RESEARCH ARTICLE
Phytochemical Composition and Antioxidant Properties of Dissotis erecta (Guill. & Perr.) Dandy Flowers
Sciences of Phytochemistry|Vol. 4, Issue 1, pp. 40-48 (2025)
Received
Nov 19, 2024Revised
Mar 28, 2025Accepted
Apr 12, 2025Published
Apr 25, 2025
Abstract
Introduction
Reactive oxygen/nitrogen species (ROS/RNS) such as hydroxyl radicals, nitric oxide radicals, hydrogen peroxide, superoxide anions, lipid peroxides, and various singlet oxygen molecules can damage nucleic acids, proteins, enzymes, and other biomolecules within the body, leading to loss of structure and function and potentially resulting in oxidative stress (1). These ROS are implicated in the pathogenesis of numerous degenerative and chronic diseases, including Parkinson's disease, arthritis, cancer, diabetes, aging, heart disease, and various neurodegenerative conditions (2). Synthetic antioxidants like butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), and propyl gallate (PG) can scavenge ROS. However, some of these compounds are inaccessible, unaffordable, and potentially carcinogenic, raising safety concerns. This has prompted the search for safer, more accessible, and more potent alternatives to plant-based materials.
Dissotis erecta is a medicinal plant that grows abundantly in Nigeria. Traditionally, it has been used to treat various ailments such as colds, fever, dysentery, edema, stomach discomfort, cough, and rheumatism. Research on this plant species is limited, although Soyinka et al. (3-4) reported its leaf extracts' antioxidant and antimicrobial activity. Additionally, Dissotis rotundifolia has been documented to possess antimicrobial, antioxidant, analgesic, and anti-infective activities (5-6). Chemical investigation revealed the presence of C-glycosyl flavones in D. rotundifola (7-8).
Flowers occur widely in nature and are known for their beauty, fragrance, diversity, and medicinal and culinary properties. Numerous health benefits have been linked with edible flowers, including hepatoprotective, anti-inflammatory, antidiabetic, anticancer, cardioprotective, antihemolytic activities, etc. These health benefits are attributed to their rich content of bioactive compounds such as phenolic acids, flavonoids, anthocyanins, carotenoids, tocopherol, terpene compounds, etc (9-11). To the best of our knowledge, there are no scientific studies on the antioxidant properties and phytochemical constituents of D. erecta flowers, which are also rich in phytocompounds. This study investigated preliminary phytochemical constituents, levels of phenolics and flavonoids, and antioxidant activity of the methanolic extract of D. erecta flowers was investigated using various in-vitro models.
Materials and Methods
Materials
The materials used included Dissotis erecta flower extract, beaker, stirrer, mechanical shaker, distilled water, hydrochloric acid, iron (III) chloride, potassium iodide, sodium carbonate, mercury (II) chloride, chloroform, sulphuric acid, acetic acid, trolox, 2, 2-diphenyl-1-picrylhydrazyl (DPPH), acetic anhydride, 2, 2-azinobis -3-ethylbenzothiazoline-6-sulphonic acid (ABTS), ethanol, sodium phosphate buffer, potassium ferricyanide, ferrozine, iron (II) chloride, ascorbic acid, trichloroacetic acid, methanol, sodium nitrite, sodium hydroxide, aluminum chloride, Folin- Ciocalteu reagent, gallic acid and quercetin (Analytical grade, Sigma Aldrich, USA).
Sample Collection
Fresh flowers of Dissotis erecta (Guill. & Perr. ) Dandy was collected from the wild in Uruan Local Government Area, Akwa Ibom State, Nigeria, in July 2022 and was authenticated by Prof. Margaret Bassey, a Taxonomist from the Faculty of Biological Sciences, University of Uyo, with voucher number UUYH 4573. The flowers were dried in an oven at 40 °C to constant weight, powdered with an electric blender (Silver Crest; SC1589, China), and stored in an airtight container.
Solvent Extraction
The powdered material (200 g) was soaked in methanol in a conical flask, and the flask was placed on a mechanical shaker (KJ-210BD, China) and macerated for 48 h at 26 °C. This was followed by filtration of the resulting solution and evaporation of the solvent in-vacuo (RE 52A, TOPTION Instrument Co. , China) to obtain the crude methanol (MDE) extract.
Phytochemical Analysis
The qualitative preliminary phytochemical analysis of the extract was performed using standard protocol (12-13).
Alkaloids
The extract (5 mL) in a test tube was concentrated to yield a residue. The obtained residue was dissolved by adding 1.5 mL of 2% (v/v) HCl and three drops of Meyer’s reagent (0.679 g of HgCl2 and 2.5 g of KI in 50 mL distilled water). The formation of a white precipitate indicated the presence of alkaloids.
Phenols
The extract was mixed with 2 mL of 2% FeCl3 solution and observed for the formation of blue, green, or black coloration.
Flavonoids
The extract was mixed with a few pieces of magnesium, followed by a dropwise addition of concentrated HCl. After a few minutes, the formation of a pink scarlet color indicated the presence of flavonoids.
Tannins
2 mL of 5% FeCl3 was added to 2 mL of the flower extract and observed for the formation of yellow or brown precipitates.
Saponins
The extract was mixed with 5 mL of distilled water and shaken vigorously. The formation of stable foam indicated the presence of saponins.
Terpenoids
A small amount of the flower extract was dissolved in chloroform, and an equal volume of concentrated H2SO4 was added. Reddish-brown coloration at the junction of two liquids indicated the presence of terpenoids.
Sterols
The extract (2 mL) was mixed with chloroform. Overall, 1–2 mL of acetic anhydride was added, followed by one or two drops of concentrated H2SO4 from the side of the test tube. An array of red, blue, and green colors indicated the presence of sterols.
Glycosides
2 mL of glacial acetic acid, one drop each of 5% FeCl3, and concentrated H2SO4 were added to 5 mL of the extract. The appearance of a brown ring indicated the presence of glycosides.
Evaluation of Antioxidant Activity
The antioxidant activity of the extract was determined by evaluating its DPPH, ABTS radical scavenging activities, ferric reducing, and metal chelating potentials.
DPPH Free Radical Scavenging Activity
The solvent extract was prepared by dissolving 0.01 g of the flower extract in 10 mL of methanol, and the concentration was adjusted to 10 – 200 µg/mL. Precisely, 1 mL of this extract at varying concentrations was mixed with 1 mL of 0.004% methanol solution of DPPH. The mixture was shaken vigorously and allowed to stand for 30 min at room temperature in the dark. The reduction of the DPPH radical was determined by measuring the absorption at 517 nm (Jenway 7305 Spectrophotometer, UK). The procedure was repeated for the blank and control. The radical scavenging activity was calculated using Eq. 1.
The sample concentration that provided fifty percent inhibition (IC50) was calculated from the graph plotting the inhibition percentage against the extract concentration. Trolox was used as a positive control. (14).
ABTS Assay
In the ABTS assay, Li et al. (15) method was used with minor modifications. Briefly, the ABTS radical was generated by mixing 7 mM ABTS stock solution with 2.45 mM potassium persulfate, and the mixture was left in the dark for 12–16 h at room temperature. The resulting solution was diluted with distilled water to an absorbance of 0.70 at 730 nm. 1 mL of different concentrations of the flower extract (10-200 µg/mL) was added to 4 mL of ABTS solution in labeled tubes, and the tubes were incubated for 30 min, followed by measuring the absorbance at 730 nm. Trolox was used as a positive control. The radical scavenging activity was calculated using Eq. 1. The sample concentration that provided fifty percent inhibition (IC50) was calculated from the graph plotting the inhibition percentage against the extract concentration.
Ferric Reducing Power
The ferric-reducing power was determined according to a previously reported procedure (16). The sample (10 – 200 µg/mL) in ethanol (2.5 mL) was mixed with 2.5 mL of 200 mM sodium phosphate buffer (pH 6.60) and 2.5 mL of 1% potassium ferricyanide. The resulting mixture was incubated at 50 °C for 20 min. Thereafter, 2.5 mL of 10% trichloroacetic acid (w/v) was added, and the mixture was centrifuged at 200 g for 19 min. The upper layer (5 mL) was mixed with 5 mL of deionized water and 1 mL of 0.1% ferric chloride, and the absorbance was measured at 700 nm against a blank. A higher absorbance indicated a higher reducing power. IC50 value (µg/mL) is the effective concentration at which the absorbance was 0.5 for reducing power and was obtained by interpolation. Ascorbic acid was used as positive control.
Metal Chelating Activity
The metal chelating activity was determined according to the method of Bouriche et al. (17) with some modifications. Briefly, 0.5 mL of the extract was mixed with 0.05 mL of 2 mM FeCl2 and 0.1 mL of 5 mM ferrozine. The total volume was diluted with 2 mL methanol. Then, the mixture was shaken vigorously and left at room temperature for 10 min. After the mixture had reached equilibrium, the absorbance of the solution was measured spectrophotometrically at 562 nm. The percentage inhibition rate of ferrozine – Fe2+ complex formation was calculated using Eq. 2. Acontrol = absorbance of ferrozine – Fe2+ complex, and Asample = absorbance of sample. EDTA was used as a positive control.
Determination of Total Phenolics
The extract's total phenolics concentration was determined using the Folin - Ciocalteu reagent (18). Briefly, 0.1 mL of extract was dissolved in 0.5 mL (1/10 dilution) of the Folin-Ciocalteu reagent, and 1 mL of water/methanol (1: 2) was added. The solution was mixed and incubated at room temperature for 1 min. After 1 min, 1.5 mL of 20% Na2CO3 solution was added. The final mixture was shaken and incubated for 2 h in the dark at room temperature. The absorbance of the extract was measured at 760 nm using a UV-Vis spectrophotometer. Gallic acid was employed as the standard, and the results were expressed in mg gallic acid per gram (mg GAE/g).
Determination of Total Flavonoids
A slightly modified version of the spectrophotometric method was used to determine the total flavonoid content of the extract. The extract (0.1 g) was mixed with 20 mL of 80% aqueous methanol and filtered with Whatman filter paper No. 42 to obtain a clear filtrate. A 0.5mL aliquot of this filtrate was taken in a test tube, and 3 mL of distilled water and 0.3 mL of 0.5% sodium nitrite were added. The solution was mixed and allowed to stand at room temperature for 5 min. To this solution, 0.6 mL of 10% aluminum chloride was added. After 6 min, 2 mL of 1 M sodium hydroxide solution was added. The solution was then diluted with distilled water to make the final volume up to 10 mL. The absorbance was read at 510 nm. Flavonoid content was calculated using a standard calibration curve prepared from quercetin. (19).
GC-MS Analysis
A GC (Agilent 19091S-433, Agilent Technologies, USA) interfaced with a mass spectrometer, and a capillary column (length, 30 m; thickness, 0.25 m; diameter, 0.25 mm) was employed to analyze the sample. Helium gas (99.999%) was the carrier gas at 1 mL/min, and a sample injection volume of 1 µL was at a split ratio (10: 1). The oven temperature progressed from 80 °C, with an increase of 5 °C/min, to 250 °C and maintained at 325 °C. The ion source was set at 230 °C, and the ionization voltage was at 70 eV. Interpretation of GC-MS was conducted using the National Institute of Standard Technology database. The mass spectrum of the unknown compound was compared with the spectrum of the known compound available in the NIST library (20).
Statistical Analysis
All experiments were performed in triplicate. Results were presented as mean ± SD. IC50 values were calculated by non-linear regression. Pearson’s correlation analysis between variables was performed using Statistical Package for the Social Sciences (SPSS Version 20.0, IBM Corp, USA).
Result and Discussion
Interest in finding naturally occurring antioxidants from plants that can retard the development of oxidative stress-related maladies has been on the increase in recent years. Synthetic antioxidants are limited because they are expensive, toxic, and carcinogenic (21). Therefore, identifying natural, safe, and readily accessible alternatives from plants becomes imperative. Plants are rich sources of bioactive phytoconstituents with pharmacological activities, including antioxidant capabilities, which can retard the development of oxidative stress-related maladies (2-3). Research has indicated that antioxidant activity should not be established on a single experimental model. Rather, several experimental models should be taken into consideration (3). In this study, we evaluated the content of phytochemicals and antioxidant activity of the methanolic extract of D. erecta flowers by measuring its DPPH and ABTS radical scavenging activities, metal chelating ability, and ferric-reducing potential.
Phytochemical analysis of the extract (Table 1) revealed the presence of alkaloids, flavonoids, saponins, sterols, phenols, tannins, and terpenoids, while phlobatannins and glycosides were absent. These findings agree with previous reports on the ethanolic flower extracts of Delonix elata and Tagetes minuta (22-23). Similarly, a study on the methanolic flower extract of Bromelia Iaciniosa (Bromeliaceae) identified anthraquinones, coumarins, flavonoids, and tannins as the phytocmpounds present (24). Furthermore, phytochemical analysis of Eichhornia Crassipes methanolic flower extract revealed the presence of flavonoids, saponins, and terpernoids, while alkaloids and steroids were absent. (25). The presence of alkaloids, flavonoids, tannins, phenols, sterols, and terpenoids in D. erecta flowers suggests potential therapeutic benefits, as these secondary metabolites are known for their diverse biological activities, including anti-inflammatory, antidiabetic, antioxidant, and anticancer properties (24-26).
| Test type | Extract |
|---|---|
| Alkaloids Flavonoids Saponins Sterols Tannins Phlobatannins Glycosides Phenols Terpenoids | + + + + + - - + + |
In the DPPH assay, the scavenging effect of the extract on the DPPH radical increased in a dose-dependent manner (Figure 1A), with the extract scavenging 40.2% of the DPPH radical at 10 µg/mL and 75.4% at 200 µg/mL respectively. However, this was lower than the scavenging ability of the standard Trolox, which scavenged 92.85% of the radical at 200 µg/mL.
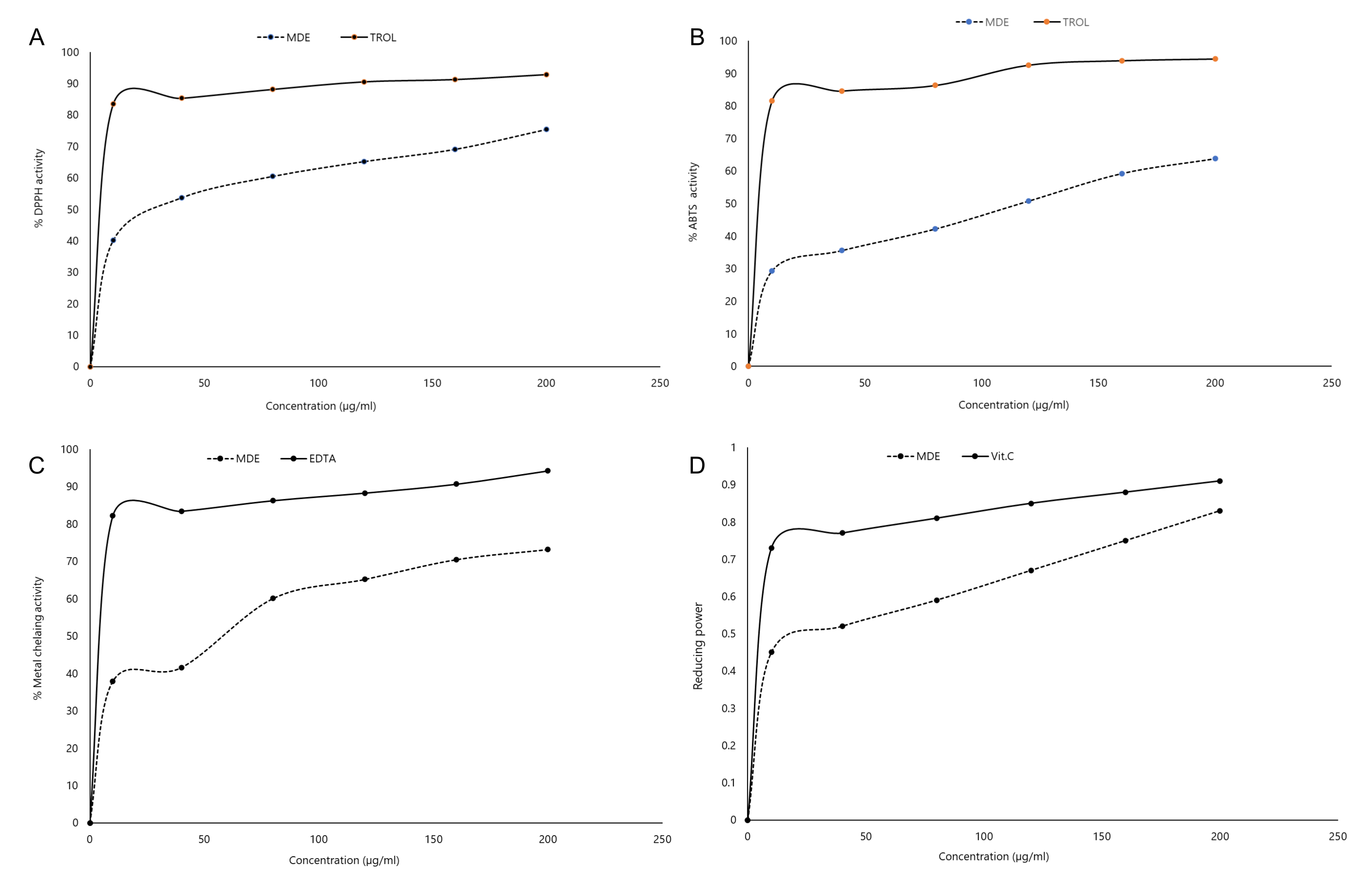
Based on this study, our extract exhibited lower DPPH scavenging activities (IC50 = 78.84 ± 0.64 µg/mL) (Table 2) than the methanolic extract of Pyrostegiavenusta flowers (IC50 = 0.026 ± 0.40 mg/mL) (21). Still, it was higher than various cultivars of Chrysanthemum morifolium (IC50 = 1.2 + 0.1 mg/mL to 7.1 ± 0.2 mg/mL) and Citrus aurantium (IC50 = 87 µg/mL) flowers (27-28). Factors such as the content of phytocompounds in flowers, climate, soil composition, and plant species may be responsible for this variation in DPPH activity (29).
| Parameter | Extract | Controls | |||
|---|---|---|---|---|---|
| Trolox*a | Trolox*b | Ascorbic acid* | EDTA* | ||
| DPPH assay* | 79.84 ± 0.64 | 13.52 ± 0.13 | |||
| ABTS assay* | 125.77 ± 2.64 | - | 7.69 ± 0.29 | ||
| Metal chelating activity* | 87.89 ± 0.91 | - | - | 11.82 ± 0.15 | |
| Reducing power* | 71.71 ± 0.55 | - | - | - | 7.34 ± 0.12 |
| Total phenolicsc | 57.47 ± 1.29 | ||||
| Total flavonoidsd | 83.52 ± 1.14 | ||||
| Note: *IC50 (μg/mL) is the effective concentration where DPPH and ABTS radical is scavenged by 50%, ferrous ion is chelated by 50% and the absorbance is 0.5 for reducing power. IC50 was obtained using the regression equation. aDPPH assay, bABTS assay; cmg GAE/g; dmg QE/g. | |||||
Similar to the DPPH assay, the ABTS radical scavenging assay is widely used to assess the radical scavenging activity of compounds and extracts. In this assay, antioxidants neutralize the ABTS radical by transferring either electrons or hydrogen atoms to the ABTS chromophore. Methanolic extract of D. erecta flowers were rapid and effective scavengers of ABTS radical in a concentration–dependent manner (Figure 1B).
Generally, our extract exhibited better ABTS scavenging abilities (IC50 = 125.77 ± 2.64 µg/mL) than flowers of Targetespatula (IC50 = 0.684 ± 0.015 TEAC mM/g) and Rosarugosa (IC50 = 1036.75 ± 8.82 µM TE/g) while higher scavenging ability was reported for flowers of Calendulaofficinalis (IC50 = 0.124 ± 0.22 TEAC mM/g), Violatricolor (IC50 = 0.454 ± 0.44 TEAC mM/g) (30-31).
The metal chelating activity of the extract was also evaluated. This is important as transition metals such as iron can catalyze hydroperoxide decomposition and Fenton-type reactions. Therefore, the ability of an extract to chelate or deactivate transition metal may be regarded as an important antioxidant mechanism (32). In this study, the chelating ability of the D. erecta flower extract increased with increasing extract concentration. At a 200 µg/mL concentration, the extract exhibited a 73.2% metal chelating ability (Figure 1C). This was, however, lower than that of the standard EDTA, with a chelating ability of 94.25% at the same concentration. Lower values were also reported for Melaleuca cajuputi flowers (33).
Like the DPPH assay, the reducing power of the extract also increased in a dose-dependent manner (Figure 1D). The result from this study indicated that the extract had a stronger reducing ability (IC50 = 71.71 µg/mL) than Cocusnucifera ethanolic flower extract (IC50 = 89.84 µg/mL), suggesting a greater ability of the extract to break the free radical chain by hydrogen donation (34).
The total phenolic and total flavonoid content of the extract showed that the extract was a rich source of phenolic compounds (57.47 ± 1.29 mg GAE/g and 83.52 ± 1.14 mg QE/g, respectively). Phenolics are powerful antioxidants whose activity is structure-related. They scavenge reactive species, chelate transition metals, and initiate deleterious free radical reactions (35). The phenolic content in this work was higher than reports for edible flower extracts from Portugal (36).
Overall, the extract exhibited strong antioxidant activity (IC50 < 150 µg/mL) in the various in-vitro models and could work through a variety of mechanisms, including scavenging radicals by either hydrogen donation or transfer of electro, sequestering transition metals, decomposing hydrogen peroxides, quenching active pro-oxidants and enhancing endogenous antioxidant defense (37-38).
The correlation between the antioxidant assays and phenolics was also evaluated, as depicted in Table 3. Total phenolics showed a good correlation with DPPH activity (R2 = 0.85), ABTS activity (R2 = 0.71), metal chelating capacity (R2 = 0.98), and reducing power (R2 = 0.95). A similar result was observed for total flavonoids. This result is in agreement with reports that antioxidant activity is related to the content of phenolics. Similarly, there was a positive correlation between total phenolics and total flavonoids, suggesting phenolics' importance in the extract's observed antioxidant activity (32, 39).
| - | DPPH | ABTS | MC | RP | TP | TF |
|---|---|---|---|---|---|---|
| DPPH | 1 | |||||
| ABTS | 0.79 | 1 | ||||
| MC | 0.98 | 0.77 | 1 | |||
| RP | 0.95 | 0.83 | 0.89 | 1 | ||
| TP | 0.85 | 0.71 | 0.92 | 0.68 | 1 | |
| TF | 0.96 | 0.78 | 0.91 | 0.99 | 0.71 | 1 |
GC-MS analysis (Figure 2) revealed the presence of a-terpineol, nerolidol, campesterol, and neocurdione as the major components. β-sitosterol, kaempferol, aminocaproic acid, piperidine, 1, 1’-thiobis, α–panasinsen, 1-(piperidin-1-yl) tetradecane-1-one and quinoline-2 (1H)-one, 1, 1-di (hydroxymethyl)- were also identified in trace amounts. α-terpineol is a volatile monoterpenoid alcohol found in several aromatic plants with wide applications in medicine and industry.
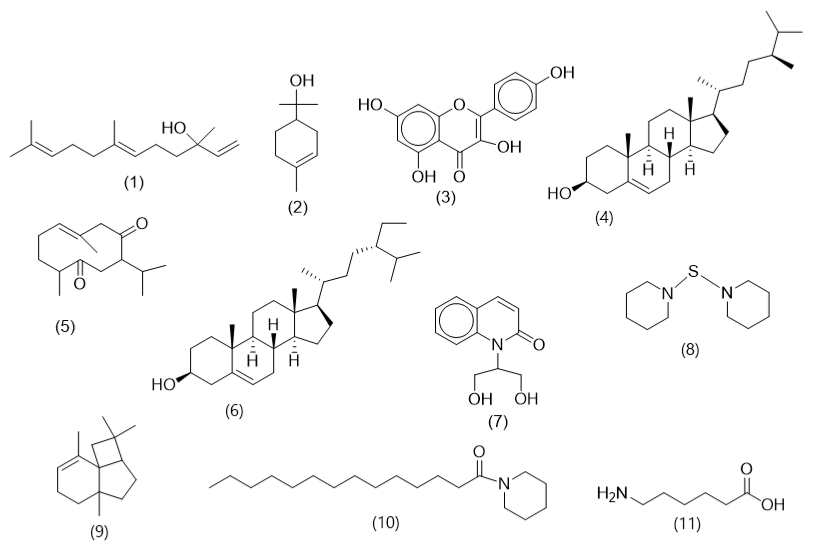
Notable biological activities exhibited by this compound include antioxidant, anticancer, anticonvulsant, antiulcer, antihypertensive, antinociceptive, anti-bronchitis, insecticidal, and skin penetrating enhancing activities (40). Nerolidol, kaempferol, β-sitosterol, and piperidine, 1, 1’ thiobis have shown numerous pharmacological activities, including antioxidant, antitumor, anti-inflammatory, cardioprotective, antidiabetic, anti-arthritis, antibacterial, and antidepressant activities (41-43). These phytocompounds in the D. erecta flower extract may be responsible for the observed antioxidant properties.
Conclusion
The methanolic extract of D. erecta flowers demonstrated potent antioxidant activity across multiple in-vitro models and was rich in phytocompounds. The GC-MS analysis identified α-terpineol, nerolidol, campesterol, kaempferol, β-sitosterol, and neocurdione as the key compounds responsible for the extract’s antioxidant properties. This study indicates that flowers could be an excellent source of alternative and sustainable plant-based antioxidants, potentially playing a crucial role in managing diseases related to oxidative stress. Additionally, the findings highlight the future potential of D. erecta in pharmaceutical, nutraceutical, and cosmetics applications, encouraging further research into its bioactive composition and therapeutic benefits.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
Not applicable.
Funding Information
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
References
- González-Palma I, Escalona-Buendía HB, Ponce-Alquicira E, Téllez-Téllez M, Gupta VK, Díaz-Godínez G, Soriano-Santos J. (2016). Evaluation of the Antioxidant Activity of Aqueous and Methanol Extracts of Pleurotus ostreatus in Different Growth Stages. Front. Microbiol. 2016; 7:1099.
- Jafri SAA, Khalid ZM, Khan MZ, Jogezai N. Evaluation of phytochemical and antioxidant potential of various extracts from traditionally used medicinal plants of Pakistan. Open Chem. 2022; 20: 1337–1356.
- Guchu BM, Machocho AK, Mwihia SK, Ngug MP. In Vitro Antioxidant Activities of Methanolic Extracts of Caesalpinia volkensii Harms., Vernonia lasiopus O. Hoffm., and Acacia hockii De Wild. Evidence-Based Complement. Altern. Med. 2020; 3586268, 1-10.
- Soyinka JO, Oguntade TO, Onawunmi GO, Idowu TO, Ogundaini AO. Antioxidant and antimicrobial constituents of Dissotis erecta and Dissotis rotundifolia. Nig. J. Pharm. Res. 2008; 7(1):76-82.
- Usanga VU, Agbo NL, Kalu ME, Ude UA, Azi, SO. Microbial contamination, antimicrobial activities of Dissotis rotundifolia leaf: A common ethnomedicine for ocular diseases. Nig. J. Pharm. Res. 2023; 19(1):71-77.
- Enin GN, Adegoke AA, Ita BN, Udosen CI, Inyang VF, Onuaha EC, Antia BS. In vitro antioxidant, mineral analysis, and antimicrobial activities of extracts and fractions from the aerial parts of Heterotis rotundifolia (Sm.) Jacq. Fel. Trop. J. Nat. Prod. Res. 2024; 8(8):8202-8211.
- Adinortey MB, Ansah C, Adinortey CA, McGiboney J, Nyarko A. In vitro H+/K+-ATPase Inhibition, Antiradical Effects of a Flavonoid-rich Fraction of Dissotis rotundifolia, and In silico PASS Prediction of its Isolated Compounds. J. Nat. Sci. Biol. Med. 2018; Jan-Jun; 9(1):47–53.
- Raith G, Toure A, Nianga M, Wolfender JL, Hostettman K. Characterisation of C-glycosyl flavones from Dissotis rotundifolia by liquid chromatography-UV diode array detection-tandem mass spectrometry. Chromatograph. 1995; 41(516): 332 - 342
- Antunes MC, Araujo S, Matos C, Correia E. Qual. Assur. Safety Crops. Foods. 2019; 11 (5): 471-478.
- Fernandes L, Casal S, Pereira JA, Saraiva JA, Ramalho E. Edible flowers: A review of the nutritional, antioxidant, antimicrobial properties and effects on human health. J. Food Compos. Anal. 2017; 60: 38-50.
- Kandylis P. Phytochemicals and antioxidant properties of edible flowers. Appl. Sci. 2022; 12(19): 9937-9948.
- Mallikharjuna PB, Rajanna LN, Seetharam YN, Sharanabasappa GK. Phytochemical Studies of Strychnos Potatorum L.F.- a Medicinal Plant. J. Chem. 2007; 4(4): 510–518.
- Harborne JB. In Phytochemical Methods. Chapman Hall: London, 1998.
- Sharma S, Awasthi L, Kumari P. Evaluating the Phytochemical Composition and Antioxidant Activity of Leaves of Different Rose Varieties. Int. J. Plant Biol. 2023; 14:1051–1063.
- Li P, Huo L, Su W, Lu R, Deng C, Liu L, Deng Y, Guo N, Lu C, He C. Free radical-scavenging capacity, antioxidant activity and phenolic content of Pouzolzia zeylanica. J. Serbian Chem. Soc. 2011; 76 (5): 709-717.
- Enin GN, Ita, BN, Jumbo B, James MU, Joseph SE, Antia BS, Thomas PS, Okokon JE. In vitro Antioxidant and Biological Activities of Extract and Fractions from Telfairia occidentalis Stems. South Asia Res. J. Nat. Prod. 2024; 7(2): 102-122.
- Bouriche H, Meziti H, Senator A, Arnhold J. Anti-inflammatory, free radical-scavenging and metal-chelating activities of Malva parviflora. Pharm. Biol. 2011; 49(9): 942-946.
- Gul H, Bhakshu I, Ahmed F, Qureshi I, Ghazi I. Evaluation of Abelmoschus moschatus extracts for antioxidant, free radical scavenging, antimicrobial and antiproliferative activities using in-vitro assays. BMC Complement. Altern. Med. 2011; 11: 64-72.
- Subhasree B, Baskar R, Keerthana RL, Susan RL, Rajasekran P. Evaluation of antioxidant potential in selected green leafy vegetables. Food Chem. 2009; 115: 1213 -1220.
- Kadhim MJ, Mohammed GJ, Hussein H. Analysis of bioactive metabolites from Candida albicans using (GC-MS) and evaluation of antibacterial activity. Int. J. Pharm. Clin. Res. 2016; 8(7):655-670.
- Roy P, Amdekar S, Kumar A, Singh V. Preliminary study of the antioxidant properties of flowers and roots of Pyrostegia venusta (Ker Gawl) Miers. BMC Complement. Altern. Med. 2011; 11:69-77.
- Okpondo FA, K’Owino JO, Chepkwony SC, Kosgei VJ. Evaluation of phytochemical composition of extractives and proximate analysis of Tagetes minuta powdered leaves and flowers. Eur. J. Appl. Sci. 2023;11(4):217-237.
- Amabye TG, Bezabh AM, Mekonen F. Phytochemical constituents and antioxidant activity of Delonix elata L. flower extract. J. Anal, Pharm. Res. 2016; 2(1):1-5.
- Goncalves de Oliveira-Junior R, Ferraz CAA, Souza GR, Guimares AL, Paula de Oliveira A, Gomes de Lima SR, Rolim LA, Rolim=Neto PJ, Almeida JR. Phytochemical analysis and evaluation of antioxidant and photoprotective activities of extracts from flowers of Bromelia iaciniosa (Bromeliaceae). Biotech. Biotech. Equip. 2017; 31(3):600-605.
- Baehaki A, Lestari SD, Agustina W, Putri SD. Phytochemical analysis and antioxidant activity of water Hyacinta flowers (Eichhorinia crassipes) extract. Pharmacog. J. 2022; 14(4) Suppl: 955-957.
- Parani K. Determination of physicochemical composition of leaves and flowers of Cassia auriculata. Eur. J. Med. Plants. 2024; 35(5): 13-20.
- Doan TTM, Tran GH, Nguyen TK, Lim JH, Lee S. Antioxidant activity of different cultivars of Chrysanthemum morifolium and quantitative analysis of phenolic compounds by HPLC/UV. Appl. Biol. Chem. 2024; 67:17-30.
- Prabawati NB, Oktavirina V, Palma M, Setyaningsih W. Edible Flowers: Antioxidant Compounds and Their Functional Properties. Hortic. 2021, 7:66-74.
- Kumar S, Yadav, M, Yadav A, Yadav AP. Impact of spatial and climate conditions on the phytochemical diversity and in vitro antioxidant activity of Indian Aloe Vera (L.) Burm f. South. Afr. J. Bot. 2017; 111: 50-59.
- Araujo S, Matos C, Correia E, Antunes MC. Evaluation of phytochemicals content, antioxidant activity and mineral composition of selected edible flowers. Qual. Assur. Safety. Crops. Foods, 2019; 11 (5): 471-478.
- Zheng J, Yu X, Maninder M, Xu B. Total phenolics and antioxidants profiles of commonly consumed edible flowers in China. Int. J. Food Prop. 2018; 21(1):1524-1540.
- Sowndhararajan K, Kang SC. Free radical scavenging activity from different extract of leaves of Bauhinia vahliii Wight and Arn. Saudi J. Biol. Sci. 2013; 20:319-325.
- Al-Ald NM, Nor ZM, Mansor M, Azhar F, Hasan MS, Kassim M. Antioxidant, antibacterial activity and phytochemical characterization of Melaleuca cajuputi extract. BMC Complement. Altern. Med. 2015; 15:385.
- Kumar V, Lemos M, Sharma M, Shiram V. Antioxidant and DNA damage protecting activities of Eulopia nuda Lindl. Free Rad. Antiox. 2013; 3:55-60.
- Fresco O, Borges F, Diniz C, Marques MPM. New insights on the anticancer properties of dietary polyphenols. Med. Res. Rev. 2006; 26:747-766.
- Gonçalves F, Gonçalves JC, Ferrão AC, Correia P, Guiné RPF. Evaluation of phenolic compounds and antioxidant activity in some edible flowers. Open Agric. 2020; 5: 857–870.
- Kholifah E, Nurazizah D, Noviyanto F. Antioxidant activity and vitamin C concentration analysis of Gandaria (Bouae macophylla Griff) ethanol extract using UV-Vis spectrophotometry. J. Fundam. Appl. Pharm. Sci. 2023; 3(2): 54-63.
- Flieger J, Flieger W, Baj J, Maciejewski R. Antioxidants: Classification, Natural Sources, Activity/ Capacity Measurements, and Usefulness for the Synthesis of Nanoparticles. Mater. 2021; 15(15): 4135.
- Jemli ME, Kamal R, Marmouzi I, Zerrouki A, Cherrah Y, Alaoui K. Radical Scavenging activity and Ferric reducing abilities of Juniperus thurifera (L.), Juniperus oxycedrus (l.), Juniperus phoenicea (L.) and Tetraclinis articulata (L.). Adv. Pharm. Sci. 2016; 6392656: 1-6.
- Khaleel C, Tabanca N, Buchbauer G. α- terpineol, a natural monoterpene: A review of its biological activities. Open Chem. 2018; 16(1): 349-361.
- Chen M, Fitzgerald P, Singh SB, O’Neill EA, Schwartz CD, Thompson CM, O’Keefe SJ, Zaller DM, Doherty JB. Synthesis and biological activity of quinolinone and dihydroquinolinone p38 MAP kinase inhibitors. Bioorg. Med. Chem. Lett. 2008; 18(6): 2222-2226.
- Babu S, Jayaraman S. An update on β-sitosterol: A potential herbal nutraceutical for diabetic management. Biomed. Pharmacother. 2020; 131: 110702.
- Bangar SP, Chaudhary V, Sharma N, Bansal V, Ozogul F, Lorenzo JM. Kaempferol: A flavonoid with wider biological activities and their applications. Crit. Rev. Food Sci. Nutr. 2023; 63(28):9580-9604.