RESEARCH ARTICLE
Isolation, Characterization, and Identification of Yeast from Fermented Catfish (pangasius Sp.) Sausage
Academic Editor: Ramanda Ahmad Rizal Rifa’i
Aquatic Functional Products|Vol. 1, Issue 1, pp. 31-38 (2025)
Received
Nov 22, 2025Revised
Dec 3, 2025Accepted
Dec 19, 2025Published
Dec 26, 2025
Abstract
Introduction
Fermented sausage is a product that results from the work of lactic acid-forming bacteria, either naturally present in meat or added as starter bacteria. Fermented sausage is made from a mixture of raw meat, fat, and other ingredients, which is then stuffed into casings and left to ferment and mature (1). The fermentation process involving microbial activity is carried out for various purposes, including to increase product shelf life, create distinctive flavor and aroma characteristics, and enhance nutritional value to produce functional food products (2). However, in addition to lactic acid bacteria (LAB), other microorganisms such as yeast are also found in the fermentation process and are thought to contribute to product quality. Yeast is one of the microorganisms used as an inoculum or starter in the fermentation process to produce certain products (3). Yeast in the fermentation process of patin fish sausage produces compounds that contribute to the aroma, flavor, and texture of the sausage. The acidic conditions formed during the sausage fermentation process affect the sensory characteristics, particularly the taste and texture of the product. The distinctive sour taste (tangy) is produced as a result of fermentation activity by yeast, giving fermented sausages their unique taste (4).
The dominant yeasts commonly found in fermented foods (fish and meat) are from the genera Candida, Saccharomyces, Pichia, Zygosaccharomyces, Debaryomyces, and Hanseniaspora (5). Yeast capable of breaking down proteins (having proteolytic activity) is thought to play an important role in the fermentation of fish such as belacan depik and fish paste (6). According to (7), yeast has various beneficial effects on human health and shows potential as a probiotic microorganism. The presence of yeast in fermented sausages affects product quality characteristics, including color, taste, and aroma, and has the ability to degrade peroxide compounds and exhibit lipolytic and proteolytic activity.
Several studies have attempted to explain the role of yeast as secondary microbiota in fermented products. Identification using classical methods of yeast isolated from sheep meat products showed the presence of several genera such as Debaryomyces, Candida, Yarrowia, Pichia, Rhodotorula, Cryptococcus, and Trichosporon (8). Evaluation of yeast diversity during sausage processing and maturation in various factories showed the presence of Candida and Trichosporon species in raw meat or in the early stages of maturation, while Candida zeylanoides, Yarrowia lipolytica, and Debaryomyces hansenii appeared in the final stages of sausage production (9). In the sausage fermentation process, lactic acid bacteria (LAB), which are commonly used as starters, have been extensively studied. However, the role of other microorganisms, such as yeast in sausage fermentation, is still not fully understood. Based on the above description, the author was interested in conducting research on the isolation, characterization, and identification of yeast in fermented patin fish sausage.
Methodology
Time and Places
This research was conducted from May 2025 to June 2025 at the Fisheries Product Processing Laboratory and the Fisheries Product Microbiology Laboratory, Faculty of Fisheries and Marine Sciences, University of Riau.
Materials
The ingredients used in making fermented sausage are catfish (Pangasius sp. ) obtained from traditional markets, polyamide casings with a diameter of 1.9, tapioca flour, carrageenan, isolate soy protein (ISP), salt, pepper (Piper nigrum L. ), garlic (Allium sativum L. ), red yeast rice (Monascus purpureus), skim milk, corn oil, frankfurters, bratwurst, smoke powder, and ice cubes. The materials used for yeast isolation, characterization, and identification testing were PDA (Potato Dextrose Agar), chloramphenicol (100 μg/mL), distilled water, 70% alcohol, physiological solution (0.9% NaCl), crystal violet, iodine, 95% ethanol, 0.5% safranin, malachite green 0.5%, CuSO solution 1%, immersion oil, Christensen's Urea Broth (1g peptone, 5g NaCl, 2g H2P4, 12μg red phenol), and 20% sterile urea).
The equipment needed to make fermented sausage includes containers, trays, coconut shells, sausage molds, mattress thread, stoves, boiling pots, scissors, and knives. Microbiological analysis uses equipment such as masks, gloves, Bunsen burners, mortars, cotton wool, label paper, aluminum foil, Erlenmeyer flasks, test tubes, Petri dishes, micropipettes, tips, measuring flasks, incubators, hot plates, autoclaves, object glasses, cover glasses, test tubes, ose nedles, Bunsen burners, and microscopes.
Research method
This Study used an exploratory approach, which aims to isolate and identify the types of yeast (genus level) found in fermented patin fish sausage. The yeast identification results were analyzed and presented in tables and figures to clearly visualize the data.
Sampling was conducted on day 0 (before fermentation) and day 6 (final fermentation stage). At each sampling time, pH, water activity (Aw), and total yeast counts were measured.
Research procedure
Making fermented catfish sausage
In the production of sausages, fresh catfish undergo a series of cleaning processes, including gutting, removal of the intestines and gills, and washing with running water. After that, the fish is cut into fillets. The catfish is minced for 60 seconds, then the prepared seasoning is added. The grinding process continues until the mixture reaches the desired consistency (smooth). Next, the mixture is stuffed into 1.9 cm diameter polyamide casings with a sausage length of 7-10 cm. The fish sausages are steamed at 40°C for 30 minutes, followed by a 24- hour conditioning process at room temperature. The next process involves baking at a temperature between 40°C and 45°C for 2 days, with each smoking session lasting 3 hours. The final stage is fermenting the sausage for 6 days (10).
Yeast Isolation
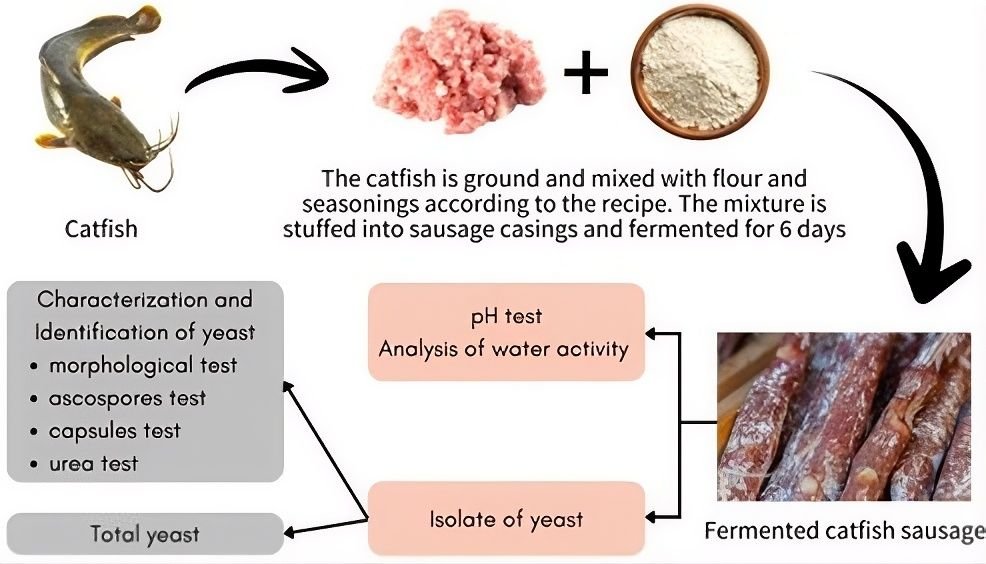
Samples of fermented catfish sausage were cultured using Total Plate Count on a medium (Potato Dextrose Agar) containing 100 μg/mL chloramphenicol (to inhibit bacterial growth), then incubated for 48 hours at 37°C. Colonies suspected to be yeast were observed morphologically under a microscope and subcultured on PDA medium and stored at 4°C (11). The overall experimental workflow, including sausage fermentation, physicochemical analysis, yeast isolation, and identification, is presented in Figure 1.
Water activity (Aw)
Water activity (Aw) was measured on days 0 and 6 of fermentation using a pre-calibrated Aw meter. A total of 4 g of crushed fermented sausage was placed in a measuring cup. The device was used until it showed the “completed” sign (12).
pH test
pH measurement was conducted on days 0 and 6 of fermentation using a calibrated pH meter. A total of 10 g of sausage sample was homogenized with 90 mL of distilled water to obtain a uniform suspension. The pH meter was calibrated using standard buffer solutions at pH 4.0 and 7.0 prior to measurement. The electrode was immersed in the homogenized sample, and the pH value was recorded once a stable reading was obtained (13).
Total yeast
Total yeast counts were determined on days 0 and 6 of fermentation. Total yeast testing begins with dilution, by homogenizing 10 g of fermented sausage sample. This homogenate is a 10-1 dilution. To obtain a 10-2 dilution, 1 mL of the 10-1 dilution is taken and added to a sterile reaction tube containing 9 mL of physiological saline, then homogenized. Continue the dilution process up to a 10⁻⁵ dilution. Then, transfer 1 mL of sample from the last four dilution series (10⁻³, 10⁻⁴, and 10⁻⁵) into Petri dishes and add 15 mL of PDA medium. Next, the petri dishes are incubated at 37°C for 3 days. After that, count the yeast that have grown on the petri dishes using the Eq. 1:
Identification and Characterization of Yeast at the Genus Level
Macroscopic and microscopic morphological observations
Macroscopic observation of yeast characteristics refers to the guidebook The Yeast Taxonomic Study (15) The morphological characteristics observed are based on the growth of colonies visible on solid media, which include the surface of the colony, the edge or margin of the colony, the texture of the colony, the color of the colony, and the elevation (16). Microscopic observation of yeast morphology was carried out by making a wet preparation, namely by placing the purified yeast isolate 3x24 hours on an object glass, then dripping sterile distilled water (17). The next step is observation under a microscope with a magnification of 4x10 to 100x10 to observe the size of the yeast, budding, and cell shape (18). The observation results refer to the book The Yeast: A Taxonomy Study by (15).
Ascospore test
The ascospore test was performed using a modified Schaeffer-Fulton technique with 0.5% malachite green and 0.5% safranin in a 1: 1 ratio. One drop of the isolate was stained with these dyes, then observed under a microscope. Mature ascospores appeared green, while vegetative yeast cells appeared red (19).
Capsule test
Capsule testing on yeast is performed using the smear method on a test slide. One ose of the isolate is placed on the test slide, which is then stained with crystal violet for ± 5 minutes and rinsed with a 1% CuSO4 solution. Observation is performed under a microscope at 1000x magnification with the addition of immersion oil. The capsule will appear as a faint blue circle surrounding the purple-colored vegetative yeast cells (19).
Urea test
The isolated yeast strain was inoculated into Christensen's Urea Broth medium, which consisted of a mixture of 1 g peptone, 5 g NaCl, 2 g H2PO4, and 12 μg phenol red dissolved in 1 L of sterile distilled water with a pH of 6.8. A total of 4.5 mL of the medium solution was transferred to a test tube and sterilized using an autoclave for 15 minutes. A total of 0.5 mL of sterile 20% urea was added to each tube. The strain was incubated at 25°C for 4 days and checked daily. A positive result of this test is the formation of a pink color on the medium in the tube (20).
Data Analysis
The data obtained is presented in tables and graphs to facilitate understanding and interpretation. Furthermore, the data is analyzed descriptively with reference to relevant literature studies to gain deeper insight into the findings.
Result and Discussion
Morphology of Yeast
Morphological observations were conducted macroscopically and microscopically on the isolates obtained. Morphological observations of the colonies included colony shape, edges, elevation, and color. Meanwhile, morphological observations of the cells were conducted to determine the cell shape of the yeast isolates. The results of the yeast morphological observations are presented in Figure 2.
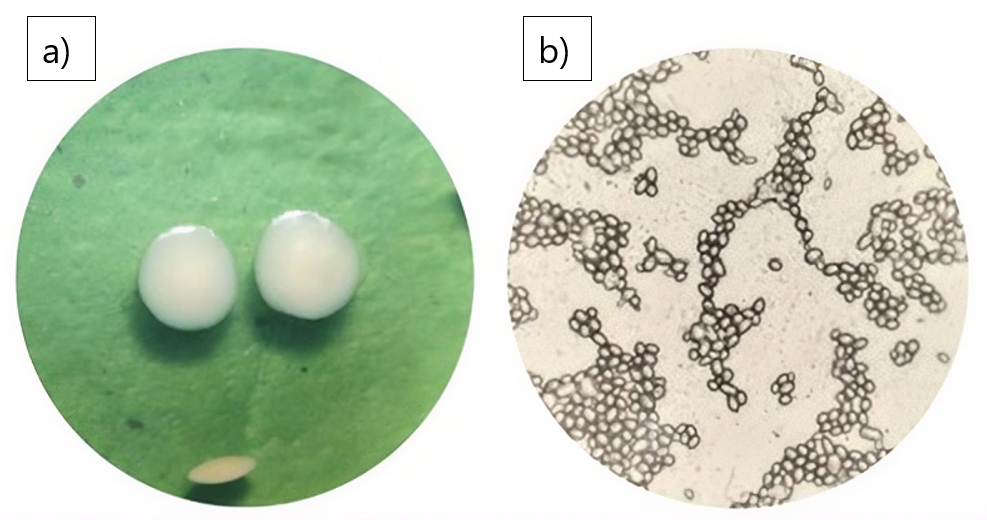
Based on Figure 2, macroscopic observations show that yeast colonies are milky white, evenly elevated, shiny with smooth and even edges. The same is reported by (21), that yeast isolates generally have a milky white color, and some yeast isolates have a creamy white, yellowish white, and brownish white color. Yeast isolates have a flat and raised elevation, all isolates have a shiny surface with smooth and flat edges. Microscopic observations show that yeast cells are round, oval, and without pseudohyphae. Based on the microscopic similarity of the isolate, it is suspected to belong to the Candida genus of yeast. This is supported by the opinion of (22), that yeast of the Candida genus have cell shapes that vary from round, oval, cylindrical to elongated, rarely apiculate, ogival, triangular or bottle-shaped with or without pseudohyphae. Asexual reproduction occurs through multilateral budding. However, to confirm that the isolated genus is indeed Candida, biochemical testing is required to ensure the yeast identification process is more accurate and clear.
Biochemical Characteristics of Yeast
Ascospore Test
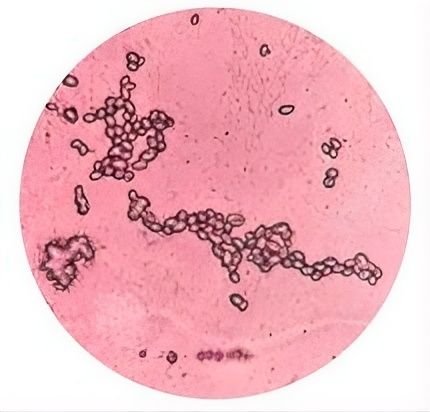
Based on Figure 3, the results of the ascosporulation test for the genus Candida were negative. No ascosporulation structures were visible after staining, because Candida is an anascoprogenic yeast that is unable to form asci or ascospores naturally (20). This is in line with the opinion of (22) that the genus Candida does not form ascospores, arthrospores, teliospores, or blastospores, but chlamydospores may form in some species that do not have carotenoid pigments and are therefore white to cream in color.
Capsule Test
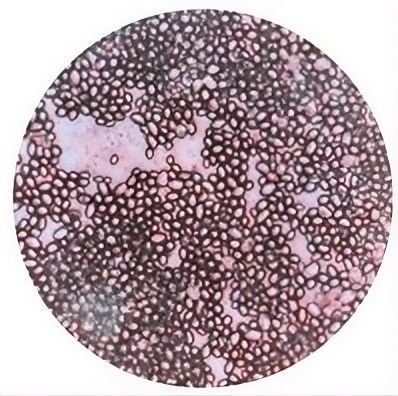
Based on Figure 4, the capsule staining test results were negative. No halo or capsule structure was detected, because Candida does not produce polysaccharide capsules (20). Capsule staining tests on yeast of the Candida genus have been conducted by (22), where based on microscopic observations, no capsules in the form of pale blue circles surrounding purple yeast cells were observed in the isolates examined.
Urease Test

Based on Figure 5, the urease test results for the Candida genus were negative. There was no change in the color of the medium to pink after incubation, indicating no urea hydrolysis (17). The absence of urease activity in this isolate supports that the genus found is consistent with the characteristics of Candida, which relies more on other metabolic mechanisms to survive and thrive in its host environment (23). This negative result distinguishes it from pathogenic yeasts such as Candida albicans, which are urease-positive and contribute to virulence in lung tissue (24). Urease testing on yeasts of the genus Candida has been conducted by (16), in which the yeast isolates tested showed no reaction of media color change to pink.
Chemical and Microbiological Analysis
| Parameter | Average | |
|---|---|---|
| Day - 0 | Day - 6 | |
| pH | 6, 70 | 4, 23 |
| Aw | 0, 67 | 0, 62 |
| Total yeast | 0 CFU/mL | 2, 6x104 CFU/mL |
Based on Table 1, it is known that fermented sausage undergoes changes in pH value during the fermentation process. Several factors that influence the decrease in pH are due to the smoking process. According to (25), pH can decrease during the smoking process because the smoke components that adhere to the sausage have an acidic taste, such as carboxylic acids, which include formic acid, acetic acid, and butyric acid. On day 0, fermented sausage was found to have a pH value of around 6. On the sixth day, the pH value of the sausage decreased to around 4. This was due to lactic acid bacteria beginning to work and producing acids that lowered the pH. According to (26), the longer the fermentation, the greater the decrease in pH due to an increase in the number of lactic acid bacteria, which also increases acid production. According to (27), this decrease in pH can also be caused by the growth of lactic acid bacteria, resulting in a lower pH of the substrate.
In addition, other factors include additives such as skim milk and liquid smoke. Skim milk can lower pH because it contains lactose, which can stimulate the growth of lactic acid bacteria, leading to fermentation (28). Liquid smoke can lower the pH value of a product (29). The pH value of liquid smoke ranges from 1.50 to 4.50, with quality classification 1 having a pH of 1.50–2.75 and quality 2 having a pH of 2.76–4.50. This value indicates the acidic nature of liquid smoke, which comes from the content of organic acid compounds such as acetic acid and formic acid (30).
Water Activity (Aw)
Based on Table 1, it is known that fermented sausage undergoes changes in water activity. The decrease in Aw value is also thought to be caused by the evaporation process due to smoking carried out before the fermentation process (10). The smoking process causes the surface of the sausage to become drier due to the evaporation of free water. The decrease in water activity (Aw) is caused by the drying process, which inhibits the growth of spoilage microorganisms (31). However, the Aw level must be maintained so that the microorganisms that act as fermenters can work properly. Microbes can generally grow on food materials with Aw 0.6–0.99 (32). This decrease in Aw is thought to be related to a decrease in product pH due to acids produced by yeast activity, causing proteins to lose their ability to retain water, resulting in dry fermented sausages (33). The longer the fermentation process lasts, the drier the fermented sausage will be and the better its appearance. This is supported by the opinion of (34) that a decrease in Aw helps reduce microbial growth and chemical reactions that damage materials, such as hydrolysis, browning, and fat oxidation.
Total Yeast
Based on the data in Table 1, it can be seen that the amount of yeast will increase as the fermentation time increases. This is in line with the research by (35), which found that fermentation time greatly affects yeast activity because the longer the fermentation, the greater the amount of yeast or the more active the yeast is in reproducing. Yeast can grow optimally under suitable conditions. Yeast growth factors can be influenced by the availability of nutrients, both macronutrients and micronutrients. The nutritional composition of the medium is very important to support yeast growth and metabolism (36). Nutrients in the growth medium will be used for growth, resulting in an increase in the number of yeast cells. 256 Another factor that also influences this is the length of time the isolate culture is stored, which can cause the nutrients in the medium to be completely consumed, leading to the death of the isolate in the storage medium. This can cause the isolate to be unable to grow in a new culture. In addition, it can also be caused by insufficient nutrients in the growth medium (37).
Conclusion
The conclusions of this study indicate that the yeast isolated from fermented catfish sausage belongs to the genus Candida sp., which exhibits diverse morphological characteristics, including round, oval, cylindrical, and elongated shapes, and may present with or without pseudohyphae. Biochemical characterization showed that Candida sp. does not form ascospores, does not possess capsules, and does not produce urea. The presence and activity of yeast were found to influence the chemical and microbiological properties of the fermented product, as reflected in an average pH value of 4.23, water activity (Aw) of 0.62, and total yeast count of 2.6 × 10⁴ CFU/mL. Furthermore, the results demonstrate that lactic acid bacteria (LAB) are not the only microorganisms involved in the fermentation process, as yeast also plays a significant role in contributing to the overall quality of the product. Consequently, these findings highlight the complex microbial synergy required to develop the unique flavor profiles and structural integrity characteristic of traditional fermented sausages.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data will be provided upon request to corresponding author.
Ethics Statement
Not applicable.
Funding Information
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
References
- Patriani P, Hafid H, Mirwandhono E, Wahyuni TH. Kualitas Daging Domba dengan Penerapan Teknologi Marinasi Menggunakan Kluwak Fermentasi terhadap Masa Simpan. Prosiding Seminar Nasional Teknologi Peternakan dan Veteriner 2019. 2019:491-499. doi: https://doi.org/10.14334/pros.semnas.tpv-2019-p.491-499
- Putra Syah S, Sumantri C, Isnafia Arief I, Taufik E. Karakteristik minuman whey yang difermentasikan dengan bakteri asam laktat indigenus asal dangke. jtip. 2017;28(2):129-138. doi: https://doi.org/10.6066/jtip.2017.28.2.129
- Yusuf M, Rahmawati R, Anshar M. Pemeriksaan kadar alkohol pada tapai ketan hitam yang difermentasikan dengan konsentrasi ragi yang berbeda. Medika. 2018;3(1):22-27. doi: https://doi.org/10.53861/jmed.v3i1.147
- Pratama A, Balia RL, Suryaningsih L. PENGARUH PENAMBAHAN YEAST (Candida apicola) PADA SOSIS FERMENTASI DAGING DOMBA TERHADAP KUALITAS FISIK, KIMIA DAN AKSEPTABILITAS. AGROINTEK: Jurnal Teknologi Industri Pertanian. 2021;15(2):574-582. doi: https://doi.org/10.21107/agrointek.v15i2.9791
- Kuncharoen N, Techo S, Savarajara A, Tanasupawat S. Identification and lipolytic activity of yeasts isolated from foods and wastes. Mycology. 2020;11(4):279-286. doi: https://doi.org/10.1080/21501203.2020.1745922
- Murlida E, Nilda C, Widayat HP, Muzaifa M. Karakterisasi dan identifikasi khamir proteolitik dari belacan depik, pasta ikan fermentasi khas Gayo. AGROINTEK: Jurnal Teknologi Industri Pertanian. 2024;18(2):455-461. doi: https://doi.org/10.21107/agrointek.v18i2.20022
- Encinas JP, López-Dı́az TM, Garcı́a-López ML, Otero A, Moreno B. Yeast populations on Spanish fermented sausages. Meat Science. 2000;54(3):203-208. doi: https://doi.org/10.1016/s0309-1740(99)00080-7
- Atit Kanti. Diversitas Khamir Indonesia untuk pengembangan Biofuel dan Bioindustri. Penerbit BRIN; 2022. doi: https://doi.org/10.55981/brin.727
- Encinas JP, López-Dı́az TM, Garcı́a-López ML, Otero A, Moreno B. Yeast populations on Spanish fermented sausages. Meat Science. 2000;54(3):203-208. doi: https://doi.org/10.1016/s0309-1740(99)00080-7
- Husni A, Brata AK, Budhiyanti SA. PENINGKATAN DAYA SIMPAN IKAN KEMBUNG DENGAN EKSTRAK ETANOLIK Padina sp. SELAMA PENYIMPANAN SUHU KAMAR. Jphpi. 2015;18(1). doi: https://doi.org/10.17844/jphpi.v18i1.9553
- Roostita R, Fleet G. The occurrence and growth of yeasts in Camembert and Blue-veined cheeses. International Journal of Food Microbiology. 1996;28(3):393-404. doi: https://doi.org/10.1016/0168-1605(95)00018-6
- AOAC Official Method 972.44Microbiological Method. New York: Oxford University Press; 2023. doi: https://doi.org/10.1093/9780197610145.003.2231
- Sardjono H, Khairiyati L, Perangin-angin WK, Syahadi M, Azzumar M, Amalia H, et al. Metrologi Kelistrikan Terapan: : Untuk Laboratorium Kalibrasi di Industri dan Perguruan Tinggi. LIPI Press; 2021. doi: https://doi.org/10.14203/press.234
- Sardjono H, Khairiyati L, Perangin-angin WK, Syahadi M, Azzumar M, Amalia H, et al. Metrologi Kelistrikan Terapan: : Untuk Laboratorium Kalibrasi di Industri dan Perguruan Tinggi. LIPI Press; 2021. doi: https://doi.org/10.14203/press.234
- Kurtzman CP, Fell JW. Preface. Elsevier; 1998. doi: https://doi.org/10.1016/b978-044481312-1/50000-9
- Maya FN, Alami NH. Uji Potensi Isolat Khamir Dari Rhizosfer Mangrove Wonorejo dan Gunung Anyar Sebagai Agen Penghasil IAA (Indole Acetic Acid). Jssits. 2019;8(1). doi: https://doi.org/10.12962/j23373520.v8i1.41855
- Musa U, Jurara AF, Mubarak AM. Isolation, Identification and Leavening Ability of Yeast from Local Fruits. Asian J Plant Biol. 2023;5(1):33-36. doi: https://doi.org/10.54987/ajpb.v5i1.825
- Yuniarti F, Hidayati W, Setiawati S, Nabilah K. Isolasi dan Uji Aktivitas Enzim Β-Galaktosidase Bakteri Asam Laktat (BAL) dari Fermentasi Buah Sirsak (Annona muricata L.). Al-Kauniyah J. Biol. 2022;15(1):28-35. doi: https://doi.org/10.15408/kauniyah.v15i1.15523
- Sinclair CG. Bergey's Manual of Determinative Bacteriology. The American Journal of Tropical Medicine and Hygiene. 1939;s1-19(6):605-606. doi: https://doi.org/10.4269/ajtmh.1939.s1-19.605
- Kurtzman CP, Fell JW, Boekhout T. Gene Sequence Analyses and other DNA-Based Methods for Yeast Species Recognition: Elsevier; 2011. doi: https://doi.org/10.1016/b978-0-444-52149-1.00010-0
- Azhara I, Rais M, Sukainah A, Praja Putra R. Isolasi dan identifikasi bakteri asam laktat pada fermentasi spontan biji kopi robusta asal bantaeng. Jtp. 2022;23(1):49-60. doi: https://doi.org/10.21776/ub.jtp.2022.023.01.5
- Maya FN, Alami NH. Uji Potensi Isolat Khamir Dari Rhizosfer Mangrove Wonorejo dan Gunung Anyar Sebagai Agen Penghasil IAA (Indole Acetic Acid). Jssits. 2019;8(1). doi: https://doi.org/10.12962/j23373520.v8i1.41855
- Booth JL, Vishniac HS. Urease testing and yeast taxonomy. Can. J. Microbiol. 1987;33(5):396-404. doi: https://doi.org/10.1139/m87-069
- Toplis B, Bosch C, Schwartz IS, Kenyon C, Boekhout T, Perfect JR, et al. The virulence factor urease and its unexplored role in the metabolism of Cryptococcus neoformans. FEMS Yeast Research. 2020;20(4). doi: https://doi.org/10.1093/femsyr/foaa031
- Jahidin JP. Pengaruh Pengasapan Sekam Padi Terhadap Kualitas Fisik dan Kimia Dendeng Batokok. jiiip. 2015;18(2):89-97. doi: https://doi.org/10.22437/jiiip.v18i2.2678
- Ramadhanti BW, Sumardianto S, Romadhon R. Karakteristik mutu dan kandungan senyawa volatil bekasam cumi-cumi dengan lama fermentasi yang berbeda. Jphpi. 2024;27(3):208-222. doi: https://doi.org/10.17844/jphpi.v27i3.48451
- Fuadi M, Surnaherman S. Cara Pengawetan Ikan Mas (Cyprinus carpio L) Dengan Menggunakan Fermentasi Limbah Kubis (Brassica oleracea). Agrintech.j.teknologi.pangan.dan.has.pertan. 2017;1(1):55-63. doi: https://doi.org/10.30596/agrintech.v1i1.1669
- Arsyad M, Hulinggi M. Formulasi Jagung Hibrida (Zea Mays L.) Dan Jagung Manis (Zea Mays Saccharata) Pada Pembuatan Susu Jagung. Perbal. 2019;7(3):178-192. doi: https://doi.org/10.30605/perbal.v7i3.1414
- Upet E, Salindeho N, Reo AR, Montolalu L, Kaparang JT, Makapedua DM, et al. Pengujian TPC, Kadar Air dan pH pada Ikan Tongkol (Euthynnus Affinis) Asap Cair Yang Disimpan Pada Suhu Ruang. Mthp. 2021;9(2):76. doi: https://doi.org/10.35800/mthp.9.2.2021.31144
- Elishian C, Zuas O. Penelitian Arsenik (As) di Indonesia. Prosiding Seminar Nasional Penerapan Ilmu Pengetahuan dan Teknologi : kampus merdeka meningkatkan kecerdasan sumberdaya manusia melalui interdispliner ilmu pengetahuan dan teknologi : Pontianak, 24 Agustus 2021. 2021;:189-198. doi: https://doi.org/10.26418/pipt.2021.40
- Ratnawati L. Pengaruh Waktu Penggilingan dan Teknik Pemasakan terhadap Kinetika Pengeringan Daging Giling. j. pangan. 2022;31(1):45-54. doi: https://doi.org/10.33964/jp.v31i1.543
- Nugroho DF, Wijayanti DA. Pengaruh penambahan sari wortel pada yoghurt ditinjau dari aw, kadar air, viskositas, total asam tertitrasi dan kadar protein. Ags. 2021;5(1):18. doi: https://doi.org/10.32585/ags.v5i1.1374
- Kurnianto MA, Munarko H. PENGARUH PENAMBAHAN KULTUR STARTER DAN METABOLIT Lactobacillus casei TERHADAP MUTU MIKROBIOLOGI SOSIS FERMENTASI IKAN PATIN (Pangasius sp.). J. Kelaut. Perikan. Terap. 2022;5(1). doi: https://doi.org/10.15578/jkpt.v5i1.10970
- Mayang Agil Ladensi, Sri Winarti, Luqman Agung Wicaksono. Pengaruh konsentrasi garam dan lama fermentasi terhadap karakteristik fisikokimia dan kandungan asam glutamat terasi nabati dari tempe gembus. jstpuho. 2024;9(4). doi: https://doi.org/10.63071/srb9k717
- Hartina F, Jannah A, Maunatin A. FERMENTASI TETES TEBU DARI PABRIK GULA PAGOTAN MADIUN MENGGUNAKAN Saccharomyces cerevisiae UNTUK MENGHASILKAN BIOETANOL DENGAN VARIASI pH DAN LAMA FERMENTASI. Al. 2014;. doi: https://doi.org/10.18860/al.v0i0.2907
- Walker G, Stewart G. Saccharomyces cerevisiae in the Production of Fermented Beverages. Beverages. 2016;2(4):30. doi: https://doi.org/10.3390/beverages2040030
- Fadila RN, Ratna M. Pemodelan Indeks Pembangunan Manusia dengan Metode Regresi Panel di Jawa Timur. Jssits. 2023;12(1). doi: https://doi.org/10.12962/j23373520.v12i1.100678