RESEARCH ARTICLE
Phytochemical Extraction from Mediterranean Plants: A Comparative Study of Traditional Techniques
Academic Editor: Khafit Wiradimafan
Sciences of Phytochemistry|Vol. 5, Issue 1, pp. 160-171 (2026)
Received
Feb 10, 2026Revised
Mar 30, 2026Accepted
May 5, 2026Published
May 14, 2026
Abstract
Introduction
Medicinal and aromatic plants constitute a rich source of diverse phytochemicals, primarily phenolics, alkaloids, and terpenes, with well-documented antioxidant, antimicrobial, and therapeutic properties, making them suitable alternatives to synthetic compounds in several areas, including pharmaceutical and cosmetic applications (1, 2). The method used for extracting these compounds, including the process, conditions, and solvents, is crucial to optimize the yield, efficacy, and selectivity of the target compounds. In recent years, several studies have used advanced methods to extract these bioactive compounds, such as microwave and ultrasound extraction. However, traditional techniques such as maceration and Soxhlet remain widely used due to their robustness, reproducibility, and good preservation of phytochemicals (3). Although advanced extraction methods such as ultrasound-, microwave-, and pressurized-fluid-assisted extraction may improve efficiency, conventional techniques remain relevant as benchmark approaches because they are widely used, operationally simple, and suitable for controlled comparison of solvent effects across different plant matrices. Solvent systems have also been extensively investigated, including a new generation of solvents such as natural deep eutectic solvents, ionic liquids, and pressurized fluids. However, classical solvents including water, methanol and ethanol, remain predominant due to their proven efficiency, safety, and regulatory acceptance. Solvent polarity is a key parameter governing extraction efficiency and selectivity (4). Among the diversity of botanical sources, phytochemicals found in olive leaves (Olea europaea), rosemary (Salvia rosmarinus), bay laurel (Laurus nobilis), and herb Robert (Geranium robertianum), show great potential in several areas, such as cosmetic, nutraceutical, and pharmaceutical applications. Importantly, these phytochemical classes differ markedly in polarity, molecular weight, and thermal stability, which directly govern their extractability under different solvent systems and extraction regimes.
Olive leaves represent one of the most abundant by-products of the olive oil industry, generating in Europe ca. 20 million tons of residues annually (5, 6). They are particularly rich in phenolics such as oleuropein (ca. 14%), hydroxytyrosol, tyrosol, luteolin, apigenin, and rutin (7). The phenolic profile depends strongly on cultivar, geographic origin, harvest time, and processing parameters (6, 8). These metabolites exhibit antioxidant, antimicrobial, and anti-aging properties, and have been increasingly used in cosmetic formulations as dual-function ingredients that both protect against oxidative stress and stabilize active compounds (5, 9).
Various methods of extraction have been applied to olive leaves, such as maceration, Soxhlet, ultrasound, microwave, and pressurized-liquid extraction (8). Reported studies typically express efficiency in terms of total phenolic content (mg gallic acid equivalents, GAE) or antioxidant capacity (trolox equivalent antioxidant activity (TEAC), 2, 2-diphenyl-1-picrylhydrazyl (DPPH), 2, 2′-azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS)) (5, 10–12), but rarely provide extraction yield data. For instance, Mylonaki et al. obtained up to 250 mg GAE/g extract using a water/ethanol/citric acid mixture (11), while Guebebia et al. reported 8.43 mg GAE/g dry biomass for Soxhlet extraction of wild oleasters (13). Similar trends were observed in deep eutectic and hydrogen-rich water systems showing strong antioxidant activity (up to 26.26 mg TEAC/g) but lacking yield information (5, 12). Ramirez et al. concluded that the drying temperature of olive leaves affects the phenolic content. At temperatures above 60 °C, phenolic compounds can be degraded or oxidized (14). Overall, these studies indicate that olive leaf extracts are dominated by moderately polar phenolics whose recovery is strongly dependent on solvent polarity and extraction temperature, yet comparative data on extraction yield across classical techniques remain scarce.
G. robertianum L. (herb Robert) is a European species known for its strong antioxidant, antimicrobial, and anti-inflammatory potential (15). Methanolic extracts are particularly rich in phenolic compounds (up to 284 mg GAE/g extract), including quercetin, kaempferol, ellagic acid, and gallic acid, and display remarkable antioxidant activity, together with antimicrobial effects against Staphylococcus aureus and Bacillus cereus (16). Neagu et al. reported that ultrafiltered hydroalcoholic extracts also exhibited a strong antioxidant capacity (17) and the ability to inhibit the growth of cancer cells (18). More recently, Świątek et al. also demonstrated anticancer activity using extracts obtained with hexane and ethyl acetate. Additionally, the antibacterial activities, mainly against g-positive bacteria together with moderate antifungal activity were described (19). In addition, G. robertianum leaves are rich in flavonoids, tannins, and B-vitamins (riboflavin, niacin, thiamine), highlighting their nutritional and multifunctional potential (20). Despite the extensive characterization of herb Robert extracts obtained with organic solvents, direct comparisons between extraction techniques and solvent polarity in terms of extraction yield and chemical profiles are still limited.
S. rosmarinus Spenn. (rosemary) is another Mediterranean species with pharmacological and cosmetic relevance due to its antioxidant, antimicrobial, anti-inflammatory, and photoprotective properties. This plant is rich in diterpenes (carnosic acid, carnosol, rosmanol), phenolic acids, and flavonoids (21–23). Zeroual et al. extracted the phytochemicals using methanol and ethanol, resulting in extracts with high antioxidant and antimicrobial activity against S. aureus and C. albicans (22). Damianova et al. also found that ethanolic extracts are rich in tannins and flavonoids and display strong activity against Aspergillus niger and gram-positive bacteria (24), and Aamer et al. described that the methanolic extract has antifungal properties against Rhizoctonia solani, Fusarium oxysporum, with performance comparable to commercial fungicides (25). Using acetone, it is also possible to extract phenolic compounds, as described by Mena et al., who reported the extraction of 57 phenolic compounds and 63 volatiles (23). These findings highlight rosemary as a chemically complex matrix rich in lipophilic and semi-polar diterpenes, making it a relevant model to assess solvent- and technique-dependent extraction selectivity.
L. nobilis L. (bay laurel) is a Mediterranean species widely used in culinary applications. This plant can be considered an agricultural residue, since less than 1% of the harvested biomass is effectively utilized (26). The leaves of bay laurel contain diverse polyphenols, including flavonoids (kaempferol, quercetin, isorhamnetin derivatives), phenolic acids (p-coumaric, ferulic), tannins, and lignans, responsible for antioxidant, antimicrobial, and neuroprotective activities. Reported total phenolic contents vary markedly (53–9200 mg GAE/100 g extract) depending on solvent composition, temperature, and pre-treatment. Hydroalcoholic solvents and moderate temperatures (≤80 °C) enhance recovery, while microwave- and ultrasound-assisted extraction offer greener and faster alternatives (27). Comparative extractions by heat-reflux, microwave, and ultrasound methods yielded 32–54 mg GAE/g, with microwave extraction providing the highest recovery in a shorter time, while solvent polarity and temperature remain key factors influencing phenolic yield and bioactivity (28). Pressurized liquid extraction and mechanochemical-assisted extraction have recently emerged as sustainable methods, recovering up to 49–75 mg GAE/g within min while preserving phenolic integrity (26, 29). The wide variability reported for bay laurel phenolic content suggests a strong influence of extraction conditions, underscoring the need for systematic comparative studies using conventional techniques and solvents.
Although the phytochemical composition and bioactivity of O. europaea, G. robertianum, S. rosmarinus, and L. nobilis have been widely reported, the available literature remains fragmented. Most studies focus on total phenolic content, antioxidant activity, or single-species extractions, often under markedly different extraction conditions, which hinders direct comparison across solvents, extraction regimes, and plant matrices. In particular, comparative data integrating extraction yield, solvent polarity, conventional extraction technique, and GC–MS-accessible chemical profiles across multiple Mediterranean plant species remain limited. Despite the increasing use of advanced extraction technologies, direct cross-study comparison is frequently complicated by the simultaneous variation of equipment-specific parameters, energy input, extraction kinetics, and analytical endpoints. In the present work, conventional techniques were selected not because they are inherently superior to assisted methods, but because they provide operationally distinct and chemically interpretable benchmark extraction conditions: infusion represents a water-based extraction regime, maceration probes room-temperature solid–liquid partitioning with minimal thermal input, and Soxhlet provides continuous hot-solvent renewal under sustained extraction. This simplified experimental framework allows the influence of solvent polarity and extraction regime to be compared across different plant matrices under controlled and operationally comparable laboratory conditions.
Accordingly, the present study was guided by the hypothesis that extraction yield and GC–MS-accessible comparative fingerprints would vary systematically with both solvent polarity and extraction technique across the four selected plant species. More specifically, it was expected that more polar solvents would generally recover a greater mass of extractable material, whereas heated continuous extraction would modify the relative representation of GC–MS-accessible semi-volatile constituents when compared with room-temperature maceration. To address this hypothesis within an exploratory screening design, the present work comparatively evaluated infusion, maceration, and Soxhlet extraction using solvents of different polarity, with assessment based on extraction yield, qualitative phytochemical screening, and GC–MS-accessible chemical profiles.
Experimental Section
Materials
Methanol (99.85%), ethanol (98%), hexane (98%), sodium hydroxide (>99%) were purchased from José Manuel Gomes dos Santos, Lda). Chloroform (>99%), dichloromethane (> 99%), ethyl acetate (>99%), and BSTFA were obtained from Sigma-Aldrich. Sulfuric acid (96%) was acquired from Panreac, hydrochloric acid (37%) from Fisher, ferric chloride (77%) from Fluka, ammonia solution (24%) from Vencilab, Lda and Dragendorff’s reagent from VWR Chemicals. All reagents were used as received. Leaves of olive (O. europaea), bay laurel (L. nobilis), rosemary (S. rosmarinus), and herb Robert (G. robertianum) were collected in the Coimbra region (Portugal) and dried in a ventilated oven at 50 °C until constant weight. The dried plant material was used as collected after oven-drying to constant weight and was not further standardized by particle-size fractionation. Accordingly, the plant material was not milled, sieved, or classified into defined particle-size fractions prior to extraction.
The extractions were therefore performed on heterogeneous dried leaf material, and differences in fragment size and sample geometry among species may have affected solvent accessibility, diffusion path length, and mass-transfer efficiency. For this reason, the extraction-yield data are interpreted as descriptive comparative indicators within a screening framework rather than as strictly normalized extraction efficiencies.
Methods
Phytochemical Extraction
For the infusion method, 0.5 g of plant material was added to 20 mL of water. The mixture was kept under reflux at 100 °C, during 1, 2, and 5 h.
For the maceration procedure, 0.5 g of plant material was mixed with 20 mL of solvent (methanol, ethanol, ethyl acetate, dichloromethane or hexane) under continuous stirring at 200 rpm using a magnetic stir bar. The flask was wrapped in aluminum foil to prevent light exposure and maintained at room temperature. Extraction was carried out for 1, 3, and 5 days.
For the Soxhlet extraction, 0.5 g of plant material was placed in a filter paper thimble and positioned in the extraction chamber. A total of 150 mL of solvent (water, methanol, ethanol, ethyl acetate, dichloromethane or hexane) was used. For Soxhlet extraction, the process was stopped when the solvent returning to the siphon chamber became visually colorless under constant observation. This endpoint was used as an operational criterion for screening purposes and should be regarded as approximate rather than as a rigorously standardized exhaustion criterion.
All extractions were performed using plant material previously dried at 50 °C to constant weight; therefore, extraction yields are reported relative to oven-dried biomass. This procedure was adopted to minimize moisture-related variability among samples before extraction. However, no additional correction based on an independent residual-moisture determination was applied after oven drying. Therefore, the reported extraction yields should be interpreted as recovered extract mass relative to oven-dried plant material, rather than as values normalized for residual water content with analytical moisture correction. No further normalization of extraction efficiency, beyond recovered extract mass relative to oven-dried plant material, was applied, as the study was designed as an exploratory comparative screening exercise.
These extractions were conducted as an exploratory comparative screening study. Full replication of all extraction conditions was not performed; therefore, extraction yield values are presented as descriptive comparative indicators and should not be interpreted as statistically validated estimates. Selected maceration experiments for olive leaf extracts (ethanol and methanol) were independently repeated and showed comparable extraction yields, providing limited internal support for method consistency.
Qualitative Phytochemical Screening
Chemical tests were done based on standard procedures (30–32):
Flavonoids: A qualitative screening for flavonoids was performed by adding 1 mL of distilled water and 0.5 mL of 10% aqueous ammonia solution to 0.1 g of extract. The appearance of a yellow coloration was taken as an indicator of flavonoid presence.
Phenolic compounds: For the detection of phenolic constituents, 0.1 g of extract was treated with 1 mL of 10% aqueous sodium hydroxide and allowed to stand until a yellow coloration was obtained. The decolorization of the solution after adding 0.5 mL of 5% aqueous hydrochloric acid confirmed the presence of phenolic compounds.
Tannins: To evaluate tannins, 0.1 g of extract was mixed with 3 mL of distilled water followed by 0.1 g of ferric chloride. The formation of a blue or green coloration was considered a positive diagnostic test for tannins.
Alkaloids: For alkaloid determination, 0.1 g of extract was reacted with 0.5 mL of Dragendorff’s reagent. The apparent red to orange coloration was taken as evidence for the presence of alkaloids.
Terpenoids: To detect terpenoids, 0.1 g of extract was combined with 1 mL of chloroform, followed by the careful addition of 1.5 mL of concentrated sulfuric acid. The appearance of a red to brown coloration was considered a positive reaction for terpenoids.
GC-MS
Extracts (5 mg) were derivatized with 250 μL BSTFA, dried under nitrogen, and reconstituted in 250 μL dichloromethane for GC–MS injection. Analyses were carried out on an Agilent 7820A GC coupled to an Agilent MSD 5975 with an HP-5MS column (30 m × 250 μm × 0.25 μm), using helium (1 mL/min) as carrier gas. The oven was programmed as follows: 80 °C (3 min), ramp to 250 °C at 10 °C/min (10 min hold), then to 280 °C at 5 °C/min (5 min hold). Injector, interface, quadrupole, and ion source temperatures were 250, 280, 150, and 250 °C, respectively. Compounds were identified as silylated derivatives by comparison with in-house, Wiley 10th, and NIST spectral libraries of the corresponding silylated derivatives. No external calibration standards were used for compound quantification or formal confirmation of identity. Accordingly, GC–MS was used here as a comparative fingerprinting tool rather than as a validated quantitative analytical method. Compound assignments were based on mass spectral library matching of the derivatized peaks, and no external authentic standards or calibration-based confirmation procedures were employed. Therefore, the reported identities should be regarded as tentative, and the chromatographic data should be interpreted qualitatively for comparative purposes only.
Results and Discussion
Infusion
Extraction Yield
A descriptive comparison of aqueous infusion at 100 °C for 1, 2, and 5 h showed marked differences among the four plant species, Figure 1. For aqueous extractions, the reported yield may also reflect the co-extraction of inorganic species and the formation of hydrolysis products; therefore, yield values should be interpreted as a global mass recovery indicator rather than a direct measure of phytochemical content. Olive leaves and rosemary exhibited the highest yields overall, ranging from approximately 26% to 34%, whereas bay laurel and herb Robert produced lower values, between 2% and 9%. For herb Robert, extraction yield increased from 9% after 1 h to nearly 20% after 5 h, suggesting slower recovery of water-extractable material under the tested conditions. Conversely, olive and rosemary leaves reached near-maximum yields within the first h, with only minor fluctuations at longer times. Bay laurel showed a modest but consistent increase across time, achieving its maximum yield after 5 h. A similar extraction yield (18%) was reported by El Faqer et al. for Moroccan L. nobilis L. , using an infusion procedure in which the plant material was immersed in water preheated at 70 °C and subsequently allowed to cool gradually (33).
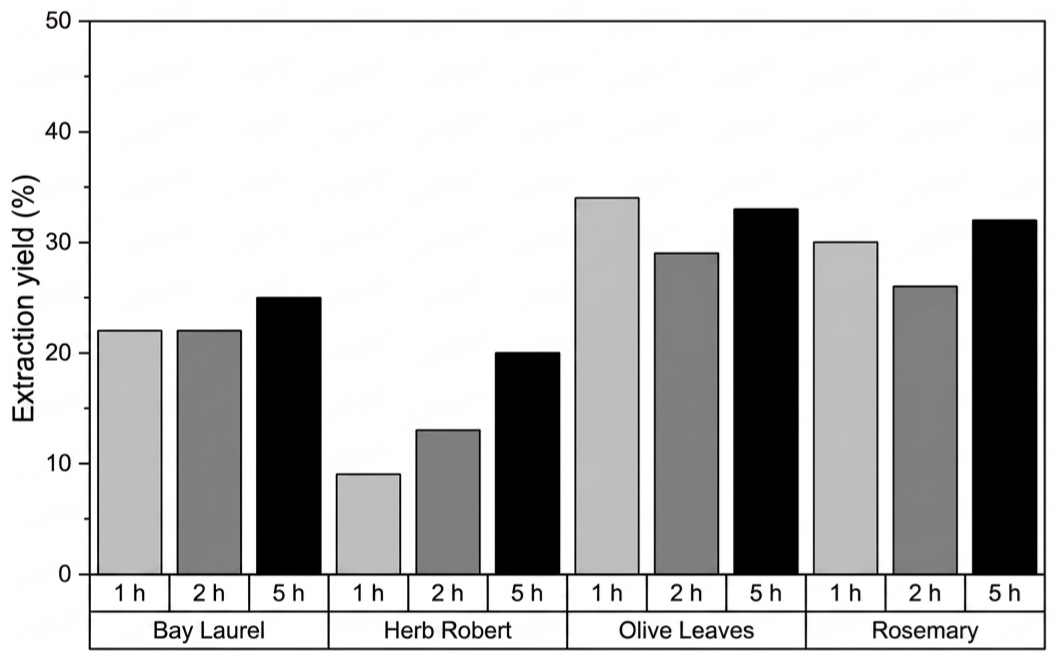
The slight non-monotonic variations observed for olive and rosemary extracts at 2 h may arise from experimental variability or from compositional changes occurring during prolonged heating. Overall, these descriptive results suggest species-dependent differences in the recovery of water-extractable material by infusion.
Qualitative Phytochemical Screening
The qualitative phytochemical screening of the aqueous infusion extracts indicated the presence of terpenoids, alkaloids, phenolic compounds, tannins, and flavonoids in all four plant species. Because these colorimetric assays are subject to cross-reactivity, the results should be interpreted as preliminary class-level screening rather than compound-level identification.
Compared with Vinha et al. (34), who found no terpenoids or alkaloids in olive leaves infusions prepared for only 10 min, our longer infusions (1–5 h) showed positive responses for both classes. Mrabet et al. (35) reported that aqueous maceration of L. nobilis at room temperature extracted alkaloids, phenolics, flavonoids, and tannins, but no terpenoids. Similarly, Kesiya et al. detected the presence of saponins and alkaloids in aqueous Laurus nobilis extracts prepared at ambient temperature, whereas flavonoids, tannins and phenolic compounds were not detected (36). These differences may reflect the influence of extraction time and temperature on class-level assay responses.
Maceration
Extraction Yield
A descriptive comparison of maceration yields across solvents of different polarity suggested differences associated with solvent type and extraction duration, Figure 2. When alcoholic solvents are employed, particularly under extended extraction times, transesterification reactions of ester-containing phytochemicals cannot be completely ruled out. Consequently, some detected ester derivatives may originate from native plant constituents, solvent-induced transformations, or a combination of both, and should therefore be interpreted with appropriate caution. Methanol generally gave the highest yields, followed by ethanol, while ethyl acetate, dichloromethane, and especially hexane resulted in lower extract mass recovery. For methanol and ethanol, yields tended to increase from 1 to 3 days, after which further changes were limited. Overall, within the limits of this screening design, the more polar solvents and extraction times up to three days tended to provide higher extract recovery.
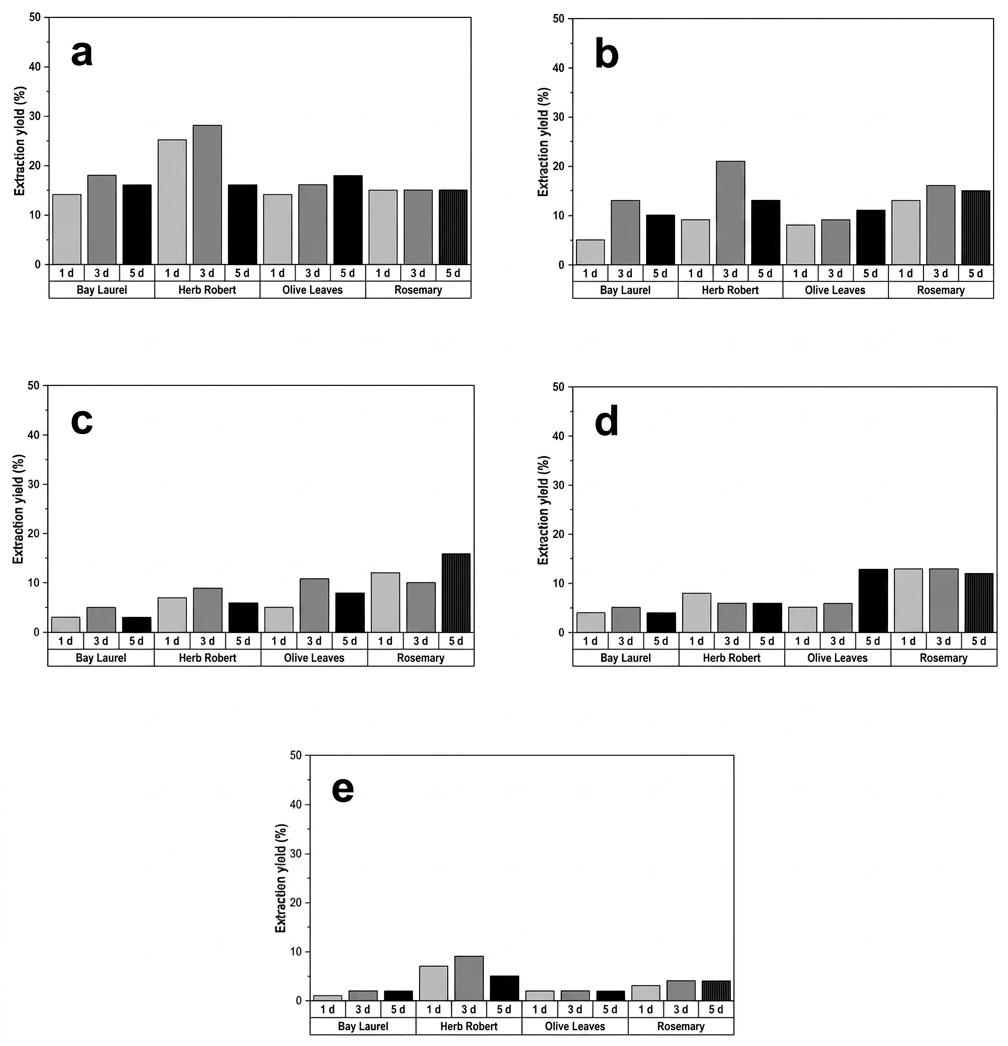
El Faqer et al. prepared an ethanolic extract of Moroccan L. nobilis L. by maceration for 48 h at room temperature, obtaining a yield of 17%, which was slightly higher than the yield we achieved (13%), possibly due to the higher purity of the ethanol used in their extraction. In the study by Zeroual et al., the maceration of rosemary was carried out twice consecutively for 72 h each, and the combined extracts were used to calculate the total extraction yield (22). Despite this extended extraction protocol and the use of finely powdered plant material, the reported yields (ranging from 4% to 19%), depending on the solvent and plant origin) are similar to those obtained in our work.
Qualitative Phytochemical Screening
The qualitative phytochemical screening of extracts obtained with solvents of different polarity showed differences in class-level assay responses among extraction conditions (Table 1). Methanolic extracts showed the broadest qualitative coverage, with all five classes detected in all species. In contrast, ethanolic extracts consistently lacked detectable tannins across all plant species, despite the presence of other classes. Extracts obtained with ethyl acetate and dichloromethane displayed intermediate selectivity, with tannins detected exclusively in bay laurel. In dichloromethane extracts, terpenoids were further restricted to olive leaves and rosemary. Hexane extracts also showed positive responses for several phytochemical classes; however, given the very low polarity of this solvent, these results should be interpreted cautiously as indicative signals rather than definitive evidence of efficient extraction. Overall, these colorimetric results are interpreted only as preliminary class-level indicators and not as compound-specific evidence of compositional differences.
| Solvent | Class | Bay Laurel | Herb Robert | Olive Leaves | Rosemary |
|---|---|---|---|---|---|
| Methanol | Terpenoids | + | + | + | + |
| Alkaloids | + | + | + | + | |
| Phenolic compounds | + | + | + | + | |
| Tannins | + | + | + | + | |
| Flavonoids | + | + | + | + | |
| Ethanol | Terpenoids | + | + | + | + |
| Alkaloids | + | + | + | + | |
| Phenolic compounds | + | + | + | + | |
| Tannins | - | - | - | - | |
| Flavonoids | + | + | + | + | |
| Ethyl Acetate | Terpenoids | + | + | + | + |
| Alkaloids | + | + | + | + | |
| Phenolic compounds | + | + | + | + | |
| Tannins | + | - | - | - | |
| Flavonoids | + | + | + | + | |
| Dichloromethane | Terpenoids | - | - | + | + |
| Alkaloids | + | + | + | + | |
| Phenolic compounds | + | + | + | + | |
| Tannins | + | - | - | - | |
| Flavonoids | + | + | + | + | |
| Hexane | Terpenoids | + | + | + | + |
| Alkaloids | + | + | + | + | |
| Phenolic compounds | + | + | + | + | |
| Tannins | + | - | - | - | |
| Flavonoids | + | + | + | + |
Soxhlet
Extraction Yield
The extraction yields obtained by Soxhlet extraction varied among solvents and plant species (Figure 3). Methanol and ethanol generally gave the highest yields, whereas ethyl acetate, dichloromethane, and hexane resulted in lower extract recovery. Herb Robert was an exception, as water-based Soxhlet extraction gave the highest yield for this species, exceeding 35%, while water produced much lower recoveries in the other plants. This pattern suggests that the recovery of extractable material under Soxhlet conditions was strongly species dependent.
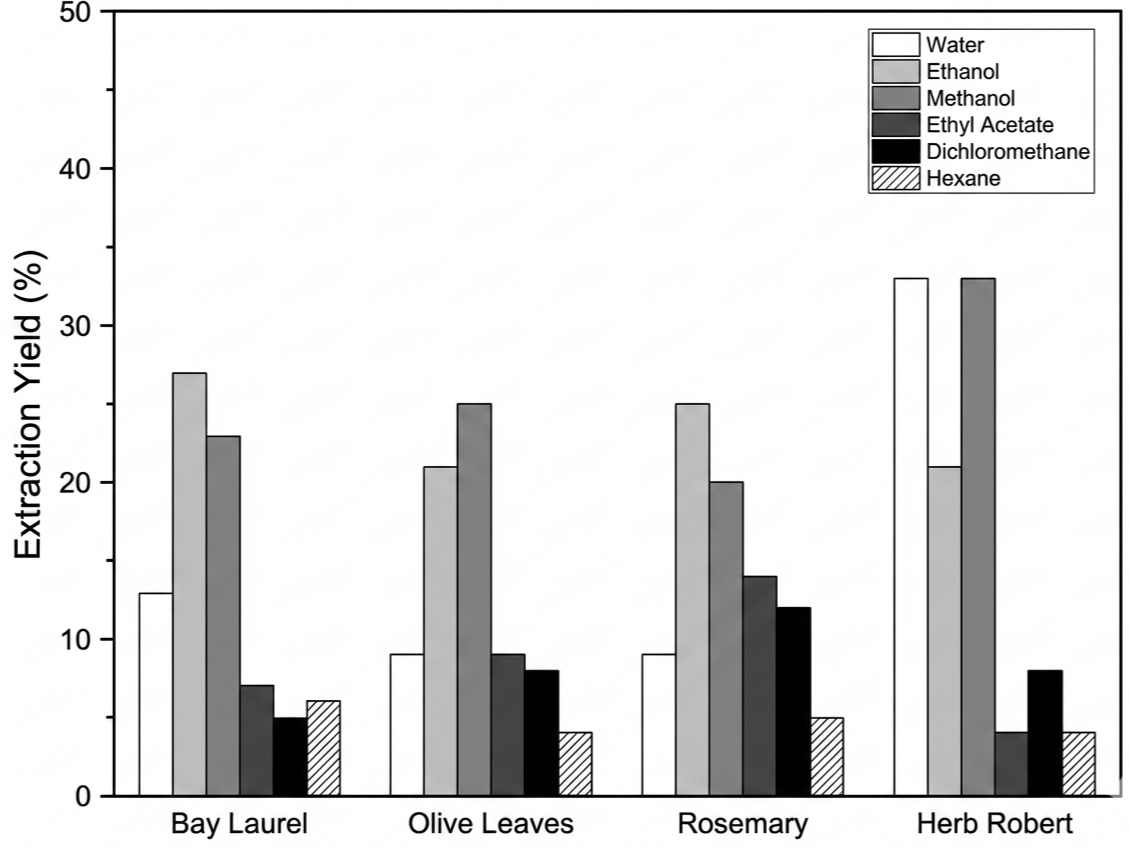
When compared descriptively with the maceration and infusion results, water again appeared particularly effective for herb Robert, whereas alcohol-based solvents tended to perform better for the other species. Because Soxhlet extraction involves prolonged heating and repeated solvent renewal, these differences should be interpreted cautiously within the exploratory scope of the study.
In our study, the ethanolic Soxhlet extraction of laurel leaves yielded 27%, which is slightly higher than the 22% reported by Peña-Ortiz et al., who performed the extraction using leaves previously ground to below 20 mesh (ca. 850 µm) (37). This result is notable because substantial extract recovery was still obtained without prior milling of the leaves. Compared with Tarhane et al., who obtained 48–49 % for finely ground leaves (< 250 µm), the differences can be mainly attributed to particle size, which strongly affects solvent accessibility and diffusion path length (38). Nevertheless, our yield remains within the same magnitude as that reported for rosemary by Zeroual et al., even though they used finely powdered material and performed two consecutive Soxhlet runs (22). Under the conditions tested, a single Soxhlet extraction yielded substantial extract recovery, even from intact or coarsely cut leaves.
GC-MS
Gas chromatography–mass spectrometry (GC–MS) was performed on ethanolic extracts because ethanol gave relatively high extraction yields and produced GC–MS-accessible profiles within the scope of this screening study, while remaining a more sustainable solvent and enabling straightforward GC–MS injection. The resulting chromatograms for extracts obtained by maceration and Soxhlet are shown in Figure 4a and Figure 4b, respectively. It should be noted that GC–MS is inherently biased toward volatile and semi-volatile, thermally stable, and relatively lipophilic compounds. Consequently, highly polar, non-volatile, or high-molecular-weight constituents may be underrepresented or not detected, and the reported profiles should be interpreted as a partial representation of the extract composition. All plant species were analyzed, and Table 2 summarizes the compounds tentatively identified in each extract. Accordingly, the GC–MS profiles discussed herein should be regarded as comparative fingerprints rather than exhaustive compositional descriptions, suitable for evaluating method- and species-dependent trends. Moreover, GC–MS provides information only on compounds amenable to volatilization and derivatization; therefore, non-volatile lipidic species such as triglycerides are not detected under the applied conditions.
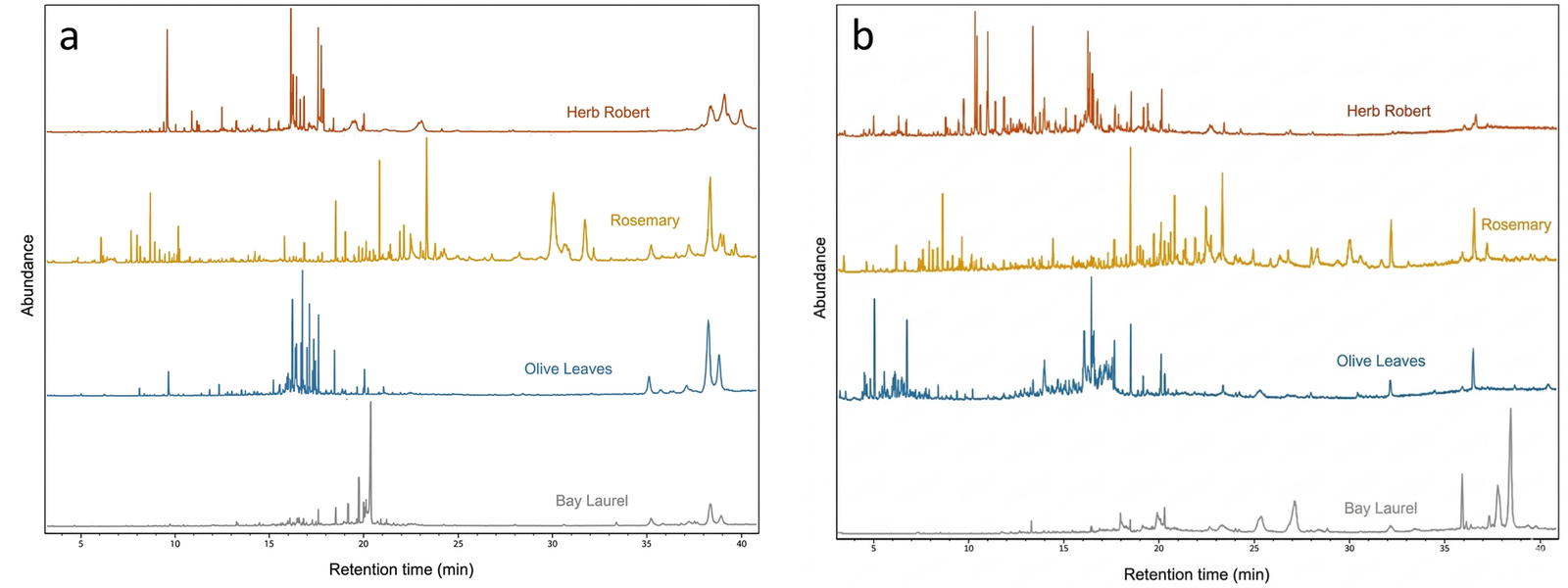
A descriptive difference between extraction methods was observed when comparing the two methods: Soxhlet extracts generally display more intense and more complex peak patterns at longer retention times, whereas maceration produces simpler chromatograms with comparatively reduced late-eluting signal.
Rosemary displayed one of the richest GC–MS profiles in both chromatograms, particularly in the mid-to-late retention-time region, where signals tentatively assigned to characteristic rosemary phenolic diterpenes such as carnosic acid, carnosol, rosmanol, rosmaquinone, and epirosmanol were observed. These tentative assignments are consistent with the richer chromatographic profile observed for rosemary.
In herb Robert, relatively intense peaks were observed at early-to-mid retention times, including signals tentatively assigned to simple phenolic derivatives (e. g. , vanillin and ethyl homovanillate) and carbohydrate-related compounds (aldoses/ketoses and a C5 alditol). In addition, signals tentatively assigned to lipid-derived constituents such as hexadecenoic acid were observed in the mid-to-late retention-time region. Silylated sugar derivatives formed after BSTFA derivatization were also prominent in olive leaf extracts, together with additional triterpenoid-related signals at longer retention times. Bay laurel displayed a comparatively more modest chromatographic profile, with fewer detectable peaks than the other species. Prominent peaks around 18–21 min included signals tentatively assigned to lipid-derived compounds, while terpenoid-related signals were also observed at longer retention times.
| Compound | Retention time (min) | |||||||
|---|---|---|---|---|---|---|---|---|
| Herb Robert | Rosemary | Olive leaves | Bay Laurel | |||||
| M | S | M | S | M | S | M | S | |
| Ethyl homovanillate | 13.3 | 13.3 | 13.0 | 13.3 | - | 13.3 | 13.3 | 13.3 |
| Vanillin | 14.2 | 14.4 | 14.4 | 14.4 | - | - | - | - |
| Aldoses/Ketoses | 16.0 -16.9 | 16.0-16.9 | 16.2 - 16.8 | 16.0-16.9 | 16.0-16.9 | 16.1 - 16.9 | 16 - 16.8 | - |
| Alditol C5 | 17.1 - 17.4 | 17.0 - 17.4 | - | 17.0 - 17.4 | 17.0 - 17.4 | 17.0 - 17.4 | 17.2 - 17.4 | - |
| Coumaric acid | - | - | - | - | - | - | 17.5 | - |
| Hexadecanoic acid | 18.0; 18.5 | 18.5 | 18.5 | 18.5 | 18.5 | 18.5 | 18.5 | 18.0 |
| Oxylipid/long-chain hydroxy acid (C18) | - | 18.8 | - | - | - | - | - | - |
| Phytol | - | - | - | - | 20.0 | - | - | 19.9 |
| α/β-Amyrin | 19.3 - 19.5; 21.1; 23.1 | - | - | - | - | - | - | - |
| Linoleic acid | 20.1 | 20.1 | 20.1 | 20.1 | 20.1 | 20.1 | 20.1 | - |
| N-octadecanol | - | 20.3 | 20.3 | 20.3 | - | 20.3 | 20.3 | 20.3 |
| Oleic Acid | - | - | 20.7 | - | - | - | - | - |
| b-Sitosterol | 22.6 | 22.6 | 22.4 | - | - | - | - | 22.6 |
| Carnosic acid | - | - | 23.3 | 23.3 | - | - | - | - |
| Lupeol | - | - | - | - | - | 25.3 | - | 25.4 |
| Carnosol | - | - | - | 26.6 | - | - | - | - |
| Rosmanol | - | - | - | 26.8 | - | - | - | - |
| Rosmanol-quinone | - | - | - | 28.0 | - | - | - | - |
| Epirosmanol | - | - | - | 28.2 | - | - | - | - |
| Betunilic acid | - | - | - | - | - | - | - | 27.1 |
| Triterpenoids (pentacyclic triterpene derivatives) | 37.1 - 40.0 | 36.5 | - | 30.0 - 31.6 | - | 32.1 - 37.8 | 35.9 - 37.7 | 32.1; 35.9 - 37.8 |
| Nonacosane | - | - | 32.1 | 32.2 | - | - | - | - |
| Lupenone | - | - | - | - | 35.2 - 37.2 | - | 35.3 | - |
| Tritriacontane | - | - | - | 36.5 | - | - | - | - |
| Oleanolic acid | - | - | - | 37.2 | - | - | - | - |
| Betulin | - | - | - | - | - | - | - | 38.5 |
| Triterpenoid derivative with higher mass (ester or more functionalized derivative) | - | - | - | - | - | 38.7; 40.4 | 38.4; 38.9 | - |
| Note: (-) non detected or low intensity compounds. Compounds were tentatively identified based on mass spectral library matching. | ||||||||
Limitations
These findings should be interpreted as descriptive comparative observations rather than as statistically validated quantitative conclusions. The main limitations of the study include the absence of full systematic replication across all extraction conditions, the lack of particle-size standardization, the absence of additional analytical correction for residual moisture beyond oven drying to constant weight, the non-specific nature of the qualitative colorimetric phytochemical assays, and the tentative character of GC–MS compound assignment in the absence of external standards for formal confirmation. Accordingly, the present results provide a preliminary comparative framework rather than a predictive optimization model, and they should serve as a basis for future studies incorporating replicated experiments, standardized sample preparation, and quantitative targeted phytochemical analysis.
Conclusion
This exploratory comparative screening study suggests that extraction yield and GC–MS-accessible phytochemical fingerprints varied under the tested conditions according to solvent polarity, extraction technique, and plant matrix. Among the organic solvents tested, methanol generally provided the highest extract mass recovery, whereas ethanol represented a comparatively efficient and more sustainable alternative under the conditions evaluated. Within the analytical window of GC–MS, Soxhlet extracts tended to display more complex late-eluting profiles than maceration extracts, and species-dependent fingerprints were observed, including diterpene-enriched rosemary extracts and stronger carbohydrate-related signals in herb Robert and olive leaves.
Abbreviations
ABTS = 2, 2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid); BSTFA = Bis (trimethylsilyl)trifluoroacetamide; DPPH = 2, 2-Diphenyl-1-picrylhydrazyl; GAE = Gallic acid equivalents; GC-MS = Gas Chromatography – Mass Spectrometry; TEAC = Trolox Equivalent Antioxidant Capacity.Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The data generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Statement
Not applicable.
Funding Information
This research was funded by COMPETE 2030, Portugal 2030 and European Union, through project Adikte - Sustainable Beauty Products (no 14342).
References
- Bitwell C, Indra SS, Luke C, Kakoma MK. A review of modern and conventional extraction techniques and their applications for extracting phytochemicals from plants. Scientific African. 2023;19:e01585. doi: https://doi.org/10.1016/j.sciaf.2023.e01585
- Altemimi A, Lakhssassi N, Baharlouei A, Watson D, Lightfoot D. Phytochemicals: Extraction, Isolation, and Identification of Bioactive Compounds from Plant Extracts. Plants. 2017;6(4):42. doi: https://doi.org/10.3390/plants6040042
- El Maaiden E, Bouzroud S, Nasser B, Moustaid K, El Mouttaqi A, Ibourki M, et al. A Comparative Study between Conventional and Advanced Extraction Techniques: Pharmaceutical and Cosmetic Properties of Plant Extracts. Molecules. 2022;27(7):2074. doi: https://doi.org/10.3390/molecules27072074
- Kumar A, P N, Kumar M, Jose A, Tomer V, Oz E, et al. Major Phytochemicals: Recent Advances in Health Benefits and Extraction Method. Molecules. 2023;28(2):887. doi: https://doi.org/10.3390/molecules28020887
- Öztürk M, Ender S, Kurtulbaş E, Şahin S. Olive leaf as a functional cosmetic ingredient and analysis of the principal component. Analytical Biochemistry. 2025;700:115802. doi: https://doi.org/10.1016/j.ab.2025.115802
- Abaza L, Taamalli A, Nsir H, Zarrouk M. Olive Tree (Olea europeae L.) Leaves: Importance and Advances in the Analysis of Phenolic Compounds. Antioxidants. 2015;4(4):682-698. doi: https://doi.org/10.3390/antiox4040682
- Asmaey MA, Elsoghiar AAM, Shaaban M, Moharram AM, El-Gaby MSA. Phenolics and Other Structural Compounds from Leaves of Olea europaea L.: Extraction Techniques and Pharmacological Activities. Chemistry Africa. 2024;7(10):5123-5148. doi: https://doi.org/10.1007/s42250-024-01110-1
- Markhali FS, Teixeira JA, Rocha CMR. Olive Tree Leaves—A Source of Valuable Active Compounds. Processes. 2020;8(9):1177. doi: https://doi.org/10.3390/pr8091177
- Clodoveo ML, Crupi P, Annunziato A, Corbo F. Innovative Extraction Technologies for Development of Functional Ingredients Based on Polyphenols from Olive Leaves. Foods. 2021;11(1):103. doi: https://doi.org/10.3390/foods11010103
- d’Avanzo N, Mancuso A, Mare R, Silletta A, Maurotti S, Parisi OI, et al. Olive Leaves and Citrus Peels: From Waste to Potential Resource for Cosmetic Products. Cosmetics. 2024;11(2):41. doi: https://doi.org/10.3390/cosmetics11020041
- Mylonaki S, Kiassos E, Makris DP, Kefalas P. Optimisation of the extraction of olive (Olea europaea) leaf phenolics using water/ethanol-based solvent systems and response surface methodology. Anal Bioanal Chem. 2008;392(5):977-985. doi: https://doi.org/10.1007/s00216-008-2353-9
- Alwazeer D, Elnasanelkasim MA, Engin T, Çiğdem A. Use of hydrogen-rich water as a green solvent for the extraction of phytochemicals: Case of olive leaves. Journal of Applied Research on Medicinal and Aromatic Plants. 2023;35:100472. doi: https://doi.org/10.1016/j.jarmap.2023.100472
- Guebebia S, Othman KB, Yahia Y, Romdhane M, Elfalleh W, Hannachi H. Effect of genotype and extraction method on polyphenols content, phenolic acids, and flavonoids of olive leaves (Olea europaea L. subsp. europaea). Int. J. Plant Based Pharm. 2021;2(1):17-24. doi: https://doi.org/10.62313/ijpbp.2022.9
- Ramírez EM, Brenes M, Romero C, Medina E. Olive Leaf Processing for Infusion Purposes. Foods. 2023;12(3):591. doi: https://doi.org/10.3390/foods12030591
- Catarino MD, Silva AMS, Cruz MT, Cardoso SM. Antioxidant and anti-inflammatory activities of Geranium robertianum L. decoctions. Food Funct. 2017;8(9):3355-3365. doi: https://doi.org/10.1039/c7fo00881c
- Graça VC, Ferreira IC, Santos PF. Phytochemical composition and biological activities of Geranium robertianum L.: A review. Industrial Crops and Products. 2016;87(9):363-378. doi: https://doi.org/10.1016/j.indcrop.2016.04.058
- Paun Roman G, Neagu E, Moroeanu V, Nechifor G, Lucian Radu G. Antioxidant activity of Geranium robertianum concentrated extracts by ultrafiltration process. Planta Med. 2009;75(09):321-325. doi: https://doi.org/10.1055/s-0029-1234557
- Neagu E, Paun G, Constantin D, Radu GL. Cytostatic activity of Geranium robertianum L. extracts processed by membrane procedures. Arabian Journal of Chemistry. 2017;10:S2547-S2553. doi: https://doi.org/10.1016/j.arabjc.2013.09.028
- Świątek Ł, Wasilewska I, Boguszewska A, Grzegorczyk A, Rezmer J, Rajtar B, et al. Herb Robert’s Gift against Human Diseases: Anticancer and Antimicrobial Activity of Geranium robertianum L. Pharmaceutics. 2023;15(5):1561. doi: https://doi.org/10.3390/pharmaceutics15051561
- I. O I, A. E. E. Phytochemical Analysis and Determination of Vitamin Contents of Geranium Robertianum. Iosrjdms. 2014;13(6):44-47. doi: https://doi.org/10.9790/0853-16634447
- González-Minero FJ, Bravo-Díaz L, Ayala-Gómez A. Rosmarinus officinalis L. (Rosemary): An Ancient Plant with Uses in Personal Healthcare and Cosmetics. Cosmetics. 2020;7(4):77. doi: https://doi.org/10.3390/cosmetics7040077
- Zeroual A, Sakar EH, Mahjoubi F, Chaouch M, Chaqroune A, Taleb M. Effects of Extraction Technique and Solvent on Phytochemicals, Antioxidant, and Antimicrobial Activities of Cultivated and Wild Rosemary (Rosmarinus officinalis L.) from Taounate Region (Northern Morocco). Biointerface Res Appl Chem. 2021;12(6):8441-8452. doi: https://doi.org/10.33263/briac126.84418452
- Mena P, Cirlini M, Tassotti M, Herrlinger K, Dall’Asta C, Del Rio D. Phytochemical Profiling of Flavonoids, Phenolic Acids, Terpenoids, and Volatile Fraction of a Rosemary (Rosmarinus officinalis L.) Extract. Molecules. 2016;21(11):1576. doi: https://doi.org/10.3390/molecules21111576
- Damianova S, Tasheva S, Stoyanova A, Damianov D. Investigation of Extracts from Rosemary (Rosmarinus officinalisL.) for Application in Cosmetics. Journal of Essential Oil Bearing Plants. 2010;13(1):1-11. doi: https://doi.org/10.1080/0972060x.2010.10643784
- Aamer HA, Al-Askar AA, Gaber MA, El-Tanbouly R, Abdelkhalek A, Behiry S, et al. Extraction, phytochemical characterization, and antifungal activity of Salvia rosmarinus extract. Open Chemistry. 2023;21(1):20230124. doi: https://doi.org/10.1515/chem-2023-0124
- Rincón E, Balu AM, Luque R, Serrano L. Mechanochemical extraction of antioxidant phenolic compounds from Mediterranean and medicinal Laurus nobilis: A comparative study with other traditional and green novel techniques. Industrial Crops and Products. 2019;141:111805. doi: https://doi.org/10.1016/j.indcrop.2019.111805
- Dobroslavić E, Repajić M, Dragović-Uzelac V, Elez Garofulić I. Isolation of Laurus nobilis Leaf Polyphenols: A Review on Current Techniques and Future Perspectives. Foods. 2022;11(2):235. doi: https://doi.org/10.3390/foods11020235
- Dobroslavić E, Elez Garofulić I, Zorić Z, Pedisić S, Dragović-Uzelac V. Polyphenolic Characterization and Antioxidant Capacity of Laurus nobilis L. Leaf Extracts Obtained by Green and Conventional Extraction Techniques. Processes. 2021;9(10):1840. doi: https://doi.org/10.3390/pr9101840
- Dobroslavić E, Elez Garofulić I, Šeparović J, Zorić Z, Pedisić S, Dragović-Uzelac V. Pressurized Liquid Extraction as a Novel Technique for the Isolation of Laurus nobilis L. Leaf Polyphenols. Molecules. 2022;27(16):5099. doi: https://doi.org/10.3390/molecules27165099
- Sharma V, Agarwal A. Physicochemical and antioxidant assays of methanol and hydromethanol extract of ariel parts of Indigofera tinctoria Linn. Indian J Pharm Sci. 2015;77(6):729. doi: https://doi.org/10.4103/0250-474x.174995
- Akinseye OR, Morayo AE, Olawumi AS. Qualitative and quantitative evaluation of the phytochemicals in dry, wet and oil extracts of the leaf of Morinda lucida. J Biol Agric Healthcare. 2017;7(7):22–25.
- Godlewska K, Pacyga P, Najda A, Michalak I. Investigation of Chemical Constituents and Antioxidant Activity of Biologically Active Plant-Derived Natural Products. Molecules. 2023;28(14):5572. doi: https://doi.org/10.3390/molecules28145572
- El Faqer O, Rais S, Elkoraichi I, El Amrani A, Dakir M, Zaid Y, et al. Phytochemical characterization and immunomodulatory effects of aqueous and ethanolic extracts and essential oil of Moroccan Laurus nobilis L. (Lauraceae) on human neutrophils. J Herbmed Pharmacol. 2022;12(1):92-99. doi: https://doi.org/10.34172/jhp.2023.08
- Vinha A. Phytochemical Characterization and Radical Scavenging Activity of Aqueous Extracts of Medicinal Plants from Portugal. Ejmp. 2012;2(4):335-347. doi: https://doi.org/10.9734/ejmp/2012/1946
- Mrabet A, Abdelfattah B, El Mansouri F, Simou A, Khaddor M. Bay Laurel of Northern Morocco: A Comprehensive Analysis of Its Phytochemical Profile, Mineralogical Composition, and Antioxidant Potential. Biophysica. 2024;4(2):238-255. doi: https://doi.org/10.3390/biophysica4020017
- Kesiya K, Poorane B, Vinoth A. Comparative Study of Phytochemical Screening and Antibacterial Activity of Laurus Nobilis and Pleurotus Ostreatus. Ijser. 2017;5(8):14-18. doi: https://doi.org/10.70729/ijser171786
- Peña-Ortiz M, Serrano L, Romero AA, García A. Bay Leaves Extracts as Active Additive for Food Protective Coatings. Foods. 2023;12(20):3741. doi: https://doi.org/10.3390/foods12203741
- Tarhane AK, Tarhane S, Dursun İ, Büyük F, Coşkun MR, Ersoy Y. GC-MS analysis of chemical composition and determination of antimicrobial activity of Laurel Leaf Extracts prepared by different methods and solvents. Pvj. 2024;44(3):619–28. doi: https://doi.org/10.29261/pakvetj/2024.207