RESEARCH ARTICLE
In Vitro and In Silico Evaluation of Brugmansia suaveolens' Ability to Treat Asthma
Sciences of Phytochemistry|Vol. 2, Issue 1, pp. 121-130 (2023)
Received
Apr 10, 2023Revised
May 30, 2023Accepted
Jun 1, 2023Published
Jun 1, 2023
Abstract
Introduction
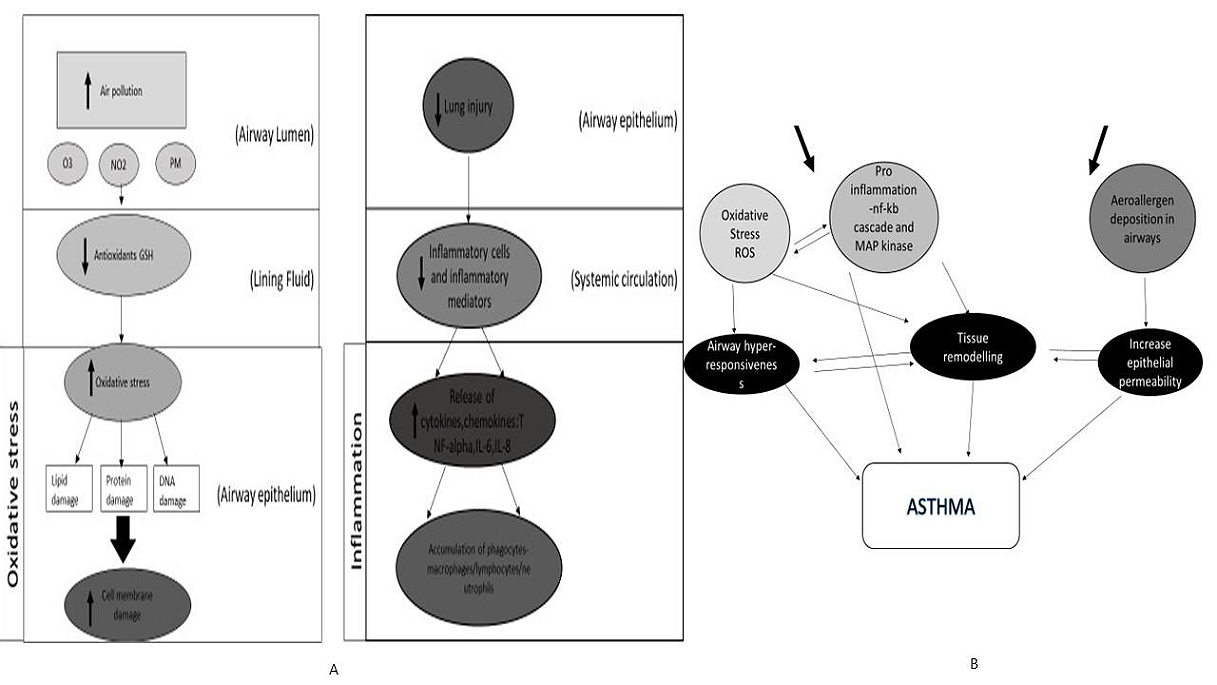
Chronic respiratory disease is one of the main causes of mortality and morbidity worldwide. People of all ages are susceptible to asthma, a common chronic inflammatory disease of the airways. With more than 300 million people currently suffering from asthma worldwide, the burden of asthma is enormous (1). It has long been believed that asthma is the classic T helper type 2 (TH2) illness of the airways due to the high eosinophil count in the airways of persons with moderate asthma and results from mouse models. It is now known that some eosinophilic inflammation and some neutrophilic inflammation in asthma are controlled by type 2 innate lymphoid cells (ILC2 cells) acting in performance with basophils (2). The link between the main causes of asthma, airway inflammation, and oxidative stress in asthma is depicted in Figure 1. Exposure to external agents, such as allergens found indoors and outdoors, air pollutants like O3, NO2, PM (particulate matter), and respiratory tract infections, as seen in Figure 1A, are the most frequent causes of these conditions. Oxidative stress is the phrase used to describe the imbalance between oxidants, such as reactive oxygen species (ROS) and reactive nitrogen species (RNS), and antioxidants in favour of oxidants after exposure. This illness could be categorized as a heterogeneous disorder. The primary environmental factors influencing the physiopathology of asthma are sensitization to dust, pollen, and diet. T lymphocytes and eosinophils release pro-inflammatory cytokines such as tumor necrosis factor- β (TNF-β), interleukins IL-2, IL-3, IL-4, IL-5, prostaglandins, histamine, and leukotrienes in response to antigens recognized by mast cells covered by IgE antibodies (Figure 1B).
In silico studies have developed into a substantial and straight forward tool for analyzing diverse chemical compounds against numerous targets, with many advantages, including a significant decrease in the cost and time of drug discovery as well as the use of laboratory animals (3). There are a lot of pharmacologically active chemicals found in medicinal plants. Pharmaceutical components that can be employed in the creation of new drugs can be found in abundance in medicinal plants (4, 5). Beyond that, these plants have a big impact on how human societies have evolved all around the world. Anything about the biological function of possible drug candidates, including sequence-based properties and interactions with bodily structures, is considered a part of bioinformatics methods for drug development (6).
Bioinformatics methods for drug discovery include anything related to the biological function of potential drug candidates, including sequence-based characteristics, interactions with body structures (metabolites, proteins, cells, tissues, etc. ), pathway perturbations, and toxicity, among others. Multi-omics and high-throughput sequencing are also major areas within bioinformatics (7). In the case of natural products; researchers make use of an entire range of techniques related to the organisms that produce the compounds. Drug discovery enabled by bioinformatics and computational methods is an increasing shift toward data-driven drug discovery. Traditionally, drug discovery has been performed as follows: scientists first find a target structure in the human body related to a disease or illness, followed by screening for “lead” compounds that show an affinity for the target. Although natural drug discovery has always been a hallmark activity in pharmacology, pharmaceutical researchers have only applied these chemo-informatics techniques to natural products recently (8).
This plant has been used in traditional medicine throughout cultures as a muscle relaxant, nematicide, aphrodisiac, hallucinogen, and treatment for inflammation, rheumatism, asthma, and allergies (10). Different chemical substances have been detected in the plant's flowers, fruits, stems, and roots, including alkaloids, volatile chemicals mostly terpenes, coumarins, flavonoids, steroids, and hydrocarbons. This medicinal species has been the subject of chemical investigations since 1996 (11). These investigations were the first qualitative research on particular classes of substances, which included amines, sugars, alkaloids, phenolic compounds, flavonoids, steroids, terpenoids, tannins, anthraquinone glycosides, saponins, and triterpenes (12). The ethanolic extract of the flowers was also used to quantify the alkaloids (5.903 ± 0.01333 mg/g), phenolic compounds (3.435 ± 0.0110 mg/g), and flavonoids (9.945 ± 0.0256 mg/g) (13). The concentrations of these substances can alter, much like the volatile chemical profiles of living flowers, which are constantly changing due to both internal (genetic) and environmental (light, temperature, and water stress) variables. The loss of volatile chemicals and rapid deterioration are problems with cut flowers (14).
In the present study, our attention is on the anti-inflammatory process and the application of herbal plants that have anti-inflammatory properties. This study looks at the most recent information available in various search engines regarding the distribution, phytochemistry, pharmacology, and toxicology of B. suaveolens (Solanaceae) using chemical compounds or extracts at this time (9). To detect structural activity, we employed bioinformatics tools like PASS Online, Molinspiration, SEA, SWISS, and SLAP data. Data from these three phytoconstituents were compared to Phytoconstituents from B. sauveolens (15, 16). We evaluated B. suaveolens fractions and their in vitro anti-inflammatory assay. We intended to conduct the anti-oxidative and the anti-inflammatory assays for all fractions, however, Covid-19 prevented us from doing so (17).
Experimental Section
Materials and Methods
Materials for In Silico Approach With the Anti-Inflammatory Phytoconstituents
Databases: We used biological databases such as PubChem, PDB (Protein Data Bank), and NCBI for a literature search for anti-inflammatory, Phytoconstituents in Indian medicinal plants used in asthma.
Software: Online tools were used for various in silico predictions. PASS software was used to predict the biological activities of Phytoconstituents (9). Molinspiration software was used to predict the bioactivity score of the compounds against human molecular targets/ receptors such as GPCRs, ion channels, kinases, nuclear receptors, proteases, and enzymes (18-20). SEA is a tool for conducting structure-based fingerprint similarity searches and returns a ranked list of predictions, and comparative information on ligands known to share those targets (20). SWISS target returns a list of targets, with links to the relevant ChEMBL and Uniprot records, ranked by the pseudo-probability of ligand–targeted interaction (7). In SLAP, we sought to use such semantic methods to integrate and annotate the data about drug-target interaction (19).

Materials for In Vitro Approach
Collection of Plant Material
Plants were collected from several areas in Kolkata during October and November.
Chemicals and Instruments
Methanol, petroleum ether, chloroform, ethyl acetate, n-butanol, sodium chloride, and aspirin were used. Centrifuge, electronic balance, and UV-VIS double beam spectrophotometer were used.
Methods for In Silico Approach
Retrieval of Phytochemicals and Target Protein
Firstly, we chose an anti-asthmatic formulation used in the market. Then we identified herbs in these formulations and categorized them based on their anti-inflammatory activity. The anti-inflammatory activities of herbs were collected in the literature analysis. Literature searches were based on experimentally determined properties. We chose phytoconstituents such as cinnamaldehyde, piperine, and linolenic acid for further studies and standardization of the chosen parameters.
Protein Data Bank (PDB) archive is the single worldwide repository of information about the 3D structures of large biological molecules. 3D structures were downloaded in SDF format and canonical smiles were downloaded. 3D structures in SDF format were converted to PDB format.
PASS (Prediction of Activity Spectra for Substances)
PASS software was utilized to predict the interactions of phytochemicals with selected targets (see Table 1). PASS can be accessed from the site www. way2drug. com/PASSOnline. PASS tool interpreted the biological active spectra using the 2D structure of molecules. PASS activity selected anti-inflammatory activity which has Pa (probability "to be active") greater than Pi (probability "to be inactive") and Pa greater than 0.5.
Molinspiration, SEA, SWISS, and SLAP
The compounds were analyzed using the Molinspiration web tool based on Lipinski’s Rule of Five (www. molinspiration. com). Along with this, the bioactivity score was also predicted. A similarity ensemble approach (SEA) was used to study the potential targets of the test compounds (https://sea. bkslab. org/). Human targets related to inflammation were identified. Results showing Tanimoto coefficients (Tc) greater than 0.6 were selected. SWISS Target was used to study the potential human targets and a probability not to be less than 0.5 was used to collect significant data (http://www. swisstargetprediction. ch/). SLAP was also used to predict the targets of the test compounds and the benchmark p-value was set at 0.05 (http://cheminfov. informatics. indiana. edu: 8080/slap/).

Methods for Extraction, Fractionation, and In Vitro Approach for the Anti-Inflammatory Assay
The leaves of B. sauveolens were subjected to shade drying. The dried plant material was further crushed to powder and stored in an airtight container for further analysis. The plant materials were first extracted with petroleum ether for defatting followed by methanol in the Soxhlet extractor. Further, the methanolic extract of the plant was fractionated with chloroform and ethyl acetate based on their polarity. The extracts and fractions were filtered and the solvent was evaporated to dryness over the hot water bath. All the dried extracts were kept in well-closed containers under refrigeration.

In Vitro Anti-Inflammatory Activity Through Membrane Stabilization Assay
Preparation of Red Blood cell (RBC) suspension: The Blood was collected from a healthy rat to the experiment and transferred to the centrifuge tubes. The tubes were centrifuged at 3000 rpm for 10 min and then washed three times with an equal volume of normal saline. The volume of blood was measured and reconstituted as a 10% v/v suspension with normal saline.
Heat-Induced Hemolysis Assay: The reaction mixture (2 mL) consisted of a 1 mL test sample at different concentrations and 1 mL of a 10% RBC suspension. For the control test tube, only saline was added instead of the test sample to ensure an accurate comparison. Aspirin was used as the standard drug to evaluate the protective effect against hemolysis. All centrifuged tubes containing the reaction mixture were incubated in a water bath at 56 °C for 30 minutes to induce hemolysis. After incubation, the tubes were immediately cooled under running tap water to halt the reaction. The samples were then centrifuged at 2500 rpm for 5 minutes to separate the supernatant. The absorbance of the supernatant was measured at 560 nm to assess the extent of hemolysis (22). This method helps determine the membrane-stabilizing properties of the test compounds and their potential anti-inflammatory effects.
Results and Discussion
PASS Online: The pharmacological effects of a compound and its communication with biological entities were predicted by PASS online by uploading the SMILES string of natural compounds. PASS prediction results showed that the highest Pa value than the Pi value comes off for anti-inflammatory activity and hence selected compounds. However, all compounds have shown a significant Pa value as compared to the Pi value.
We selected data with a Pa value greater than 0.5, as it indicates strong biological activity compared to Pi. The Pa value represents the probability of a compound exhibiting a particular activity, with higher values suggesting better potential effectiveness. The concept of the 'biological activity spectrum' provides a list of activity names that reflect a compound’s interaction with biological entities. If Pa < 0.5, the compound is either unlikely to exhibit the activity or has a lower probability of doing so. In Table 1, cinnamaldehyde and linolenic acid demonstrated promising anti-inflammatory activity. Cinnamaldehyde had a highest Pa of 0.596 and Pi of 0.033, while linolenic acid had a highest Pa of 0.804 and Pi of 0.006.
| Name of the compound | Pa | Pi | Activity |
|---|---|---|---|
| Cinnamaldehyde | 0, 596 | 0, 033 | Anti-inflammatory |
| 0, 494 | 0, 008 | Anti-inflammatory, intestinal | |
| 0, 308 | 0, 067 | Anti-inflammatory, ophthalmic | |
| 0, 220 | 0, 084 | Non-steroidal anti-inflammatory agent | |
| Piperine | 0, 314 | 0, 060 | Anti-inflammatory, intestinal |
| 0, 344 | 0, 127 | Anti-inflammatory | |
| 0, 238 | 0, 207 | Anti-inflammatory, ophthalmic | |
| Linolenic acid | 0, 804 | 0, 006 | Anti-inflammatory |
| 0, 591 | 0, 004 | Anti-inflammatory, intestinal | |
| 0, 443 | 0, 017 | Non-steroidal anti-inflammatory agent | |
| 0, 373 | 0, 017 | Anti-inflammatory, ophthalmic |
| Drug/ compound | GPCRligand | Ion channel modulator | Kinase inhibitor | Nuclearreceptor ligand | Protease inhibitor | Enzyme inhibitor |
|---|---|---|---|---|---|---|
| Piperine | 0.15 | -0.18 | -0.13 | -0.13 | -0.10 | 0.04 |
| Linolenic acid | 0.33 | 0.23 | -0.19 | 0.35 | 0.13 | 0.42 |
| Cinnamaldehyde | -1.09 | -0.39 | -1.24 | -0.96 | -0.76 | -0.46 |
| Imiquimod | 0.27 | 0.21 | 0.60 | -0.77 | -0.29 | 0.48 |
| Acetyl salicylic acid | -0.76 | -0.32 | -1.06 | -0.44 | -0.82 | -0.28 |
| Target | Swiss target | SEA | SLAP | |||
|---|---|---|---|---|---|---|
| Probability | p-value | Max TC | p-value | Score | Type | |
| MAOB | 1.0 | 1.229e-05 | 1.00 | - | - | - |
| TRPV1 | - | 4.11e-07 | 1.00 | - | - | - |
| CYP3A4 | - | 4.982e-07 | 1.00 | - | - | - |
| CYP1A2 | - | 0.0002663 | 1.00 | - | - | - |
| MMP1 | - | - | - | 0.003 | 530.34 | Predicted |
| MMP9 | - | - | - | 0.004 | 404.92 | Approved expression |
| MMP3 | - | - | - | 0.0027 | 576.78 | Predicted |
| MMP8 | - | - | - | 0.0033 | 478.3 | Predicted |
| MMP13 | - | - | - | 0.0034 | 473.05 | Predicted |
| MMP2 | - | - | - | 0.0039 | 412.66 | Approved expression |
| MMP14 | - | - | - | 0.0043 | 379.97 | Predicted |
| ADAM17 | - | - | - | 0.0046 | 357.71 | Predicted |
| MMP7 | - | - | - | 0.0059 | 286.68 | Predicted |
| MMP11 | - | - | - | 0.0079 | 220.89 | Predicted |
| MMP10 | - | - | - | 0.0099 | 181.1 | Predicted |
| TNF | - | - | - | 0.0197 | 96.75 | Approved expression |
| IL1B | - | - | - | 0.0266 | 73.58 | Approved expression |
| IL6 | - | - | - | 0.0383 | 52.58 | Approved expression |
| - | - | - | - | 0.003 | 530.34 | Predicted |
The molecular properties of the compounds were calculated using Molinspiration chemo-informatics software and are presented in Table 2 and Table 3. The Molinspiration tool retrieves results using the Canonical SMILIES in PubChem and the 3D structure of the compounds. A molecule having a bioactivity score of more than 0.00 is most likely to exhibit considerable biological activities, while values −5.0 to 0.00 are expected to be moderately active and if the score was less than −5.0 it was presumed to be inactive. The results reveal that the physiological actions of different compounds might involve multiple mechanisms and could be due to the interactions with GPCR ligands, nuclear receptor ligands, and inhibit protease and other enzymes. The identified compounds showed better bioactivity scores than Imiquimod and Acetylsalicylic acid for all drug targets.
Therefore, numbers of potential compounds and active human targets with their corresponding information of probability, p-VALUE, Max TC, p-value and score, and type information were retrieved, and are given in Table 3. Swiss targets of a molecule were predicted by identifying proteins with known ligands that are highly similar to the query molecule. In piperine target MAO-B has probability 1. As target prediction accuracy is affected by the molecule size probabilities were computed separately for each ligand size. In SEA data similarities were calculated for ligands and later successfully to drug target prediction. SEA calculates a raw score by summing up the Tanimoto similarity of the chemical structure. We found SEA and SLAP data on cinnamaldehyde and linolenic acid.
| Name of compound | Target | Swiss target | SEA | |
|---|---|---|---|---|
| Probability | p-VALUE | Max TC | ||
| Cinnamaldehyde | TRPA1 | 1.0 | 2.819e-05 | 1.00 |
| Linolenic acid | PPARG | 0.739591175051 | 4.296e-09 | 1.00 |
| PPARA | 0.739591175051 | 3.843e-08 | 1.00 | |
| PPARD | 0.739591175051 | 3.052e-06 | 0.85 | |
| FABP4 | 0.714850037542 | 1.712e-07 | 0.79 | |
| FFAR1 | 0.706805409756 | 0.000163 | 1.00 | |
| PTGS1 | 0.665850737135 | 0.104 | 1.00 | |
| FABP3 | 0.665850737135 | 6.282e-27 | 1.00 | |
| OXER1 | - | 3.091e-62 | 1.00 | |
| FFAR4 | - | 6.219e-11 | 1.00 | |
In Silico Comparisons With Brugmansia Sauveolens for Anti-Inflammatory Properties
We compared the above data with B. suaveolens plant phytoconstituents, including hyoscine, norhyoscine, atropine, and apohyoscine. The Pa value represents the likelihood of a compound exhibiting biological activity, with values greater than 0.5 indicating strong potential. When analyzing Table 5, we observed that linolenic acid and cinnamaldehyde had Pa values greater than 0.5, suggesting good activity. In contrast, hyoscine, norhyoscine, atropine, and apohyoscine had Pa values below 0.5, indicating a lower probability of significant activity. While the plant phytoconstituents may still exhibit some biological effects in experimental conditions, their likelihood of demonstrating strong activity is comparatively lower. Further studies may be necessary to confirm their potential efficacy.
The molecular properties of the compound were calculated using Molinspiration Cheminformatics software and are presented in Table 6. The Molinspiration tool retrieves results using the Canonical SMILIES in PubChem and the 3D structure of the compounds.
A molecule having a bioactivity score of more than 0.00 is most expected to show significant biological activities, while values −5.0 to 0.00 are expected to be moderately active and if the score was less than −5.0 it was presumed to be inactive. In Table 6, B. sauveolens chemical constituents showed a good value of GPCR ligand, Ion channel modulator, Kinase inhibitor nuclear receptor ligand, Protease inhibitor, and Enzyme inhibitor. The identified compounds showed better bioactivity scores than Imiquimod and Acetylsalicylic acid for all drug targets.
| Name of compounds | Pa | Pi | Activity |
|---|---|---|---|
| Hyoscine | 0, 338 | 0, 130 | Anti-inflammatory |
| 0, 292 | 0, 092 | Anti-inflammatory, ophthalmic | |
| Norhyoscine | 0, 300 | 0, 079 | Anti-inflammatory, ophthalmic |
| 0, 289 | 0, 171 | Anti-inflammatory | |
| 0, 200 | 0, 189 | Anti-inflammatory, intestinal | |
| Atropine | 0, 338 | 0, 037 | Anti-inflammatory, ophthalmic |
| 0, 379 | 0, 107 | Anti-inflammatory | |
| 0, 202 | 0, 099 | Non-steroidal, anti-inflammatory agent | |
| 0, 204 | 0, 183 | Anti-inflammatory, intestinal | |
| Norhyoscine | 0, 300 | 0, 079 | Anti-inflammatory, ophthalmic |
| 0, 289 | 0, 171 | Anti-inflammatory | |
| 0, 200 | 0, 189 | Anti-inflammatory, intestinal |
| Drug/compound | GPCRligand | Ion channel modulator | Kinase inhibitor | Nuclear receptor ligand | Protease inhibitor | Enzyme inhibitor |
|---|---|---|---|---|---|---|
| Hyoscine | 0.58 | 0.23 | 0.06 | 0.11 | 0.28 | 0.35 |
| Norhyoscine | 0.51 | 0.28 | 0.06 | 0.16 | 0.47 | 0.48 |
| Atropine | 0.44 | 0.26 | -0.09 | -0.06 | 0.13 | 0.21 |
| Noratropine | 0.43 | 0.35 | -0.06 | -0.10 | 0.35 | 0.35 |
| Apohyoscine | 0.48 | 0.50 | -0.08 | 0.31 | 0.17 | 0.33 |
In Vitro Anti-inflammatory Activities (Membrane Stabilization Assay)
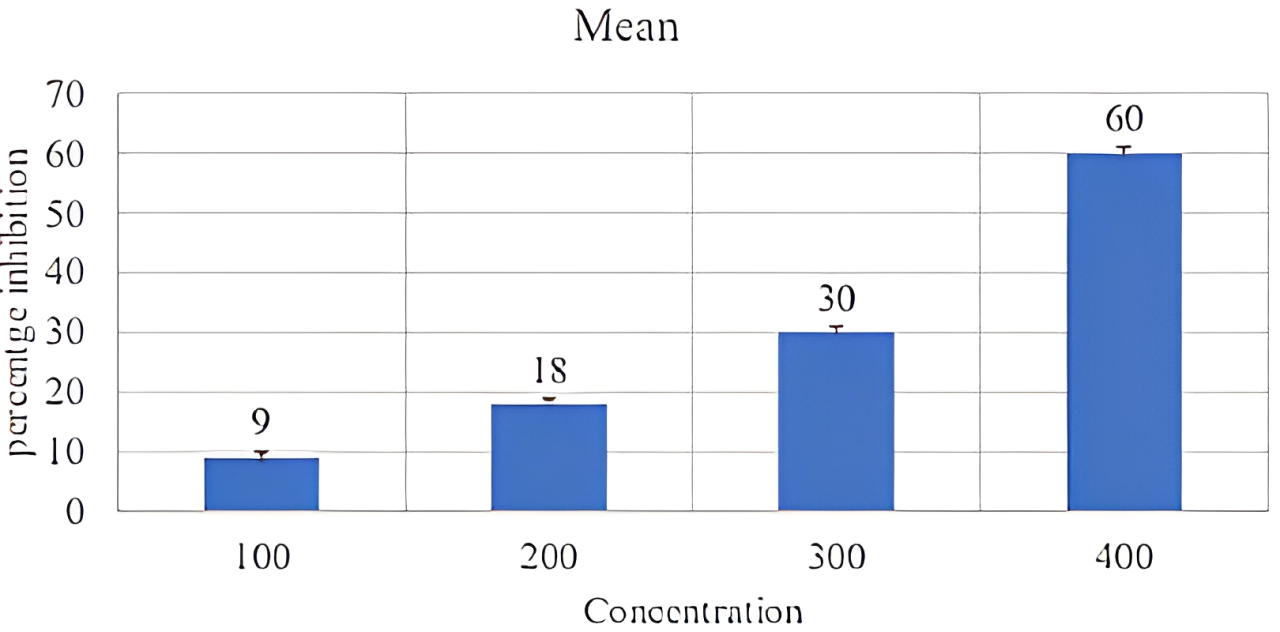
The biological potential of plant extract fractions was studied for their ability to stabilize rat RBC membrane lyses in normal saline. The results obtained were also compared with standard anti-inflammatory agent aspirin based on the % protection. Initially, we decided aspirin was used as standard in the membrane stabilization assay and we performed a pilot experiment for aspirin to choose the correct dose of aspirin. We decide on the concentration for the following assay (Table 7, Figure 5)
| Concentration (normal saline) | Mean | SD |
|---|---|---|
| 100 µg/mL | 9 | 6.350853 |
| 200 µg/mL | 18 | 8.888194 |
| 300 µg/mL | 30 | 8.717798 |
| 400 µg/mL | 60 | 3.605551 |
The fractions were analysed at selected concentrations (500 µg/mL) for anti-inflammatory activity with membrane stabilization methods. Normal saline 500 µg/mL and aspirin 100 µg/mL were used as a negative and positive control (Table 8).
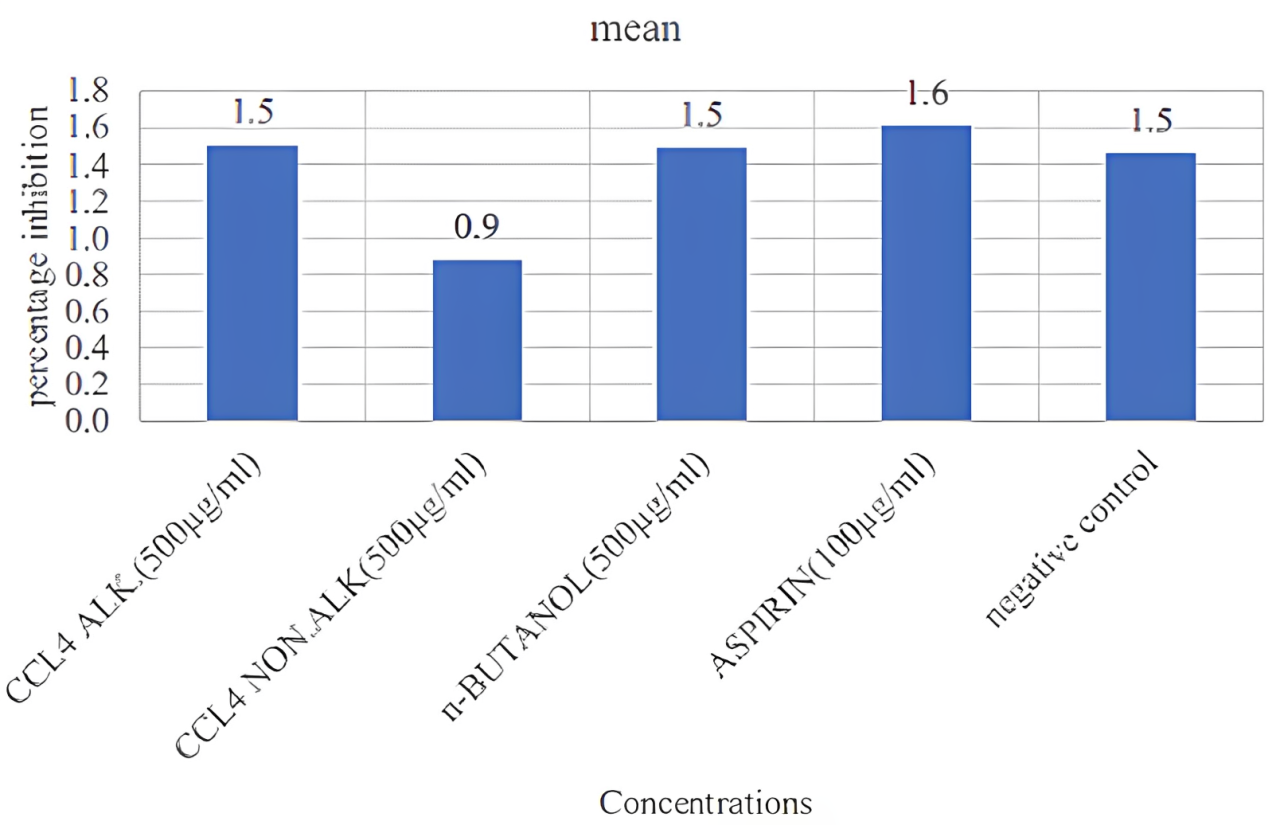
After that we decided to take only one fraction in different concentrations. So, we took CCl4 alkaloidal concentration in 100, 200, and 300 µg/mL, aspirin at 100 µg/mL and normal saline used as a negative control. But we performed only N=1 data in this (Figure 6 and Table 9).
| Concentrations | Mean | SD |
|---|---|---|
| CCL4 ALK. (500µg/mL) | 1.5 | 1.21 |
| CCL4 NON. ALK (500µg/mL) | 0.9 | 1.12 |
| n-BUTANOL (500µg/mL) | 1.5 | 1.23 |
| ASPIRIN (100µg/mL) | 1.6 | 0.74 |
| Negative control | 1.5 | 1.26 |
| Concentration | Sample 1 | Sample 2 | mean | control- sample | control – sample /control | *100 |
|---|---|---|---|---|---|---|
| 100 µg/mL | 1.396 | 1.407 | 1.4 | 0.51 | 0.267 | 26.7 |
| 200 µg/mL | 1.243 | 1.236 | 1.23 | 0.68 | 0.356 | 35.6 |
| 300 µg/mL | 0.705 | 0.691 | 0.69 | 1.22 | 0.638 | 63.87 |
| Aspirin100 µg/mL | 1.306 | 1.289 | 1.29 | 0.62 | 0.324 | 32.46 |
| Negative control | 1.901 | 1.925 | 1.91 | - | - | - |
Conclusions
Firstly, we used in silico methods for drug discovery in the natural plant during the previous decade. We choose three chemical constituents piperine, cinnamic aldehyde, and linoleic acid. After that, we identified structural activity by using bioinformatics tools such as PASS Online, Molinspiration, SEA, SWISS, and SLAP data. We compared the data of these three Phytoconstituents with B. sauveolens Phytoconstituents. On the other side, we have assessed the fractions of this plants and its in vitro anti-inflammatory assay. Based on the in silico and in vitro study, we conclude that B. sauveolens has the potential for application in the treatment of asthma. Further studies such as in vivo studies are required to validate the present findings.
Declarations
Acknowledgment
The authors are thankful to Techno India University and Shoolini University, for supporting its publication. The authors performed the study in their laboratory, and data was collected through online servers and software.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
Not applicable.
Funding Information
Not applicable.
References
- Pawankar R, Canonica GW, Holgate ST, Lockey RF. Allergic diseases and asthma: a major global health concern. Curr Opin Allergy Clin Immunol. (2012) 12(1):39-41.
- Lambrecht BN, Hammad H. The immunology of asthma. Nat Immunol. (2015) 16(1):45-56.
- Martonen T, Fleming J, Schroeter J, Conway J, Hwang D. In- silico modeling of asthma. Adv Drug Deliv Rev. (2003) 55(7):829-49.
- Majumdar S, Ghosh A, Saha S. Modulating interleukins and their receptors interactions with small chemicals using in- silico approach for asthma. Curr Top Med Chem. (2018) 18(13):1123-34.
- Huang S, Wiszniewski L, Derouette JP, Constant S. In vitro organ culture models of asthma. Drug Discov. Today Dis Models. (2009) 6(4):137-44.
- Xu P, Wang L, Chen D, Feng M, Lu Y, Chen R, Qiu C, Li J. The application of proteomics in the diagnosis and treatment of bronchial asthma. Ann Transl Med. (2020) 8(4): 132; 1-7.
- Jeyasri R, Muthuramalingam P, Suba V, Ramesh M, Chen JT. Bacopa monnieri and their bioactive compounds inferred multi-target treatment strategy for neurological diseases: A cheminformatics and system pharmacology approach. Biomolecules. (2020) 10(4):536.
- Romano JD, Tatonetti NP. Informatics and computational methods in natural product drug discovery: a review and perspectives. Front Genet. (2019) 10:368.
- Petricevich VL, Salinas-Sánchez DO, Avilés-Montes D, Sotelo-Leyva C, Abarca-Vargas R. Chemical compounds, the pharmacological and toxicological activity of Brugmansia suaveolens: a review. Plants. (2020) 9(9):1161.
- Chellemi DO, Webster CG, Baker CA, Annamalai M, Achor D, Adkins S. Widespread occurrence and low genetic diversity of Colombian datura virus in Brugmansia suggest an anthropogenic role in virus selection and spread. Plant Dis. (2011) 95(6):755-61.
- Freitas AV, Trigo JR, Brown KS, Witte L, Hartmann T, Barata LE. Tropane and pyrrolizidine alkaloids in the ithomiines Placidula euryanassa and Miraleria cymothoe (Lepidoptera: Nymphalidae). Chemoecology. (1996) 7:61-7.
- Sakunthala P, Charles A, Kesavan D, Ramani VA. Phytochemical screening and adsorption studies of Brugmansia suaveolens. Chem Sci Rev Lett. (2013) 29:319-22.
- Nandakumar A, Mayil Vaganan M, Sundararaju P, Udayakumar R. Phytochemical analysis and nematicidal activity of ethanolic leaf extracts of Datura metel, Datura innoxia and Brugmansia suaveolensis against Meloidogyne incognita. Asian J Biol. (2017) 2 (4): 1-11.
- Stashenko EE, Martinez JR. Sampling flower scent for chromatographic analysis. J Sep Sci. (2008) 31(11):2022-31.
- Mai NT. Investigation on chemical constituents of the Brugmansia suaveolens flowers. J Multi discip Eng Sci Technol. (2019) 6:10021-4.
- Pundir S, Shukla MK, Singh A, Chauhan R, Lal UR, Ali A, Kumar D. A comprehensive review on angel's trumpet (Brugmansia suaveolens). S Afr J Bot. (2022) 151: 266-274.
- Taur DJ, Patil RY. Some medicinal plants with anti-asthmatic potential: a current status. Asian Pac J Trop Biomed. (2011) 1(5):413-8.
- Khan T, Dixit S, Ahmad R, Raza S, Azad I, Joshi S, Khan AR. Molecular docking, PASS analysis, bioactivity score prediction, synthesis, characterization, and biological activity evaluation of a functionalized 2-butanone those micarbazone ligand and its complexes. J Chem Biol. (2017) 10:91-104.
- Khan T, Ahmad R, Azad I, Raza S, Joshi S, Khan AR. Computer-aided drug design and virtual screening of targeted combinatorial libraries of mixed-ligand transition metal complexes of 2-butanone that micarbazon. Comput Biol Chem. (2018) 75:178-95.
- Schenone M, Dančík V, Wagner BK, Clemons PA. Target identification and mechanism of action in chemical biology and drug discovery. Nat Chem Biol. (2013) 9(4):232-40.
- Petricevich VL, Salinas-Sanchez DO, Aviles-Montes D, Sotelo-Leyva C, Abarca-Vargas R. Chemical compounds, pharmacological and toxicological activity of Brugmansia suaveolens: a review. Plants. 2020; 9(9):1161.
- Sarveswaran R, Jayasuriya WJ, Suresh TS. In vitro assays to investigate the anti-inflammatory activity of herbal extracts a review. World J. Pharm. Res. (2017) 6(17) 131- 141.
