RESEARCH ARTICLE
Antidiabetic and Antihyperlipidemic Activity of Ethanolic Extract of Mentha viridis in Alloxan Induced Diabetic Rats
Academic Editor: Pilli Govindaiah
Sciences of Pharmacy|Vol. 3, Issue 3, pp. 167-176 (2024)
Received
Jun 22, 2024Revised
Sep 12, 2024Accepted
Sep 18, 2024Published
Sep 30, 2024
Abstract
Introduction
Diabetes mellitus refers to a group of heterogeneous metabolic disorders primarily characterized by chronic hyperglycemia, defined as a persistent elevation in blood glucose levels. The primary cause is either a disruption in insulin secretion, varying degrees of insulin resistance, or, more commonly, a combination of both. Diabetes mellitus, if left untreated, inadequately treated, or undiagnosed over a longer period, is most often correlated with the increased risk of cardiovascular diseases, kidney disorders, blindness, and foot cutoff (1, 2). According to IDF (International Diabetes Atlas, 10th edition), in the 21st century, diabetes is the fastest-growing health emergency worldwide. It has been projected that about 643 million people will have diabetes by 2030 and 783 million by 2045 (3). Diabetes mellitus substantially affects food nutrients such as carbohydrate, fat, and protein metabolism and sequentially causes chronic hyperglycemia followed by lipid profile abnormalities. Long-term untreated or poorly treated hyperglycemia considerably induces numerous microvascular and macrovascular diabetic complexities that are the ultimate reasons for diabetes-related morbidity and mortality (4).
The accessible medications for diabetes are insulin and varied oral antidiabetic agents like biguanides, sulfonylureas, thiazolidinediones, non-sulfonylureas secretagogues, and α-glucosidase inhibitors, etc. , alongside insulin (5, 6). Therefore, these drugs are used as monotherapy or combined to get better glycemic control and mask serious adverse effects of each oral antidiabetic agent (7). Over the last few decades, numerous studies have been done to find any potential in medicinal plants alone or a combination of oral antidiabetic agents in improving hyperglycemia and associated complications of DM in animal models (8). Plenty of plants have been carefully evaluated as a primary origin of dominant antidiabetic agents because herbal plants are a rich wellspring of phytoconstituents with insignificant toxicity or no side effects, making them a potential therapeutic choice for treating diabetes (9-11). Moreover, herbal medicine offers treatment at a cheaper rate than conventional medicine (9). An extensive review by Salehi et al. has covered many medicinal plants claimed to possess antidiabetic activity (8).
Mentha viridis (M. viridis), also known as Mentha spicata or spearmint, is a medicinal plant member of the Lamiaceae family widely grown in Europe, Asia, and North America but currently cultivated worldwide (12). This medicinal plant has many beneficial effects in its phytoconstituents and is utilized in numerous disorders such as diabetes, respiratory diseases, and skin disorders (13-15). According to Benkhnigue et al. , for diabetes therapy, the leaf of M. viridis is orally given as a decoction in the locality of Al Haouz-Rhamna in Morocco (16). In another study reported by Idm’ hand et al. , the leaf and stem of M. viridis are used to treat diabetes as a decoction or infusion form (13). Aqueous ethanolic extract of M. viridis exhibited blood glucose lowering and hypolipidemic effect in alloxan-caused diabetic rats (17). Additionally, aqueous leaf extract showed positive results in hyperglycemia and lipid abnormalities in diabetic animals (18). Phenolic leaf extract of M. viridis had antidiabetic effects in chemically induced diabetic rats, as reported elsewhere (19).
Notably, numerous studies reported that preliminary screening of M. viridis disclosed the existence of phytoconstituents, for example, tannins, polyphenols, steroids, flavonoids, triterpenes, and glycosides (20). Per one study, ethanolic extracts of M. viridis contain a substantial amount of phenolic compounds, including polyphenols, flavonoids, and caffeic acid derivatives (21). In essential oils extracted from M. viridis, carvone was an entire primary component besides trans-carveol, limonene, linalool, menthone, piperitone, piperitone oxide, and isomenthone (8). This research work was aimed to investigate the chemical composition of the ethanolic extract of M. viridis and to evaluate the antihyperglycemic and antihyperlipidemic activities of M. viridis separately and in blending with metformin in chemically induced diabetic rats by alloxan.
Methods and Materials
Chemicals
All the chemicals employed in this investigation were of analytical grade. Alloxan was bought from German-based Sigma Chemicals. Glucose was procured from Glaxo Smith Kline. The rest of the chemicals, like triglycerides (TG), ligh-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and total cholesterol (TC) kits, were obtained from (LINEAR CHEMICALS S. L. , Spain). The reference drug, metformin, was purchased from Chadwell Health Essex, England. The required solutions were prepared on each day of the experiment.
Plant Collection
Leaves and barks of M. viridis were collected from Kawranbazar, Dhaka 1215, Bangladesh. The whole plant (leaves and barks) was identified by a Bangladesh National Herbarium specialist in Mirpur, Dhaka, Bangladesh. The accession number is DACB–41939.
Extract Preparation
Leaves and barks of M. viridis were rigorously cleaned with water and afterwards dried for seven days under sunlight. The coarse powder was obtained from the plant parts after grinding with an appropriate grinder machine. The dried and powdered materials (100 g) from each plant part were immersed in 500 mL of 90% ethanol for two weeks at ambient temperature with periodic shaking. Initially, a cotton filter and finally, a Whatman No. 1 filter paper were utilized to filter the solution. A rotary evaporator from Bibby Sterlin Ltd, UK, concentrated the filtrate at 40°C. A semisolid extract (2.08 g each) was obtained when the extra solvent evaporated.
Experimental Animal
Long Evans male rats (100–120 g) aged nine weeks were obtained from the ICDDR, B (International Centre for Diarrheal Disease Research, Bangladesh). The standard atmospheric states, such as 22–25°C temperature, 60–65% humidity, light/dark cycle (12/12 h), etc. , were maintained when rats were kept in animal cases. Throughout the experiment, all rats were given food like standard laboratory diet (Purina rat chow) from ICDDR, B, Dhaka, and pure drinking water. All animal experiments were performed after the agreement with the Committee of Animal Ethics of Southeast University, Department of Pharmacy.
Acute Toxicity Studies
Long Evans male rats were fasted overnight and were selected for the study. Each extract (whole plant and leaf extract only) was administered orally to two groups (n = 5) of rats. The doses were 250 mg/kgBW and 500 mg/kgBW (for whole plant and leaf extract). Following administration of all the extracts and metformin, the animals were perceived intimately, particularly for the initial three h, for expression in abnormalities, such as salivation, surged motor activity, chronic convulsions, coma, and death. Regular inspections were done at a uniform gap for a single whole day. This regular monitoring was continued for 4 days.
Organizing of Investigational Rats
Long-Evans rats were arbitrarily allocated into 7 groups. Each group contains five rats (n = 5) and utilized test studies, including the blood glucose estimation, evaluation of lipid profile, etc. , following 14-day treatment protocols.
- Group I: Healthy normal rats with no treatment (Normal control)
- Group II: Diabetic control rats (Untreated Group) (Negative control)
- Group III: Diabetic rats administered leaf extract (500 mg/kgBW)
- Group IV: Diabetic rats administered herb extract (500 mg/kgBW)
- Group V: Diabetic rats administered metformin (850 mg/70 kgBW)
- Group VI: Diabetic rats administered a blending of leaf extract (250 mg/kgBW) and metformin (425 mg/70 kgBW)
- Group VII: Diabetic rats administered a blending of whole plant extract (250 mg/kgBW) and metformin (425 mg/70 kgBW)
Diabetes Induction
A newly prepared alloxan solution (120 mg/kgBW) in distilled water was injected intraperitoneally singly into each rat after 12 h of overnight fasting. These animals were given a 10% glucose solution to drink to deal with alloxan-induced low blood sugar, as there was an instantaneous rise in blood insulin just after the alloxan injection within minutes, called the initial transient hypoglycemic phase (22, 23). Blood glucose content was estimated from the tail vein of diabetic rats 72 h later. With marked hyperglycemia (FBG (fasting blood glucose) ≥25.70 mmol/L were chosen for the successive investigation.
Preparation of Dosage of Reference Drug and Plant Extract
Preparation of Extract Solution (Leaf and Whole Plant)
The extracts (leaf and whole plants) were semisolid and sparingly soluble in water. The suspension form of the water dosages was prepared so that each 0.1 mL of solution contained plant extract as specified by the 500 mg/kgBW dose.
Preparation of Metformin Solution
The physical appearance of metformin was a white crystalline solid and was highly soluble in water. That’s why the dosages were prepared in solution form using distilled water so that each 0.1 mL of solution contained metformin following the dose of 850 mg/70 kgBW. In humans, this drug works effectively in the same dose.
Preparation of Leaf Extract and Metformin Combination
The dosage was prepared individually so that each 0.1 mL of solution contained leaf extract and metformin in line with the dose of 250 mg/kgBW of leaf extract and 425 mg/70 kgBW of metformin in the given order.
Preparation of Whole Plant Extract and Metformin Combination
The dosage was prepared individually so that each 0.1 mL of solution contained plant extract and metformin as per the dose of 250 mg/kgBW of plant extract and 425 mg/70 kgBW of metformin in the order given.
Blood Serum Collection
Following the completion of the two weeks of treatment with the drug and extracts (whole plant and leaf), chloroform was employed to anesthetize the rats. After confirming that the rats had become unconscious, the thoracic artery was opened by cutting their abdominal skin. 3–4 mL of blood collected directly from the thoracic artery by syringe immediately after opening their skin. The blood sample was centrifuged at 4000 rpm (rotate per minute) for 20 min using a centrifuge machine (Digisystem Laboratory Instrument Inc. Taiwan). The supernatant plasma samples were decanted and stored at -4°C until biochemical examinations were done.
Lipid Profile
Lipid profile parameters like TC, TG, HDL-C, and LDL-C were estimated colorimetrically by a hematology analyzer using wet reagent diagnostic kits from Randox, UK.
Phytochemical Screening
Ethanolic leaf extract of M. viridis was utilized to carry out the phytochemical screening. Standard procedures were followed to test the phytochemicals such as alkaloids, carbohydrates, flavonoids, resins, saponins, steroids, tannins, and phenols (24–27).
Alkaloids
In a beaker, plant extract of M. viridis (2 mg), distilled water (5 mL), and 1% hydrochloric acid (8 mL) were taken and stirred carefully and very gently until a reaction happened. In 2 mL of this mixture, Dragendorff’s reagent (1 mL) was put on dropwise. The appearance of turbidity or precipitate designates the existence of alkaloids.
Carbohydrates
10 mL of distilled water mixed with 2 mg of plant extract was filtered, and the filtrate was condensed afterwards. Newly prepared 20% α-naphthol (2 drops) was added into this filtrate, and then 2 mL of concentrated sulphuric acid was added dropwise. The formation of a red violet ring indicates the presence of carbohydrates, which fades away from the surplus inclusion of alkali.
Flavonoids
Plant extract (2 mg) was dissolved in ethanol (5 mL) and filtered in a small beaker. Concentrated hydrochloric acid (a few drops) was added carefully to this filtrate. Then, a small piece of magnesium was incorporated, and the pink or reddish coloration proved the presence of flavonoids.
Resins
Plant extract (1 mL) was mixed with copper acetate. The solution was shaken vigorously and left for a few minutes to separate. A green color appearance in the solution demonstrated the presence of resins.
Saponins
Plant extract (0.5 mg) was dissolved in 10 mL of distilled water. Then, the solution was shaken, covered, and left for 30 min. The solution formed a honeycomb-like foam that denoted the appearance of saponins.
Steroids
A 0.2 mg of dry extract was shaken with 2 mL of chloroform, and then concentrated sulphuric acid was added to this mixture carefully by the sides of the test tube. The appearance of a reddish-brown color at the interphase revealed the presence of steroids.
Tannins
10 mL distilled water was mixed with 1–2 mg of plant extract and filtered. A few drops of 0.1% FeCl3 solution were added gently to this filtrate. The presence of tannins was confirmed by the green, blue-green, or blue-black precipitate.
Phenols
Plant extract (0.2 mg) was dissolved in a 5% FeCl3 solution. The formation of green precipitate specified the presence of phenols.
Terpenoids
Plant extract (0.5 mg) was added in 2 mL chloroform, and then the solution was mixed with 3 mL concentrated sulphuric acid. The appearance of a reddish-brown color confirmed the presence of terpenoids.
Statistical Analysis
The statistical analysis was carried out by one-way analysis of variance (ANOVA), followed by Dunnett’s post-hoc test or students paired or unpaired T-test where applicable. The results were represented as mean ± SEM (standard error of the mean). Results were set as significant when p< 0.05.
Results
Effect on Blood Glucose Level
After diabetes induction, group III to group VII rats were treated with the ethanolic extract of leaf and whole plant of M. viridis and a combination of metformin and plant extract (leaf and whole plant) for two weeks. The effects of handling extracts and combination therapy for two weeks on blood glucose levels in alloxan-induced diabetic rats were represented in Figure 1. As can be seen, the BGL (blood glucose level) was decreased significantly (62.82%) (p< 0.05) with leaf extract (500 mg/kgBW), and (72.89%) (p< 0.05) for the combination of whole plant and metformin (250 and 425 mg/70kgBW), rather than single whole plant extract (43.76%), and combination therapy of metformin with leaf extract (45.88%) compared to untreated DC rats (Group II) (31.80 mmol/L). On the other hand, singly metformin (850 mg/70 kgBW) decreased BGL (65.11%), which was notable (p< 0.05) in contrast to the untreated DC rats. The leaf extract decreased blood glucose levels from 31.8 mmol/L to 4.9 mmol/L, which was very significant (p< 0.05).
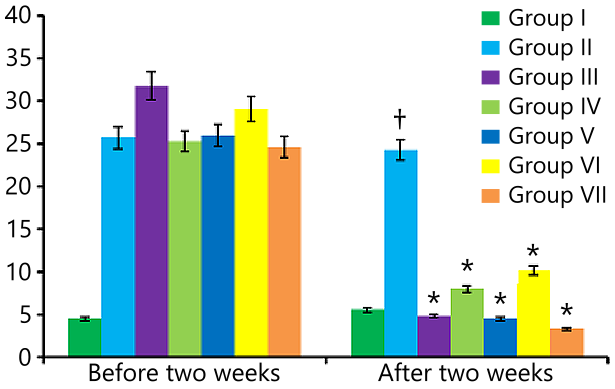
In the other treatment, combining whole plant extract with metformin decreased BGL from 24.6 mmol/L to 3.4 mmol/L compared to the untreated DC group, which was also significant (p< 0.05). Notably, all the treatments, leaf extract, whole plant extracts, single metformin, and both the combination therapies reduced the blood glucose level remarkably (p< 0.05) in treated diabetic groups (III-VII) as opposed to group II (untreated DC group) (see Figure 1). Here, one combination therapy of metformin with whole plant extract showed the maximum reduction in BGL, which was 72.89%.
Effect on Lipid Profiles
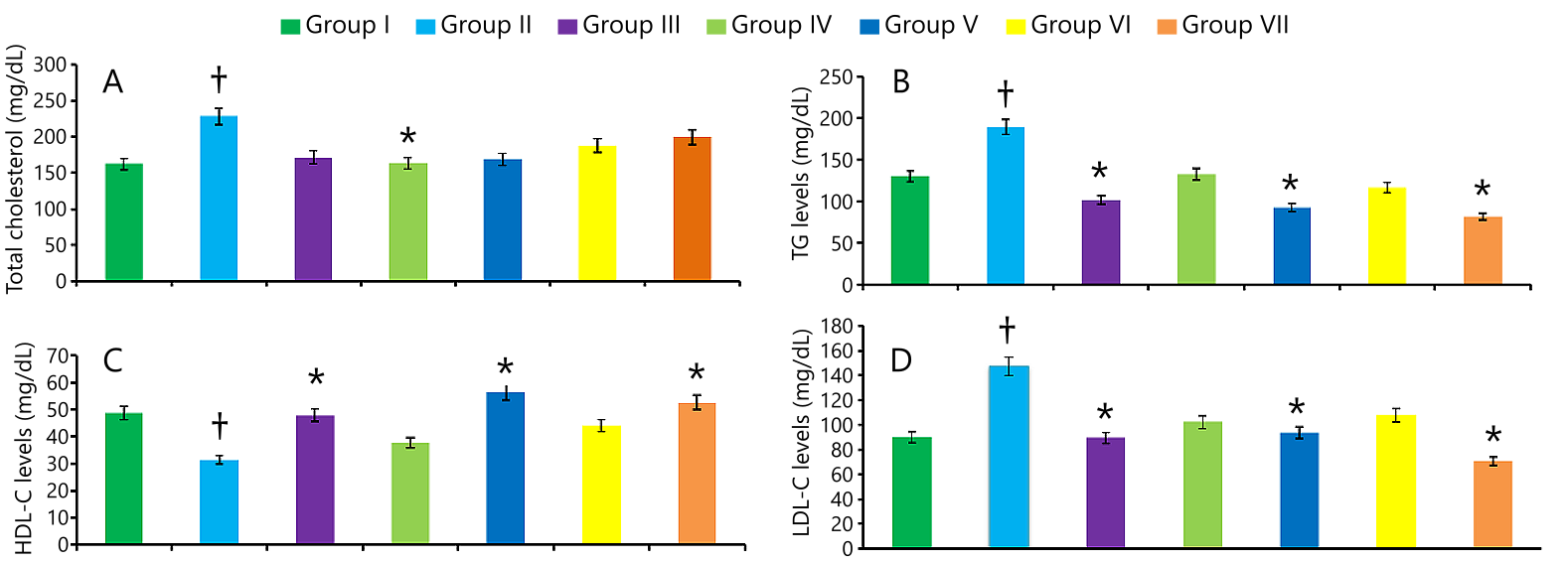
Several studies reported that diabetes caused by alloxan surged the blood glucose levels effectively in animal models. Together, they can raise TC, TG, and LDL-C cholesterol levels and decline HDL-C levels sequentially (28-30). It can be seen from Figure 2 that, in DC rats with no treatment (group II), all the lipid profiles, for instance, TC, TG, and LDL-C increased when compared to NC rats (group I). On the contrary, HDL-cholesterol decreased in the untreated DC group compared to the NC group. These findings agree with the previously reported work: alloxan induction aggressively deteriorates the lipid profiles in untreated DC rats, in addition to blood glucose level increments (22, 23). After two weeks of treatment with the leaf extract, plant extract, and blending therapy of metformin with the leaf, the plant extract reduced the elevated levels of TC, TG, and LDL-C. They increased the low levels of HDL-C in respective diabetic rats (group III – group VII), as seen in Figure 2. It has been noticed that the TC level (29.17%), TG level (89.55%) (p< 0.05), LDL-C level (68.67%) (p< 0.05) increased, and HDL-cholesterol level (25.52%) decreased in alloxan-induced untreated diabetic rats as contrasted with their corresponding normal rats after diabetes induction.
Interestingly, it was observed that a noticeable drop in TG, LDL-C (p< 0.05) and surge in HDL-C levels (p< 0.05) in alloxan-induced diabetic rats handled with ethanolic leaf extract of M. viridis, metformin alone, and metformin with whole plant extract after two weeks treatment. In contrast, whole plant extracts and one combination of metformin with leaf extract improved the dyslipidemic condition, particularly TG, LDL-C, and HDL-C in treated diabetic rats. However, it did not seem very satisfactory. On the other hand, no therapy showed a noteworthy response in reducing TC in treated hyperglycemic rats except the whole plant extract. Therefore, the conclusion drawn here is that both the leaf extract and one blending therapy of metformin with the entire plant extract produced beneficial effects on TG, LDL-C, and HDL-C levels in diabetic rats following two weeks of treatment.
| Phytochemical constituents | Result |
|---|---|
| Alkaloids | + |
| Carbohydrates | - |
| Resins | + |
| Terpenoids | + |
| Tannins | + |
| Saponins | - |
| Flavonoids | + |
| Phenols | + |
| Steroids | + |
Preliminary Phytochemical Screening
Phytochemicals estimations were carried out for qualitative analysis to reveal the existence or lack of major secondary metabolites such as alkaloids, carbohydrates, phenolic contents, terpenoids, saponins, resins, steroids, etc. , in the extracts of the leaf of M. viridis. The results of phytochemical screening are summed up in Table 1. Alkaloids, resins, tannins, steroids, terpenoids, flavonoids, and phenolic compounds appeared in the plant extract. However, saponins and carbohydrates were absent in the extract.
Discussion
The primary focus of selecting the M. viridis medicinal plant and applying it as a therapy in diabetic rats was to see any beneficial outcome in the ethanolic extract in managing chemically induced diabetes mellitus. However, this helpful result would come either by reducing elevated blood glucose levels or correcting any lipid profile disturbance. To induce diabetes in experimental animals, a single intraperitoneal injection of alloxan (2, 4, 5, 6-tetraoxypyrimidine) was utilized at a dose of (120 mg/kgBW). The mechanism of how alloxan induces diabetes in experimental animals and, consequently, its deteriorated effect is reasonably well understood. Following the previously reported study, two rationales might be accountable. One is the particular retardation of glucokinase, a glucose sensor of the beta cell resulting in insulin secretion inhibition in response to glucose. Second is the capability of alloxan to prompt reactive oxygen species (ROS) formation, which in succession causes the selective death of beta cells (22). Noteworthy, chemically, alloxan is a diabetogenic agent that is inactive but converted to an active toxin inside the body called protoxin. This foreign substance is subjected to intracellular metabolism, generating ROS in redox cycling reactions between alloxan and dialuric acid (22). It has been observed in this study that only one dose of alloxan injection caused the BGL increment significantly in untreated DC rats & diabetic rats (group III-VII) as compared to the NC group (with no alloxan treatment) as can be seen from Figure 1. The significant increase in BGL in animals might be caused by the selective uptake of alloxan to beta cells via glucose transporter, GLUT2, followed by several factors such as origination of ROS, gradual deterioration of beta cell function, beta cell demise by necrosis, and finally diabetes mellitus which is insulin dependent.
All these phenomena produced chemical diabetes termed alloxan diabetes (22). After daily treatment for two weeks with the leaf and plant extract of M. viridis singly, combining these extracts with metformin reduced the elevated BGL significantly (p< 0.05), as found in Figure 1. Among the treatments, leaf extract (500 mg/kgBW) (62.82%) and the amalgamation of whole plant and metformin (250 and 425 mg/70kgBW) (72.89%) decreased BGL more effectually. Single oral administration of metformin reduced BGL (65.11%) significantly compared to the untreated DC group. Similar observations were found in previously reported studies where combination therapy worked more effectively in managing hyperglycemia than a single therapy (29–33). This result suggests that the leaf of M. viridis and the combination of whole plant extract with an oral antidiabetic agent, metformin, can control hyperglycemia by stimulating or regenerating the beta cells to secrete insulin from islets of Langerhans.
On the contrary, the mode of action of the reference drug, metformin, on lowering BGL is explicit (34, 35). Here, the observed significant antihyperglycemic effect of leaf extract might be due to phytochemicals such as flavonoids, terpenoids, phenols and tannins. The most significant synergistic effect in lowering blood sugar levels was observed with the combination of whole plant extract and metformin. According to the literature, phytochemicals such as flavonoids (36, 37), tannins (38), and terpenoids (39, 40) present in different plant extracts were found in the management of diabetes (41).
In insulin-dependent type 1 diabetes with poor glycemic control, lipid abnormalities such as increased plasma triglycerides, LDL-C, and low levels of HDL-C are often noticed (42, 43). Insulin deficiency, insulin resistance, and hyperglycemia could change plasma lipid/lipoprotein metabolism, resulting in lipoprotein abnormalities (44, 45). This study also investigated TG, TC, LDL-C, and HDL-C lipid profiles. Compared to the NC group in the untreated DC group and diabetic (II-VII) groups, all the parameters, for instance, TC, TG, and LDC-C levels were climbed, whereas HDL-C declined notably. Similar observations were found in previously reported studies (29-31, 46). Diabetic rats treated with either M. viridis extract (leaf and whole plant) or one combination therapy of extract with reference antidiabetic drug, metformin (leaf with metformin) for two weeks exhibited a remarkable drop in TG and LDL-C levels and a notable rise in HDL-C level when compared with untreated DC group. No noticeable response was found in reducing the serum TC level in hyperglycemic rats following two weeks of treatment with leaf extract and combination therapy.
Only whole plant extract provided a significant outcome in reducing the TC level, as can be seen in Figure 2A. This result of correcting dyslipidemia agrees well with previous studies in animal models with diabetes (41). The findings here suggested that ethanolic crude extract of M. viridis may restore the beta cells and thus potentiate insulin release in pancreatic islets of alloxan-induced diabetic rats. In a previously reported study, a water-soluble alcoholic extract of Gymnema sylvestre possessed antidiabetic features, including better management of blood glucose level, betterment in hyperlipidemia, enhancement in beta cell functions, and beta cell retrieval in animal diabetic models (47). Several studies reported that phytochemicals found in medicinal plants have multiple beneficial effects in fighting diabetes mellitus and associated complications. For example, flavonoids, terpenoids, and phenolic acids have antidiabetic potentialities (48). Flavonoids could revive the defective beta cells in chemically induced hyperglycemic rats (49). Phenolics could act as effective antihyperglycemic agents in animal diabetic models (50). The phytochemical screening of the ethanolic extract of M. viridis showed that it contains abundant terpenoids, flavonoids, phenols, steroids, and alkaloids. Several studies have demonstrated that the phytoconstituents from different plant extracts, for instance, phenolic compounds, flavonoids, terpenoids, alkaloids, tannins, saponins, glycosides, glycolipids, etc. , have been found to have strong antidiabetic effects (51). The equeous extract of Piper longum root showed significant antihyperglycemic activity due to the presence of phytoconstituents such as alkaloids and glycosides (52). The antidiabetic effect was also found in Hibiscus sabdariffa extract, and the authors suggested that phytoconstituents such as flavonoids, triterpenes, tannins, and phenols possessed the potential to reduce blood glucose levels (41). Phytochemicals such as tannins, flavonoids, terpenoids, phenolic compounds, etc. , from the leaf of Discopodium penninervum Hoechst, were found to be effective against antihyperglycemic and antihyperlipidemic effects (37).
Therefore, phytochemicals present in ethanolic extract of M. viridis might provide practical antihyperglycemic and antihyperlipidemic effects in treated diabetic rats by several mechanisms, such as boosting up the insulin secretion by energizing the beta cells of islets of Langerhans or marked transport of glucose to peripheral tissue and regenerating the damaged beta cells as mentioned earlier (53). It was evident from the data that treatment with leaf and a combination of metformin with whole plant extract significantly improved the lipid abnormalities. The obvious lipid lowering consequence might be due to declined fatty acid and cholesterol synthesis in treated diabetic animals with plant extracts (53). Diabetes and its associated complications can be more effectively managed if the TC and TG levels are sufficiently low (54). On the contrary, the lipid-lowering effect of metformin was due to rectifying abnormal glucose metabolism and/or lowering the hepatic production of very low-density lipoprotein (55). In this study, metformin provided a significant improvement in normalizing the dyslipidemia and betterment the glycemic control in diabetic rats (Group V). Since the amalgamation of extract with metformin made additional augmentation in lowering BGL and in healing the lipid profiles excluding the TC that produced by metformin or extract alone, it might be advised that M. viridis might potentiate the hypoglycemic and hypolipidemic effect of metformin. The factual mode of action of antihyperglycemic and antihyperlipidemic outcome of M. viridis extract remains unclear and more investigations are necessary to unfold this observation.
Conclusion
The preliminary investigations suggest that M. viridis had the potential to diminish the elevated blood glucose singly and had synergistic results in lowering hyperglycemia with metformin. M. viridis extracts and amalgamation of metformin with extracts significantly mitigated the abnormalities in lipid profiles in alloxan-induced diabetic rats as opposed to untreated DC rats. These pharmacological effects could be ascribed to the existence of phytochemicals such as flavonoids, terpenoids, phenolic constituents, resins, tannins, steroids, etc. This investigation suggests that M. viridis could be a safe and valuable mono or adjuvant treatment with a reference oral hypoglycemic agent to achieve glycemic control and more effective antihyperlipidemic action. The dynamic pharmacological actions found in M. viridis extract could be combined wisely for maximum therapeutic action with minimum adverse effects. However, more investigations are essential to reveal the mechanism behind the antihyperglycemic and antihyperlipidemic effects.
Declarations
Acknowledgment
All authors are thankful to the Department of Pharmacy, Southeast University for providing all the support and equipment to conduct this research work. Special thanks to ICDDRB for providing experimental rats. Authors also thank the Bangladesh National Herbarium for identifying our experimental plants.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
All animal experiments were conducted in accordance with the approval of the Animal Ethics Committee of Southeast University, under notification letter number SEU/Pharm/CECR/122/2023.
Funding Information
Not applicable.
References
- Petersmann A, Nauck M, Mü ller-Wieland D, Kerner W, Mü ller UA, Landgraf R, et al. Definition, classification and diagnostics of diabetes mellitus. Journal of Laboratory Medicine. 2018; 42 (3): 73-9.
- Schleicher E, Gerdes C, Petersmann A, Mü ller-Wieland D, Mü ller UA, Freckmann G, et al. Definition, classification and diagnosis of diabetes mellitus. Experimental and Clinical Endocrinology & Diabetes. 2022; 130 (S 01): S1-S8.
- Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes research and clinical practice. 2022; 183: 109119.
- Banday MZ, Sameer AS, Nissar S. Pathophysiology of diabetes: An overview. Avicenna journal of medicine. 2020; 10 (04): 174-88.
- de Souza BVC, Moreira Araú jo RSdR, Silva OA, Faustino LC, Gonç alves MFB, Dos Santos ML, et al. Bauhinia forficata in the treatment of diabetes mellitus: a patent review. Expert opinion on therapeutic patents. 2018; 28 (2): 129-38.
- Jugran AK, Rawat S, Devkota HP, Bhatt ID, Rawal RS. Diabetes and plant‐ derived natural products: From ethnopharmacological approaches to their potential for modern drug discovery and development. Phytotherapy Research. 2021; 35 (1): 223-45.
- Zhou K, Pedersen HK, Dawed AY, Pearson ER. Pharmacogenomics in diabetes mellitus: insights into drug action and drug discovery. Nature Reviews Endocrinology. 2016; 12 (6): 337-46.
- Salehi B, Ata A, V. Anil Kumar N, Sharopov F, Ramí rez-Alarcó n K, Ruiz-Ortega A, et al. Antidiabetic potential of medicinal plants and their active components. Biomolecules. 2019; 9 (10): 551.
- Arumugam G, Manjula P, Paari N. A review: Anti diabetic medicinal plants used for diabetes mellitus. Journal of Acute Disease. 2013; 2 (3): 196-200.
- Singab AN, Youssef FS, Ashour ML. Medicinal plants with potential antidiabetic activity and their assessment. Med Aromat Plants. 2014; 3 (151): 2167-0412.
- Rí os JL, Francini F, Schinella GR. Natural products for the treatment of type 2 diabetes mellitus. Planta medica. 2015; 81 (12/13): 975-94.
- Liu KH, Zhu Q, Zhang JJ, Xu JF, Wang XC. Chemical composition and biological activities of the essential oil of Mentha spicata Lamiaceae. Advanced Materials Research. 2012; 524: 2269-72.
- Idm’ hand E, Msanda F, Cherifi K. Ethnopharmacological review of medicinal plants used to manage diabetes in Morocco. Clinical Phytoscience. 2020; 6: 1-32.
- Jamila F, Mostafa E. Ethnobotanical survey of medicinal plants used by people in Oriental Morocco to manage various ailments. Journal of ethnopharmacology. 2014; 154 (1): 76-87.
- Salhi N, Bouyahya A, Fettach S, Zellou A, Cherrah Y. Ethnopharmacological study of medicinal plants used in the treatment of skin burns in occidental Morocco (area of Rabat). South African journal of botany. 2019; 121: 128-42.
- Benkhnigue O, Akka FB, Salhi S, Fadli M, Douira A, Zidane L. Catalogue des plantes mé dicinales utilisé es dans le traitement du diabè te dans la ré gion d’ Al Haouz-Rhamna (Maroc). J Anim Plant Sci. 2014; 23 (1): 3539-68.
- MA M. Anti-diabetic and anti-hyperlipidemic action of aqueous ethanolic extracts of mentha spicata (leaves), plumeria alba (leaves) and nymphaea alba (flowers and rhizomes). IJBPAS. 2017; 6: 108-24.
- Bayani M, Ahmadi-Hamedani M, Javan AJ. Study of hypoglycemic, hypocholesterolemic and antioxidant activities of Iranian Mentha spicata leaves aqueous extract in diabetic rats. Iranian Journal of Pharmaceutical Research: IJPR. 2017; 16 (Suppl): 75.
- Al-Fartosi KG, Radi H, Al-Rekabi EA. Lipid profile of diabetic male rats treated with phenolic compounds of leaves extracts from mentha longifolia and mentha spicata. Int J Pharm Biol Med Sci. 2014; 3 (2): 26-31.
- El-Haoud H, Boufellous M, Berrani A, Tazougart H, Bengueddour R. Screening phytochimique d’ une plante medicinale: Mentha spicata L. American Journal of Innovative Research and Applied Sciences. 2018; 7: 226-33.
- Benedec D, Vlase L, Oniga I, Mot AC, Silaghi-Dumitrescu R, Hanganu D, et al. LC-MS analysis and antioxidant activity of phenolic compounds from two indigenous species of Mentha. Note I. Farmacia. 2013; 61 (2): 262-7.
- Lenzen S. The mechanisms of alloxan-and streptozotocin-induced diabetes. Diabetologia. 2008; 51 (2): 216-26.
- Szkudelski T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiological research. 2001; 50 (6): 537-46.
- Dubale S, Kebebe D, Zeynudin A, Abdissa N, Suleman S. Phytochemical screening and antimicrobial activity evaluation of selected medicinal plants in Ethiopia. Journal of experimental pharmacology. 2023: 51-62.
- Oloya B, Namukobe J, Ssengooba W, Afayoa M, Byamukama R. Phytochemical screening, antimycobacterial activity and acute toxicity of crude extracts of selected medicinal plant species used locally in the treatment of tuberculosis in Uganda. Tropical medicine and health. 2022; 50 (1): 16.
- Ayoola G, Coker H, Adesegun S, Adepoju-Bello A, Obaweya K, Ezennia EC, Atangbayila T. Phytochemical screening and antioxidant activities of some selected medicinal plants used for malaria therapy in Southwestern Nigeria. Tropical journal of pharmaceutical research. 2008; 7 (3): 1019-24.
- Saeed N, Khan MR, Shabbir M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC complementary and alternative medicine. 2012; 12: 1-12.
- Trivedi N, Mazumdar B, Bhatt J, Hemavathi K. Effect of shilajit on blood glucose and lipid profile in alloxan-induced diabetic rats. Indian journal of pharmacology. 2004; 36 (6): 373-6.
- Zannah S, Islam MS, Rahman AT, Asaduzzaman M, Al Bari AA, Ali Y, et al. Antidiabetic drugs in combination with hydroxychloroquine improve glycemic control in alloxan induced diabetic rats. Pharmacology & Pharmacy. 2014; 2014.
- Zehad A, Islam GJ, Rashid M, Juthy NJ, Zannah S. Antidiabetic and antihyperlipidemic activities of methanolic leaf extract of Stephania japonica in Alloxan Induced Diabetic Rats. Pharmacology & Pharmacy. 2017; 8 (04): 109.
- Islam M, Sarwar M, Rahman A, Asaduzzaman M, Ali Y, Zannah S, Rashid M. Fenofibrate potentiates the antihyperglycemic, antidyslipidemic and hepatoprotective activity of pioglitazone in alloxan-induced diabetic rats. British Journal of Pharmaceutical Research. 2016; 9 (6): 1-9.
- Ali H, Rahman AT, Islam S, Mamun A, Zannah S, Alam AK, et al. Combined therapy of pioglitazone and atorvastatin alleviate diabetes in rats more effectively than that of mono therapy. Pharmacology & Pharmacy. 2014; 2014.
- Begum MM, Rahman AT, Islam S, Asaduzzaman M, Ali H, Zannah S, et al. Simvastatin potentiates the antihyperglycemic, antidyslipidimic and antioxidative effect of glibenclamide on alloxan-induced diabetic rats. Pharmacology & Pharmacy. 2014; 5 (11): 1059.
- Krentz AJ, Bailey CJ. Oral antidiabetic agents: current role in type 2 diabetes mellitus. Drugs. 2005; 65: 385-411.
- Wiernsperger NF, Bailey CJ. The antihyperglycaemic effect of metformin: therapeutic and cellular mechanisms. Drugs. 1999; 58 (Suppl 1): 31-9.
- Hussain F, Hafeez J, Khalifa AS, Naeem M, Ali T, Eed EM. In vitro and in vivo study of inhibitory potentials of α-glucosidase and acetylcholinesterase and biochemical profiling of M. charantia in alloxan-induced diabetic rat models. American journal of translational research. 2022; 14 (6): 3824.
- Jifar WW, Debele GR, Kanfe SG, Mule CT. Evaluation of in vivo antidiabetic, antidyslipidemic and in vitro anti-oxidant activity of extract and solvent fractions of discopodium penninervum hoschst leaf in mice: normoglycemic and streptozocin-induced model. Journal of Experimental Pharmacology. 2022: 317-30.
- Kumari M, Jain S, Dave R. Babul (Acacia nilotica) A potential source of tannin and its suitability in management of type II diabetes. Nutrition & Food Science. 2014; 44 (2): 119-26.
- Kako M, Miura T, Nishiyama Y, Ichimaru M, Moriyasu M, Kato A. Hypoglycemic activity of some triterpenoid glycosides. Journal of natural products. 1997; 60 (6): 604-5.
- Ngugi MP, Kimuni N, Ngeranwa N, Orinda O, Njagi M, Maina D, et al. Antidiabetic and safety of Lantana rhodesiensis in alloxan induced diabetic rats. 2015.
- Hamadjida A, Metechie LC, Tchiengang FDT, Otto GLN, Eteme ON, Njintang NY, Mingoas JPK. Antidiabetic potential of Hibiscus sabdariffa extract in alloxan-induced diabetic rats. GSC Biological and Pharmaceutical Sciences. 2023; 23 (1): 193-203.
- Vergè s B. Lipid disorders in type 1 diabetes. Diabetes & metabolism. 2009; 35 (5): 353-60.
- Ohno T, Horio F, Tanaka S, Terada M, Namikawa T, Kitoh J. Fatty liver and hyperlipidemia in IDDM (insulin-dependent diabetes mellitus) of streptozotocin-treated shrews. Life sciences. 1999; 66 (2): 125-31.
- Goldberg IJ. Diabetic dyslipidemia: causes and consequences. The Journal of Clinical Endocrinology & Metabolism. 2001; 86 (3): 965-71.
- Rajalingam R, Srinivasan N, Govindarajulu P. Effects of alloxan induced diabetes on lipid profiles in renal cortex and medulla of mature albino rats. Indian J Exp Biol. 1993.
- Islam M, Islam MS, Zannah S, Sadik G, Rashid M. Momordica charantia (Bitter melon) in combination with metformin potentiates hypoglycemic and hypolipidemic effects in alloxan-induced diabetic rats. Bangladesh Pharm J. 2018; 21 (2): 109-17.
- Tiwari AK, Rao JM. Diabetes mellitus and multiple therapeutic approaches of phytochemicals: Present status and future prospects. Current science. 2002: 30-8.
- Zaman K. Medicinal plants with hypoglycemic activity. Journal of ethnopharmacology. 1989; 26 (1): 1-55.
- Chakravarthy B, GUPTA S, Gambhir S, Gode K. PANCREATIC BETA-CELL REGENERATION-A NOVEL ANTIDIABETIC MECHANISM OF PTEROCARPUS MARSUPIUM, ROXB. Indian journal of pharmacology. 1980; 12 (2): 123-7.
- Manickam M, Ramanathan M, Farboodniay Jahromi M, Chansouria J, Ray A. Antihyperglycemic activity of phenolics from Pterocarpus marsupium. Journal of natural products. 1997; 60 (6): 609-10.
- Pourmorad F, Hosseinimehr S, Shahabimajd N. Antioxidant activity, phenol and flavonoid contents of some selected Iranian medicinal plants. African journal of biotechnology. 2006; 5 (11).
- Nabi SA, Kasetti RB, Sirasanagandla S, Tilak TK, Kumar MVJ, Rao CA. Antidiabetic and antihyperlipidemic activity of Piper longum root aqueous extract in STZ induced diabetic rats. BMC complementary and alternative medicine. 2013; 13: 1-9.
- Hakkim FL, Girija S, Kumar RS, Jalaludeen M. Effect of aqueous and ethanol extracts of Cassia auriculata L. flowers on diabetes using alloxan induced diabetic rats. International Journal of Diabetes and Metabolism. 2007; 15 (3): 100-6.
- Luc G, Fruchart J-C. Oxidation of lipoproteins and atherosclerosis. The American journal of clinical nutrition. 1991; 53 (1): 206S-9S.
- DeFronzo RA, Goodman AM, Group MMS. Efficacy of metformin in patients with non-insulin-dependent diabetes mellitus. New England Journal of Medicine. 1995; 333 (9): 541-9.