective effects against heat-induced hemolysis of erythrocytes (Table 1).
The extract exhibited an overall increase in membrane stabilization across the tested range, with % inhibition ranging from 21.16% at 100 µg/mL to 62.06% at 1000 µg/mL. The IC₅₀ value was determined to be 391.6 µg/mL, indicating moderate membrane-stabilizing activity.
This trend is further illustrated in Figure 2, confirming the nonlinear dose–response pattern observed in membrane stabilization. The graphical profile indicates progressive membrane stabilization with increasing concentration, with slight deviations at intermediate concentrations, reflecting complex interactions between extract constituents and erythrocyte membranes.
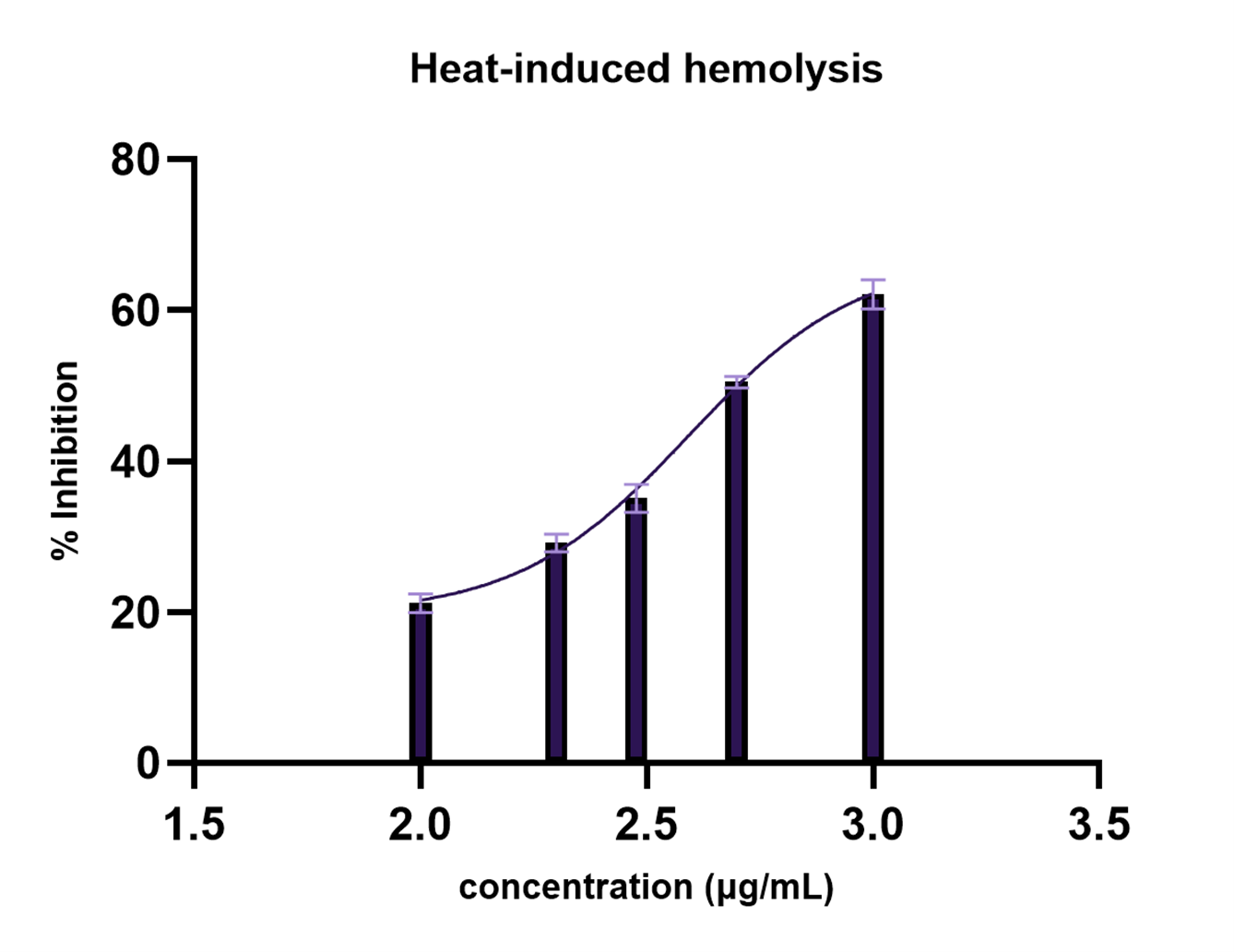
Statistical analysis using one-way ANOVA showed a highly significant effect of treatment (F = 692.2, p < 0.0001; R² = 0.9966). Dunnett’s multiple comparisons test revealed that all concentrations significantly reduced hemolysis compared to the control (p < 0.0001).
In comparison with acetylsalicylic acid (aspirin), the extract exhibited lower inhibitory activity, indicating reduced potency relative to conventional anti-inflammatory agents. At 1000 µg/mL, the extract showed 62.06% inhibition compared with 67.39% for acetylsalicylic acid, indicating lower activity relative to the standard. However, the IC₅₀ value obtained in this study is comparable to those reported for some plant extracts in similar assays. For instance, Solanum obtusifolium Dunal fruit used in a hemolysis study reported a lower IC₅₀ value of 196.81 ± 4.59 µg/mL (28), indicating higher potency than the present extract, whereas Ficus religiosa seeds extract used in a hemolysis study reported substantially higher IC₅₀ values ranging from 790.30 to 1094.65 µg/mL depending on the solvent fraction (33), suggesting lower activity compared to the current findings. These comparisons further support the classification of the acetone extract of C. excelsa as having moderate membrane-stabilizing activity.
While erythrocyte membranes share structural similarities with lysosomal membranes, extrapolation of these findings to in vivo lysosomal stabilization remains theoretical. Therefore, further in vivo studies are required to confirm this mechanism.
Conclusion
This study demonstrated that the acetone extract of green coffee beans (C. excelsa) exhibited measurable anti-inflammatory activity in vitro, as evidenced by its ability to inhibit protein denaturation and stabilize erythrocyte membranes under heat-induced conditions. Although the extract was less potent than acetylsalicylic acid (standard drug), the observed concentration-dependent effects suggest the presence of bioactive compounds that may contribute to its activity, possibly through mechanisms related to the modulation of inflammatory mediators and protection of cellular structures under stress conditions. These findings indicate that crude extracts, despite their lower potency compared to standard drugs, may still offer valuable pharmacological potential as alternative or complementary therapeutic agents.
However, the absence of phytochemical characterization and in vivo studies limits the ability to attribute the activity to specific compounds or predict therapeutic relevance. In addition, pharmacokinetic properties and toxicity profiles of the extract remain unknown, which are critical factors for its potential clinical application. Further research is needed to identify the active constituents, explore their mechanism of action, and evaluate their efficacy and safety in animal models to better understand the potential of green coffee beans as an anti-inflammatory agent, as well as to support the development of standardized formulations and optimized extraction methods for future therapeutic use.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
All data generated or analyzed during this study are included in this published article.
Ethics Statement
The study utilized anonymized discarded human blood samples obtained from Federal Medical Center, Owerri, and complied with institutional guidelines for minimal-risk in vitro studies. No identifiable donor information was used.
Funding Information
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
References
- Thomas E, Stewart LE, Darley BA, Pham AM, Esteban I, Panda SS. Plant-Based Natural Products and Extracts: Potential Source to Develop New Antiviral Drug Candidates. Molecules. 2021;26(20):6197. doi: https://doi.org/10.3390/molecules26206197
- Chaachouay N, Zidane L. Correction: Chaachouay, N.; Zidane, L. Plant-Derived Natural Products: A Source for Drug Discovery and Development. Drugs Drug Candidates 2024, 3, 184–207. Ddc. 2026;5(1):14. doi: https://doi.org/10.3390/ddc5010014
- Rajčević N, Bukvički D, Dodoš T, Marin PD. Interactions between Natural Products—A Review. Metabolites. 2022;12(12):1256. doi: https://doi.org/10.3390/metabo12121256
- Singh S, Rai V, Kanojia A, Yadav A. Natural Products and Traditional Medicine: Investigating the Role of Natural Products and Traditional Medicine in Modern Pharmacology. J. Res. Appl. Sci. Biotechnol. 2024;3(4):40-53. doi: https://doi.org/10.55544/jrasb.3.4.6
- Ouma JJ, Mungai N, Gathirwa JW, Orwa JA. A Study of the Antimalarial Activity of Combined Plant Extracts Traditionally Used in Kenya. Jocamr. 2022:13-21. doi: https://doi.org/10.9734/jocamr/2022/v17i130322
- Ssenku JE, Okurut SA, Namuli A, Kudamba A, Tugume P, Matovu P, et al. Medicinal plant use, conservation, and the associated traditional knowledge in rural communities in Eastern Uganda. Trop Med Health. 2022;50(1). doi: https://doi.org/10.1186/s41182-022-00428-1
- Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2017;9(6):7204-7218. doi: https://doi.org/10.18632/oncotarget.23208
- Abdulkhaleq LA, Assi MA, Abdullah R, Zamri-Saad M, Taufiq-Yap YH, Hezmee MNM. The crucial roles of inflammatory mediators in inflammation: A review. Vet World. 2018;11(5):627-635. doi: https://doi.org/10.14202/vetworld.2018.627-635
- Raziyeva K, Kim Y, Zharkinbekov Z, Kassymbek K, Jimi S, Saparov A. Immunology of Acute and Chronic Wound Healing. Biomolecules. 2021;11(5):700. doi: https://doi.org/10.3390/biom11050700
- Soliman AM, Barreda DR. Acute Inflammation in Tissue Healing. Ijms. 2022;24(1):641. doi: https://doi.org/10.3390/ijms24010641
- Chaiya P, Senarat S, Phaechamud T, Narakornwit W. In vitro anti-inflammatory activity using thermally inhibiting protein denaturation of egg albumin and antimicrobial activities of some organic solvents. Materials Today: Proceedings. 2022;65:2290-2295. doi: https://doi.org/10.1016/j.matpr.2022.04.916
- Yesmin S, Paul A, Naz T, Rahman ABMA, Akhter SF, Wahed MII, et al. Membrane stabilization as a mechanism of the anti-inflammatory activity of ethanolic root extract of Choi (Piper chaba). Clin Phytosci. 2020;6(1). doi: https://doi.org/10.1186/s40816-020-00207-7
- Aidoo DB, Konja D, Henneh IT, Ekor M. Protective Effect of Bergapten against Human Erythrocyte Hemolysis and Protein Denaturation In Vitro. International Journal of Inflammation. 2021;2021:1-7. doi: https://doi.org/10.1155/2021/1279359
- Hasan MM, Islam ME, Hossain MS, Akter M, Rahman MAA, Kazi M, et al. Unveiling the therapeutic potential: Evaluation of anti-inflammatory and antineoplastic activity of Magnolia champaca Linn’s stem bark isolate through molecular docking insights. Heliyon. 2024;10(1):e22972. doi: https://doi.org/10.1016/j.heliyon.2023.e22972
- Alhamadi N, Asiri AH, Alshahrani FM, Alqahtani AY, Al Qout MM, Alnami RA, et al. Gastrointestinal Complications Associated With Non-steroidal Anti-inflammatory Drug Use Among Adults: A Retrospective, Single-Center Study. Cureus. 2022. doi: https://doi.org/10.7759/cureus.26154
- Sohail R, Mathew M, Patel KK, Reddy SA, Haider Z, Naria M, et al. Effects of Non-steroidal Anti-inflammatory Drugs (NSAIDs) and Gastroprotective NSAIDs on the Gastrointestinal Tract: A Narrative Review. Cureus. 2023. doi: https://doi.org/10.7759/cureus.37080
- Lee IC, Lee JS, Lee JH, Kim Y, So WY. Anti-Oxidative and Anti-Inflammatory Activity of Kenya Grade AA Green Coffee Bean Extracts. ijph. 2020. doi: https://doi.org/10.18502/ijph.v48i11.3521
- Şemen S, Mercan S, Yayla M, Açıkkol M. Elemental composition of green coffee and its contribution to dietary intake. Food Chemistry. 2017;215:92-100. doi: https://doi.org/10.1016/j.foodchem.2016.07.176
- Hall RD, Trevisan F, de Vos RC. Coffee berry and green bean chemistry – Opportunities for improving cup quality and crop circularity. Food Research International. 2022;151:110825. doi: https://doi.org/10.1016/j.foodres.2021.110825
- Revuelta-Iniesta R, Al-Dujaili EAS. Consumption of Green Coffee Reduces Blood Pressure and Body Composition by Influencing 11β-HSD1 Enzyme Activity in Healthy Individuals: A Pilot Crossover Study Using Green and Black Coffee. BioMed Research International. 2014;2014:1-9. doi: https://doi.org/10.1155/2014/482704
- Bharath N, Sowmya N, Mehta D. Determination of antibacterial activity of green coffee bean extract on periodontogenic bacteria like Porphyromonas gingivalis, Prevotella intermedia, Fusobacterium nucleatum and Aggregatibacter actinomycetemcomitans: An in vitrostudy. Contemp Clin Dent. 2015;6(2):166. doi: https://doi.org/10.4103/0976-237x.156036
- Shah S, Kumar A. Coffee: Constituents and Health Benefits. Bji. 2020;:22-38. doi: https://doi.org/10.9734/bji/2020/v24i530115
- Mizushima Y, Kobayashi M. Interaction of anti-inflammatory drugs with serum proteins, especially with some biologically active proteins. Journal of Pharmacy and Pharmacology. 1968;20(3):169-173. doi: https://doi.org/10.1111/j.2042-7158.1968.tb09718.x
- Juvekar A, Sakat S, Wankhede S, Juvekar M, Gambhire M. Evaluation of antioxidant and anti-inflammatory activity of methanol extract of Oxalis corniculata. Planta Med. 2009;75(09). doi: https://doi.org/10.1055/s-0029-1234983
- Pavela R. Insecticidal activity of certain medicinal plants. Fitoterapia. 2004;75(7-8):745-749. doi: https://doi.org/10.1016/j.fitote.2004.08.005
- Shinde U, Phadke A, Nair A, Mungantiwar A, Dikshit V, Saraf M. Membrane stabilizing activity — a possible mechanism of action for the anti-inflammatory activity of Cedrus deodara wood oil. Fitoterapia. 1999;70(3):251-257. doi: https://doi.org/10.1016/s0367-326x(99)00030-1
- Masson P, Lushchekina S. Conformational Stability and Denaturation Processes of Proteins Investigated by Electrophoresis under Extreme Conditions. Molecules. 2022;27(20):6861. doi: https://doi.org/10.3390/molecules27206861
- Abdnim R, Bouslamti M, El-Mernissi R, Farihi A, Chelouati T, Mahamat OB, et al. In vitro and in silico antiglycation antihyperglycemic and anti-inflammatory properties of chemically profiled fruit extracts of Solanum obtusifolium Dunal. Sci Rep. 2026;16(1). doi: https://doi.org/10.1038/s41598-026-39203-1
- Al Jaafreh AM. Investigation of the phytochemical profiling and antioxidant, anti-diabetic, anti-inflammatory, and MDA-MB-231 cell line antiproliferative potentials of extracts from Ephedra alata Decne. Sci Rep. 2024;14(1). doi: https://doi.org/10.1038/s41598-024-65561-9
- Singh H, Kumar S, Arya A. Evaluation of antibacterial, antioxidant, and anti-inflammatory properties of GC/MS analysis of extracts of Ajuga. integrifolia Buch.-Ham. leaves. Sci Rep. 2024;14(1). doi: https://doi.org/10.1038/s41598-024-67133-3
- Simonaro CM. Lysosomes, Lysosomal Storage Diseases, and Inflammation. Journal of Inborn Errors of Metabolism and Screening. 2016;4:232640981665046. doi: https://doi.org/10.1177/2326409816650465
- Bonam SR, Wang F, Muller S. Lysosomes as a therapeutic target. Nat Rev Drug Discov. 2019;18(12):923-948. doi: https://doi.org/10.1038/s41573-019-0036-1
- Pinipay F, Rokkam R, Botcha S, Tamanam RR. Metabolomic and network Pharmacology insights into the anti-inflammatory and anti-urolithiatic potential of Ficus religiosa seeds. Sci Rep. 2025;15(1). doi: https://doi.org/10.1038/s41598-025-26906-0