RESEARCH ARTICLE
Alpha Lipoic Acid Improves Memory and Antioxidant Enzymes Activity in Diabetic Wistar Rats
Academic Editor: Pilli Govindaiah
Sciences of Pharmacy|Vol. 2, Issue 4, pp. 216-224 (2023)
Received
Aug 12, 2023Revised
Sep 25, 2023Accepted
Sep 25, 2023Published
Oct 2, 2023
Abstract
Keywords:
Introduction
As of 2021, diabetes mellitus (DM) was responsible for an estimated USD 966 billion in global health expenditure 2021. This represents a 316% increase over the last 15 years. The prevalence of DM is estimated at 537 million adults surpassing the 400 million people estimated by the World Health Organisation in 2016 (1), and is projected to reach 693 million by 2025 (2), with type 2 making up about 90% of the cases. Its incidence is increasing rapidly, especially in Africa, with an estimated over 130% increase in the next 25 years (3). DM occurs throughout the world but is more common (especially type 2) in developing countries. The greatest increase in prevalence is, however, expected to occur in Asia and Africa, where most patients will probably be found by 2045 (3).
The pathogenesis of brain impairment caused by chronic hyperglycemia is complex and includes mitochondrial dysfunction, neuroinflammation, neurotransmitters' alteration, and vascular disease, which lead to cognitive impairment (CI), neurodegeneration, and loss of synaptic plasticity, brain aging, and dementia (4, 5). Poor glycemic control has been associated with the progression of cognitive dysfunction (6, 7). The brain pathology underlying cognitive dysfunction is heterogeneous and highly complicated. Traditionally, Alzheimer's disease (AD) is considered the major diagnosis of dementia (8). Although the exact pathophysiology of DM-mediated dementia has not been fully elucidated, existing evidence has shown that both cerebrovascular changes and neurodegeneration are implicated in the development and progression of DM-mediated cognitive dysfunction (9).
High levels of oxidants formed in DM by glucose oxidation, protein glycation, and the subsequent degradation of glycated proteins, and the simultaneously declined antioxidant enzyme levels/activities lead to cell damage, inactivation of enzymes, and lipid peroxidation (10). Reactive oxygen species (ROS) and reactive nitrogen species (RNS) are released from activated immune cells in response to an inflammatory stimulus (11, 12). During this, phagocytic cells release reactive oxygen and nitrogen species (RONS), and non-phagocytic cells are stimulated to produce RONS by pro-inflammatory cytokines (13). Alpha lipoic acid has many biological functions including reducing inflammation, chelating the transitional metal ions, and modulating the signal transduction of nuclear factor (14). It has antioxidant properties hence, can effectively inhibit pathologies in which ROS have been implicated, such as diabetic neuropathy, ischemia-reperfusion injury, radiation injury, and DM-induced oral implant failure (15, 16). This research was aimed at determining the effect of alpha lipoic acid on memory and oxidative stress in diabetic Wistar rats.
Experimental section
Materials
All drugs and reagents were obtained commercially and were of analytical grades. The drugs, reagents, equipment, and other materials used for the study include alpha lipoic acid purchased from Puritan's Pride Inc. (Ronkonkoma, New York, USA). A digital glucometer was used for blood glucose determination (Accu-Check Advantage, Roche Diagnostic, Germany).
Animals, Induction of Diabetes, and Experimental Design
A total of 30 male Wistar rats weighing 200 – 250 grams were used for the study. Animals were allowed for two weeks for acclimatisation to the laboratory environment before the commencement of the experiments. The animals were handled by principles guiding the use and handling of experimental animals per the universal declaration of animals rights proclaimed in Paris on 15 October, 1978 (17). Ethical approval was obtained from Bauchi State University Gadau Committee on Animal Use and Care (BASUG/FBMS/REC/VOL.2/23).
The rats were fasted for 12 – 16h before the commencement of the experiment but were allowed water ad libitum throughout the experiment. The normal groups were fed with standard rat feed only as described by (18) and with little modification. HFD-STZ group was fed a diet with 66.5% labina feed (52% carbohydrate, 5% lipid, 22% protein, 10% water and 11% ash), 13.5% lard, and 20% sugar. The HFD has more calories from lipids (22%) and less carbohydrate (10%) and protein (12%) compared to the control diet. In this research, normal groups were fed with poultry vital feed (grower marsh) (9.7% moisture, 2 % ash, 9% crude fiber, 10% fat, 20 % crude protein and 49.3% carbohydrate), while the high fat-diet groups were fed with a high-fat diet (high-fat diet (HFD): 35% commercial feed, 25% groundnut, 25% fat, and 15% groundnut oil) for the induction of obesity and DM for six weeks followed by a single dose of streptozotocin (STZ) 40 mg/kg intraperitoneally (IP) and high-fat diet for another 2 weeks. Rats fasted for 12h and fasting blood glucose levels were measured to confirm the establishment of DM (18). Rats with fasting blood glucose levels of 16 mmol/L or more were considered diabetic and selected for the study (18). The drug administration, estimation of fasting blood glucose level, and neurobehavioral tests were conducted between 07: 00 am – 09: 00 am on day 0 (pre-treatment) and 21 (post-treatment). The rats were divided into six groups (each group had 5 rats). All drug administrations were done orally for 21 days as follows. Group I served as normal control and received 1 mL/kg 0.9% normal saline; Group II, III, IV, V, and VI were all diabetic and received 1 mL/kg 0.9% normal saline, 1 mg/kg glibenclamide, 100 mg/kg ALA, 200 mg/kg ALA, 400 mg/kg ALA respectively.
Novel Object Recognition Task
The Novel Object Recognition Task is an open-field evaluation of rats' innate propensity to study a novel object rather than one they are familiar with. The decision to examine the novel object and the decision to resume exploration after an object has been moved demonstrate the use of memory and learning processes (recognition) (19). The test was conducted between 07: 00 am – 09: 00 am after induction prior to the commencement of ALA administration. The illustration below is a modification of schematic diagram of novel object recognition task (20).
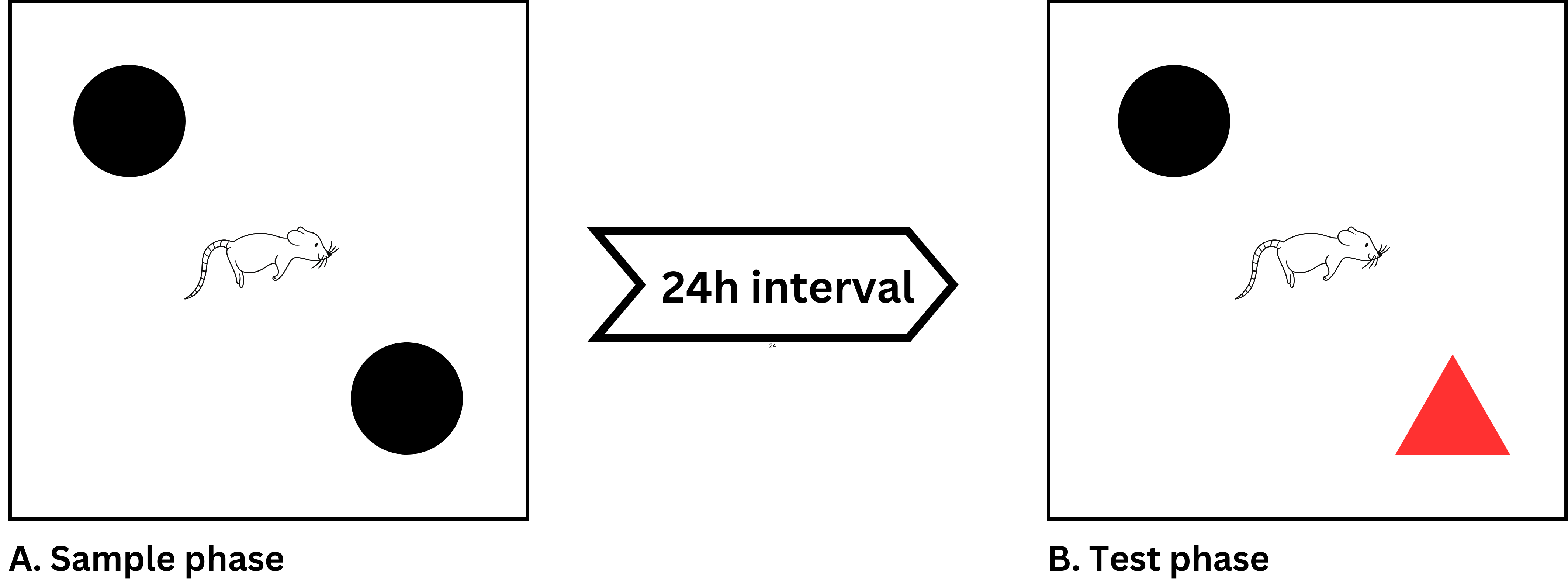
This task consists of two phases separated by a 24h period. (Retention interval): the sample phase and the test phase. The rats were shown two identical objects during the sample phase. These items were positioned 15 cm from each neighboring wall in the corners of an arena. Between the sample and test phases, each rat was put in the center of the arena and given 5min to examine the items. To get rid of smell clues, alcohol was used to disinfect all the items. Then, during the test phase, one of the objects was switched, and the rat was given 5 min to investigate the new object. The amount of time spent investigating the altered objects is compared to the amount of time spent investigating the other object (spatial memory, Ability to identify and discriminate). The rat will spend more time investigating the altered object that was changed compared to the unchanged object if its spatial memory and ability to discriminate and recognize are still functional. Difference and recognition index were calculated using Equation 1 and 2 (19, 21-23).
Where Tn means time spent exploring the novel object and Tf means time spent exploring the familiar object.
Determination of Fasting Blood Glucose Level
The blood samples were obtained from the rat tail vein on day 0 (pre-treatment) and day 21 (post-treatment). A digital glucometer was used to measure the blood glucose levels using the glucose oxidase principle (24) using the digital glucometer (Accu-Check Advantage, Roche Diagnostic, Germany), and results were expressed in mmol/L.
Termination of Experiment and Sample Collection
On day 21, all rats were subjected to light anesthesia by exposing them to chloroform soaked in cotton wool and placed in an aesthetic box. Brain tissue was collected and homogenized (25). The homogenate was used for antioxidant and lipid peroxidation analysis.
Assessment of Antioxidant Enzymes and Lipid Peroxidation
Catalase (CAT) Activity
Catalase (CAT) activity was measured using the method of (26). Exactly 10 µl of homogenate was added to a test tube containing 2.80 mL of 50 mM potassium phosphate buffer (pH 7.0). The reaction was initiated by adding 0.1 mL of freshly prepared 30 mM H2O2 and the decomposition rate of H2O2 was measured at 240 nm for 5 min using spectrophotometer. A molar extinction coefficient (e) of 0.041 mM¯1cm¯1 was used to calculate the catalase activity using Equation 3 and 4.
Superoxide Dismutase (SOD) Activity
Superoxide dismutase (SOD) was determined using the previous method (27). The basis of this assay lies in superoxide dismutase (SOD) inhibiting the autooxidation of adrenaline at pH 10.2. The assay utilized the following reagents: a 0.05 M carbonate buffer, prepared by dissolving 114.3 g of Na2CO3 and 4.2 g of NaHCO3 in distilled water and adjusting the volume to 1000 mL using a volumetric flask. The buffer's pH was then adjusted to 10.2. A fresh solution of 0.3 mM adrenaline was made by dissolving 0.01 g of adrenaline in 17 mL of distilled water. To create a 1: 10 dilution of the microsome, 0.1 mL of tissue homogenate was diluted with 0.9 mL of distilled water. A mixture containing 0.2 mL of the diluted microsome and 2.5 mL of 0.05 M carbonate buffer was aliquoted. The reaction was initiated by adding 0.3 mL of 0.3 mM adrenaline. For the reference mixture, 2.5 mL of 0.05 M carbonate buffer, 0.3 mL of 0.3 mM adrenaline, and 0.2 mL of distilled water were combined. Absorbance readings were taken at 480 nm over a time span of 30s to 150s. Increase in absorbance and percentage inhibition were calculated using Equation 5 and 6.
One unit of SOD activity is the quantity of SOD necessary to elicit 50% inhibition of the oxidation of Adrenaline to adrenochrome in one minute.
Reduced Glutathione (GSH) Concentration
The concentration of reduced glutathione (GSH) was determined using a previously established method (28). To achieve this, a supernatant of 1 mL (resulting from 0.5 mL of plasma precipitated by 2 mL of 5% trichloroacetic acid (TCA)) was utilized. Subsequently, 0.5 mL of Ellman's reagent (0.0198% DTNB in 1% sodium citrate) and 3 mL of phosphate buffer (pH 8.0) were added to the supernatant. The resulting mixture was subjected to colorimetric analysis by measuring the developed color at 412 nm.
Lipid Peroxidation
Lipid peroxidation in plasma was estimated colorimetrically by measuring malondialdehyde (MDA) using the established method (29, 30). In brief, 0.1 mL of tissue homogenate was treated with 2 mL of (1: 1: 1 ratio) TBA–TCA–HCL reagent (TBA 0.37%, 0.25N HCL, and 15% TCA) and placed in a water bath for 15 min, cooled and centrifuged and then clear supernatant was measured at 535 nm against reference blank.
Statistical Analysis
Statistical Package for the Social Sciences version 22 (SPSS 22) was used to analyse the data. Data obtained were presented as mean ± standard error of the mean (SEM). Analysis of variance (ANOVA) was employed to compare the level of significance between experimental groups and Tukey’s post hoc test was conducted to compare between different groups while paired T-test was used to compare within group. A significant level was considered at p< 0.05.
| Groups | Day 0 (mmol/L) | Day 21 (mmol/L) |
|---|---|---|
| I | 4.88 ± 0.50c | 4.84 ± 0.25c |
| II | 19.96 ± 1.20 | 20.06 ± 0.70 |
| II | 19.76 ± 1.02 | 7.02 ± 0.79a, b |
| IV | 20.86 ± 1.40 | 10.02 ± 0.71a, b |
| V | 21.44 ± 1.14 | 8.78 ± 0.94a, b |
| VI | 20.44 ± 1.01 | 8.60 ± 0.68a, b |
| Note: (a, p< 0.001) shows significant difference to its day 0 value, (b, p< 0.001) shows significant difference compared to diabetic control value (group II), and (c, p< 0.001) shows significant difference compared to all groups on the same day. | ||
Result
Alpha Lipoic Acid on Fasting Blood Glucose Level in Diabetic Rats
In order to confirm the DM condition and the effect of ALA administration, we checked the blood glucose level was checked at the beginning (day 0) and end (day 21) of the experiment. There was a significant [F (5, 30) = 55.51, p< 0.0001)] difference between the groups (see Table 1).
Alpha Lipoic Acid on the Long-term and Recognition Memory in Diabetic Rats
Figure 2 illustrates the effects of ALA (at doses of 100 mg/kg, 200 mg/kg, and 400 mg/kg) on the memory of type-2 diabetic Wistar rats. The groups treated with 400 mg/kg (group VI) and 200 mg/kg (group V) of ALA exhibited a significant increase (p< 0.05) in the time spent exploring the novel object after 21 days of administration compared to pre-treatment. Furthermore, a significant increase was observed [F (5, 30) = 6.81, p< 0.0001)] in the time spent exploring novel objects in the 400 mg/kg group after 21 days compared to the diabetic control group (group II: 26.24 ± 3.81s).
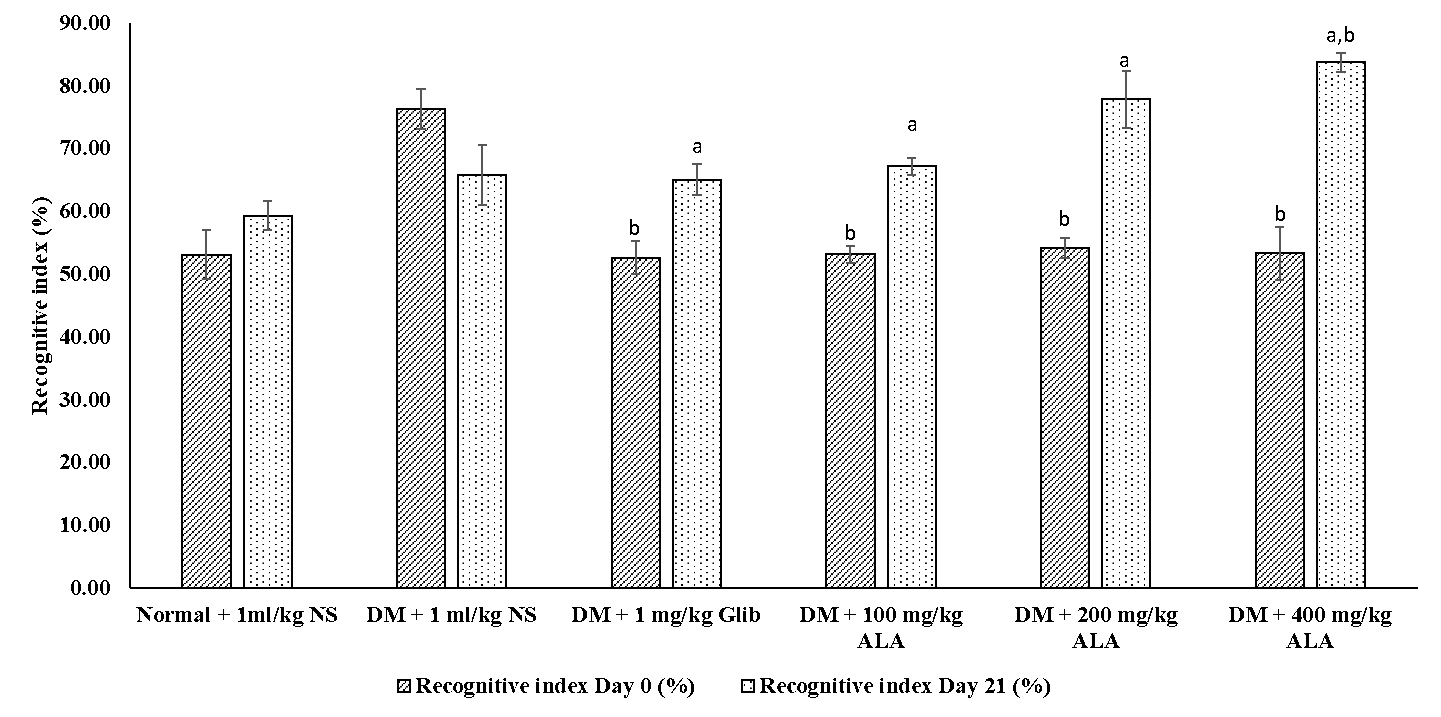
Figure 3 shows the effect of ALA (100 mg/kg, 200 mg/kg and 400 mg/kg) on recognitive index of type-2 diabetic Wistar rats. The 400 mg/kg (group VI) ALA-treated groups showed a significant increase (p< 0.05) in the percentage of recognition after 21 days of administration (83.77±1.49%) when compared to the pre-treatment (53.34 ± 4.25%). Furthermore, we observed a significant increase [F (5, 30) = 3.93, p≤0.01)] in the percentage of recognition in the group VI (83.77 ± 1.49%) after 21 days compared to the diabetic control group (group II: 65.09±2.52%).
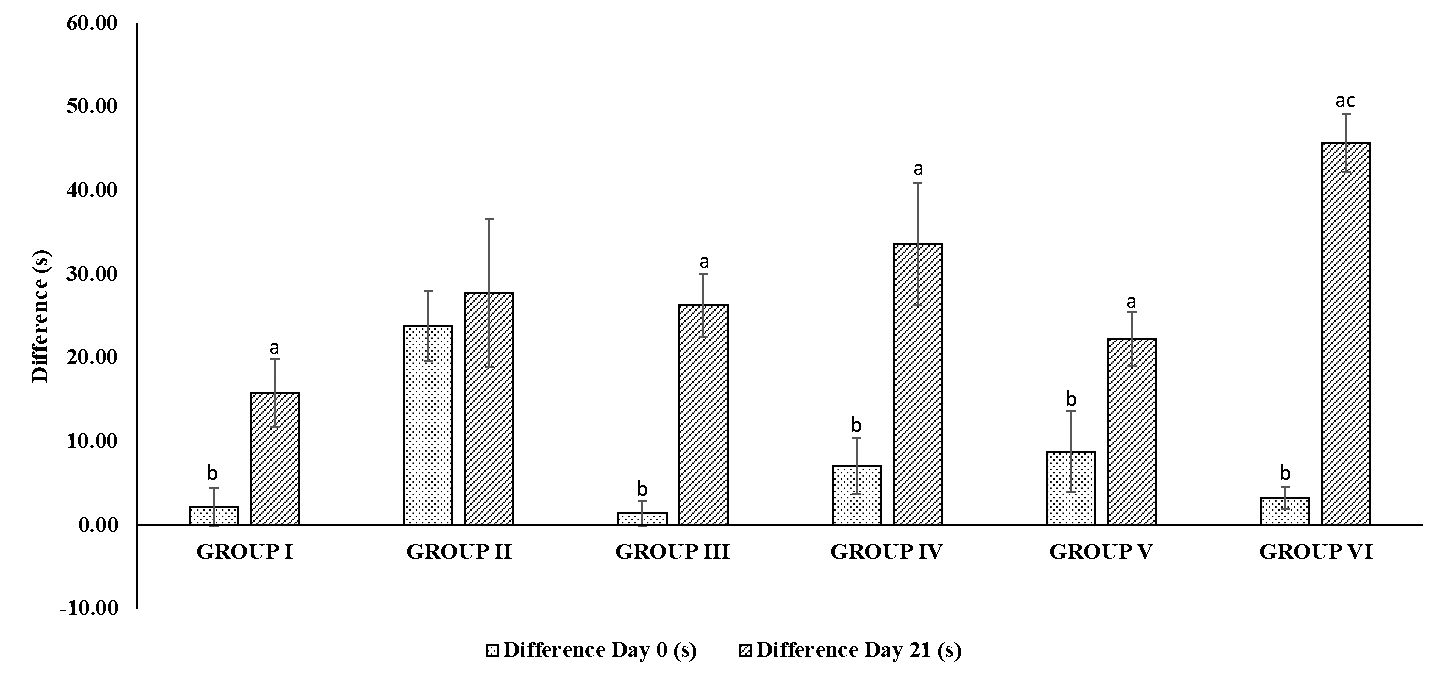
| Groups | Catalase (IU/mg) | SOD (IU/mg) | Percentage SOD Inhibition (%) | GSH (μg/mL) | MDA (nmol/L) |
|---|---|---|---|---|---|
| I | 1.57 ± 0.09a | 0.84 ± 0.05a | 42.00 ± 0.01a | 93.20 ± 5.30a | 2.20 ± 0.25a |
| II | 0.84 ± 0.03 | 0.49 ± 0.03 | 25.13 ± 1.41 | 51.64 ± 0.87 | 7.22 ± 0.42 |
| III | 1.38 ± 0.03a | 0.71 ± 0.04a | 35.08 ± 1.98a | 60.18 ± 1.95a | 4.50 ± 0.19a |
| IV | 1.32 ± 0.33a | 0.70 ± 0.04a | 35.52 ± 2.00a | 69.06 ± 0.89a | 4.78 ± 0.34a |
| V | 1.69 ± 0.01a | 0.98 ± 0.03a | 48.80 ± 1.57a | 78.48 ± 1.37a | 3.64 ± 0.18a |
| VI | 1.76 ± 0.02a | 1.02 ± 0.03a | 50.98 ± 1.68a | 91.08 ± 3.49a | 3.60 ± 0.25a |
| Note: (a, p< 0.05) shows significant difference compared to diabetic control value (group II) | |||||
Alpha Lipoic Acid on Level of Antioxidant Enzymes in Diabetic Rats
We also evaluated the concentration of catalase in the brain homogenate (Table 2) in the diabetic rats after 21 days of ALA administration at different doses (100 mg/kg, 200 mg/kg, and 400 mg/kg). We observed a significant [F (5, 30) = 5.69, p< 0.001)] increase in the concentration of CAT level in the 200 mg/kg and 400 mg/kg groups (group V: 1.69 ± 0.01 IU/mg and group VI: 1.76 ± 0.02 IU/mg) compared to the diabetic control group (0.84 ± 0.03 IU/mg).
Furthermore, the activity of SOD in the brain homogenate (Table 2) in the diabetic rats after 21 days of ALA administration at different doses (100 mg/kg, 200 mg/kg, and 400 mg/kg) were improved based on the findings of this study. We observed a significant [F (5, 30) = 27.40, p< 0.0001)] increase in the concentration of SOD in the groups treated with ALA at 200 mg/kg and 400 mg/kg (groups V and VI) as well as glibenclamide (group III) with values of 0.98 ± 0.03 IU/mg, 1.02±0.71 IU/mg and 0.71 ± 0.04 IU/mg respectively compared to the diabetic control group (0.49±0.03 IU/mg).
The activity of GSH in the brain homogenate (Table 2) in the diabetic rats after 21 days of ALA administration at different doses (100 mg/kg, 200 mg/kg and 400 mg/kg) were also improved. We observed significant [F (5, 30) = 52.11, p< 0.0001)] increase in the concentration of GSH in all the groups treated with glibenclamide (group III) and ALA (Groups IV, V and VI) with values of 60.18 ± 1.95 μg/mL and 69.06 ± 0.89 μg/mL, 78.48 ± 1.37 μg/mL and 91.08 ± 3.49 μg/mL respectively compared to the diabetic control group (51.64 ± 0.87 μg/mL).
The marker of lipid peroxidation (MDA) was determined using brain homogenate (see Table 2) in the diabetic rats after 21 days of ALA administration at different doses (100 mg/kg, 200 mg/kg and 400 mg/kg). We found a significant [F (5, 30) = 34.36, p< 0.0001)] decrease in the concentration of MDA in all the groups treated with glibenclamide (group III) and ALA (Groups IV, V and VI) with values of 4.50±0.19 nmol/mg and 4.78±0.34 nmol/mg, 3.64±0.18 nmol/mg and 3.60±0.25 nmol/mg respectively compared to the diabetic control group (7.22±0.42 nmol/mg).
Discussion
Several scientific researchers had over the years associated DM with peripheral nerve damage, Alzheimer’s disease and CI. DM also negatively impacts the lipid- and protein-intermediate metabolism (31-33). It was postulated that hyperglycemia, oxidative stress, dyslipidemia and inflammation are the major mediators for the pathogenesis and progression of memory impairment in DM (34, 35). Alpha lipoic acid (ALA) has been used for some years now as an antioxidant with antihyperglycemic and anti-inflammatory supplement and due to its extensive range of pharmacological activity, treat various illnesses (36, 37).
The results obtained in this study indicated that DM has affected long-term memory in NORT in type-2 diabetic rats. There was a significant (p< 0.05) difference between day 0 and day 21 of the diabetic untreated group (group II). This indicated that DM has affected long-term memory in NORT. However, we observed a significant increase in long-term memory in the ALA groups after 21 days compared to the pre-treatment. This could be associated with the antihyperglycemic as seen in this study and antilipidemic effect of ALA (38). This aligns with a prior study indicating that ALA enhances mouse memory, assessed through the Barnes test (36). Furthermore, Ko CY, et al. (2021) reported that ALA may ameliorate cognition impairment via alleviating cerebral IR improvement and cerebral synaptic plasticity in diabetic rats (37).
The gradual decrease in the recognition index observed in the diabetic untreated group of the present study further confirms memory impairment induced by DM. This may be associated with hyperglycemia and oxidative stress which have been implicated in learning and memory impairment. We also observed an improvement in all the ALA-treated groups with the highest dose showing a significantly higher effect compared to the diabetic untreated group after 21 days of administration. From the result observed in the ALA-treated group, it is evident that hyperglycemia is associated with impairment of long-term and recognition ability in the experimental animals which was significantly improved (p< 0.05) by the daily administration of ALA as seen in the groups treated with 100 mg/kg, 200 mg/kg and 400 mg/kg of ALA. This improvement may be associated with ALA’s antihyperglycemic and antioxidant effects as demonstrated in our results (39-41) and lipid lowering effect as demonstrated by previous study (38). However, this finding contradicts the findings of Villasana LE, et al. (2013) who reported ALA treatment impaired cortical-dependent novel object recognition in experimental animals (42). This could be because the memory impairment in the present study was secondary to diabetes, not irradiation or the doses used in the present study.
Reactive oxygen species (ROS) are released from activated immune cells in response to an inflammatory stimulus. The balance between the rate of free radical generation and elimination is important. Excess cellular radical generation can be harmful (39). The SOD offers first line of defense against ROS by scavenging and catalyzing the dismutation of superoxide, produced by cellular metabolism, into hydrogen peroxide (H2O2) and oxygen (O2) (40). Reduced glutathione reduces the oxidized form of the enzyme glutathione peroxidase, which in turn reduces hydrogen peroxide (H2O2), a dangerously reactive species within the cell (43). In this research, we found a significant improvement in the brain-homogenated antioxidant enzymes across the different doses of ALA. This further signifies the effectiveness of ALA in free radicals scavenging (44). Oxidative stress and free radicals have been implicated in most if not all of the complications of DM and diabetes-induced CI (45). This action of ALA has further explained the mechanism through which long-term and recognition memories were improved as seen in Figures 2 and 3, respectively. Furthermore, we also observed a significant reduction in the biomarker of lipid peroxidation (MDA). This further signifies that ALA at all the doses tested improved membrane integrity by reducing lipid peroxidation. This finding agrees with many other findings that reported antioxidant activity of ALA in experimental animals.
Conclusion
Results obtained in the present study demonstrated that type-2 DM causes memory impairment by affecting long-term and recognition memory in novel object recognition task through hyperglycemia and oxidative stress. This significantly improved after 21 days of administration of ALA (p< 0.05). The activities of antioxidant enzymes were also improved with significant decrease in lipid peroxidation (p< 0.05). This signifies that ALA could be a potential therapeutic target for memory impairment associated with type-2 DM.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
The study was approved by Bauchi State University Gadau Committee on Animal Use and Care with approval letter number of BASUG/FBMS/REC/VOL.2/23.
Funding Information
Not applicable.
References
- World Health Organization. World health statistics 2022 (Monitoring health of the SDGs). Monitoring health of the SDGs. 2022. 1-131 p.
- Umar AK, Luckanagul JA, Zothantluanga JH, Sriwidodo S. Complexed Polymer Film-Forming Spray: An Optimal Delivery System for Secretome of Mesenchymal Stem Cell as Diabetic Wound Dressing? Pharmaceuticals. 2022 Jul 14;15(7):867.
- International Diabetes Federation. Diabetes in Africa - 2021. Int Diabetes Fed. 2021;1–3.
- Grieco M, Giorgi A, Gentile MC, D’Erme M, Morano S, Maras B, et al. Glucagon-Like Peptide-1: A Focus on Neurodegenerative Diseases. Vol. 13, Frontiers in Neuroscience. 2019. p. 1–7.
- Rivai B, Umar AK. Neuroprotective compounds from marine invertebrates. Beni-Suef Univ J Basic Appl Sci. 2023 Jul 28;12(1):71.
- Cukierman-Yaffe T, Gerstein HC, Williamson JD, Lazar RM, Lovato L, Miller ME, et al. Relationship between baseline glycemic control and cognitive function in individuals with type 2 diabetes and other cardiovascular rIsk factors the action to control cardiovascular risk in diabetes-memory in diabetes (ACCORD-MIND) trial. Diabetes Care. 2009;32(2):221–6.
- Lu FP, Lin KP, Kuo HK. Diabetes and the risk of multi-system aging phenotypes: A systematic review and meta-analysis. Vol. 4, PLoS ONE. 2009.
- Seto SW, Yang GY, Kiat H, Bensoussan A, Kwan YW, Chang D. Diabetes Mellitus , Cognitive Impairment , and Traditional Chinese Medicine. 2015;2015.
- Herath PM, Cherbuin N, Eramudugolla R, Anstey KJ. The Effect of Diabetes Medication on Cognitive Function: Evidence from the PATH Through Life Study. Biomed Res Int. 2016;2016.
- Weidinger A, Kozlov A V. Biological activities of reactive oxygen and nitrogen species: Oxidative stress versus signal transduction. Vol. 5, Biomolecules. 2015. p. 472–84.
- Fubini B, Hubbard A. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) generation by silica in inflammation and fibrosis. Vol. 34, Free Radical Biology and Medicine. 2003. p. 1507–16.
- Ozcan A, Ogun M. Biochemistry of Reactive Oxygen and Nitrogen Species. In: Basic Principles and Clinical Significance of Oxidative Stress. 2015.
- Hussain SP, Hofseth LJ, Harris CC. Radical causes of cancer. Vol. 3, Nature Reviews Cancer. 2003. p. 276–85.
- Tabrizi R, Borhani-Haghighi A, Mirhosseini N, Lankarani KB, Naghibzadeh-Tahami A, Akbari M, et al. The effects of alpha-lipoic acid supplementation on fasting glucose and lipid profiles among patients with stroke: a systematic review and meta-analysis of randomized controlled trials. Vol. 18, Journal of Diabetes and Metabolic Disorders. Journal of Diabetes & Metabolic Disorders; 2019. p. 585–95.
- Serhiyenko V, Serhiyenko L, Suslik G, Serhiyenko A. Alpha-lipoic acid : mechanisms of action and beneficial effects in the prevention and treatment of diabetic complications. 2018;7(4):174–8.
- Vallianou N, Evangelopoulos A, Koutalas P. Alpha-lipoic acid and diabetic neuropathy. Vol. 6, Review of Diabetic Studies. 2009. p. 230–6.
- United Nations Educational Scientific and Cultural Organization. Universal Declaration of Animal Rights (15 October 1978). United Nations Educ Sci Cult Organ. 1978;(October):8–9.
- De MagalhÃes DA, Kume WT, Correia FS, Queiroz TS, Allebrandt Neto EW, Dos Santos MP, et al. High-fat diet and streptozotocin in the induction of type 2 diabetes mellitus: A new proposal. An Acad Bras Cienc. 2019;91(1):1–14.
- Antunes M, Biala G. The novel object recognition memory: Neurobiology, test procedure, and its modifications. Vol. 13, Cognitive Processing. 2012. p. 93–110.
- Kim DH, Jang YS, Jeon W, Han JS. Assessment of Cognitive Phenotyping in Inbred, Genetically Modified Mice, and Transgenic Mouse Models of Alzheimer’s Disease. Exp Neurobiol. 2019 Apr;28:146.
- Baxter MG. “I’ve seen it all before” Explaining age-related impairments in object recognition. Theoretical comment on Burke et al. (2010). Vol. 124, Behavioral Neuroscience. 2010. p. 706–9.
- Burke SN, Wallace JL, Nematollahi S, Uprety AR, Barnes CA. Pattern Separation Deficits May Contribute to Age-Associated Recognition Impairments. 2010;124(5):559–73.
- Garkuwa U, Alhassan A, Tanko Y. Effect of Curcumin on Blood Glucose Level and Some Neurobehavioral Responses in Alloxan-induced Diabetic Swiss Albino Mice. J Adv Med Pharm Sci. 2017;14(1):1–7.
- BEACH EF, TURNER JJ. An enzymatic method for glucose determination in body fluids. Clin Chem. 1958 Dec;4(6):462–75.
- Kidwai AM. Homogenization and Fractionation Techniques in Smooth Muscle. In: Daniel EE, Paton DM, editors. Smooth Muscle. Boston, MA: Springer US; 1975. p. 543–54.
- Aebi H. Catalase. In: Bergmeyer HU (Ed), editor. Method of Enzymatic Analysis. New York: Academic Press Inc.; 1974. p. 673–84.
- Fridovich I. Superoxide dismutases. An adaptation to a paramagnetic gas. J Biol Chem. 1989 May;264(14):7761–4.
- ELLMAN GL. Tissue sulfhydryl groups. Arch Biochem Biophys. 1959 May;82(1):70–7.
- Albro PW, Corbett JT, Schroeder JL. Application of the thiobarbiturate assay to the measurement of lipid peroxidation products in microsomes. J Biochem Biophys Methods. 1986 Oct;13(3):185–94.
- Das BS, Thurnham DI, Patnaik JK, Das DB, Satpathy R, Bose TK. Increased plasma lipid peroxidation in riboflavin-deficient, malaria-infected children. Am J Clin Nutr. 1990 May;51(5):859–63.
- Hossain KFB, Akter M, Rahman MM, Sikder MT, Rahaman MS, Yamasaki S, et al. Amelioration of metal-induced cellular stress by α-lipoic acid and dihydrolipoic acid through antioxidative effects in pc12 cells and caco-2 cells. Int J Environ Res Public Health. 2021;18(4):1–15.
- Zhang X, Alshakhshir N, Zhao L. Glycolytic Metabolism, Brain Resilience, and Alzheimer’s Disease. Vol. 15, Frontiers in Neuroscience. 2021. p. 1–19.
- Ehtewish H, Arredouani A, El-Agnaf O. Diagnostic, Prognostic, and Mechanistic Biomarkers of Diabetes Mellitus-Associated Cognitive Decline. Vol. 23, International Journal of Molecular Sciences. 2022.
- Duch DE. Resin transfer molded high temperature composites. In: International SAMPE Symposium and Exhibition (Proceedings). 1999. p. 1149–60.
- Mendoza-núñez VM, García-martínez BI, Rosado-pérez J, Santiago-osorio E, Pedraza-chaverri J. The Effect of 600 mg Alpha-lipoic Acid Supplementation on Oxidative Stress , Inflammation , and RAGE in Older Adults with Type 2 Diabetes Mellitus. 2019;2019.
- Farr SA, Price TO, Banks WA, Ercal N, Morley JE. Effect of alpha-lipoic acid on memory, oxidation, and lifespan in SAMP8 mice. J Alzheimer’s Dis. 2012;32(2):447–55.
- Ko CY, Xu JH, Lo YM, Tu RS, Wu JSB, Huang WC, et al. Alleviative Effect of Alpha-Lipoic Acid on Cognitive Impairment in High-Fat Diet and Streptozotocin-Induced Type 2 Diabetic Rats. Front Aging Neurosci. 2021;13(November):1–12.
- Garkuwa UA, Ibrahim M, Saleh A, Alhassan A, Kawu MU. Alpha lipoic acid improved blood glucose level and lipid profile in type-2 diabetic Wistar rats. Gadau Jorunal Pure Allied Scineces. 2023;2(2):106–12.
- Tavassolifar MJ, Vodjgani M, Salehi Z, Izad M. The Influence of Reactive Oxygen Species in the Immune System and Pathogenesis of Multiple Sclerosis. Autoimmune Dis. 2020;2020.
- Wang Y, Branicky R, Noë A, Hekimi S. Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol. 2018 Jun 4;217(6):1915–28.
- Rochette L, Ghibu S, Richard C, Zeller M, Cottin Y, Vergely C. Direct and indirect antioxidant properties of α-lipoic acid and therapeutic potential. Vol. 57, Molecular Nutrition and Food Research. 2013. p. 114–25.
- Villasana LE, Rosenthal R, Doctrow S, Pfankuch T, Garfinkel M, Raber J. Effects of alpha-lipoic acid on associative and spatial memory of sham-irradiated and 56 Fe-irradiated C57BL/6J male mice. Pharmacol Biochem Behav. 2013;103(3):487–93.
- Deponte M. Glutathione catalysis and the reaction mechanisms of glutathione-dependent enzymes. Vol. 1830, Biochimica et Biophysica Acta - General Subjects. 2013. p. 3217–66.
- Sivitz WI, Yorek MA. Mitochondrial dysfunction in diabetes: From molecular mechanisms to functional significance and therapeutic opportunities. Vol. 12, Antioxidants and Redox Signaling. 2010. p. 537–77.
- Newsholme P, Rebelato E, Abdulkader F, Krause M, Carpinelli A, Curi R. Reactive oxygen and nitrogen species generation, antioxidant defenses, and β-cell function: A critical role for amino acids. Vol. 214, Journal of Endocrinology. 2012. p. 11–20.
