RESEARCH ARTICLE
Effects of Methanolic Leaf Extract and Fractions of Irvingia Gabonensis on Hematological Parameters in Wistar Rats with Splenomegaly
Academic Editor: Mohd Shahezwan Abd Wahab
Sciences of Pharmacy|Vol. 3, Issue 1, pp. 9-17 (2024)
Received
Oct 21, 2023Revised
Nov 24, 2023Accepted
Nov 26, 2023Published
Jan 5, 2024
Abstract
Introduction
The spleen plays a significant role in hematopoiesis and immunosurveillance. Its major functions include the clearance of senescent and abnormal erythrocytes, their remnants, opsonized platelets, and white blood cells, along with the removal of microorganisms and antigens. Splenomegaly is a condition characterized by the enlargement of the spleen, which can be assessed through measurements of its size or weight (1-3). Splenomegaly is an uncommon condition, with an estimated prevalence of around 2% among the entire population of the United States. Among adults, there have been no reported patterns of prevalence based on factors such as ethnicity, gender, or age. In contrast, tropical splenomegaly is highly prevalent in regions like Asia and Africa. It's worth noting that in older individuals, the spleen's capsule tends to thin, increasing the risk of rupture (4).
Splenomegaly can influence various hematological parameters, including PCV (packed cell volume), HB (Haemoglobin), and RBC (red blood cell) count. Changes in plasma volume and red cell mass, especially in splenomegaly, may not be accurately reflected through hematocrit level determination alone (5). This implies that splenomegaly can impact PCV, HB, and RBC count. Bolognesi et al. (2002) explore the role of spleen enlargement in the context of cirrhosis with portal hypertension. They point out that splenomegaly is frequently observed in this condition and may lead to alterations in hematological parameters. While they may not specifically address PCV, HB, and RBC count, splenomegaly in cirrhosis suggests potential effects on these parameters (6).
Plants have been utilized as a source of medicinal substances for centuries, and the current trend shows an increasing use of these plants for this purpose (7). About 51 medicinal plants in Nigeria are known to have an anti-anaemic effect (7). Irvingia gabonensis is rich in polyphenols, primarily composed of ellagic acid, mono-, di-, and tri-O-methyl-ellagic acid, and their related glycosides (8). In ethnomedicinal practices, various parts of I. gabonensis are used for multiple health concerns (9). In a study on the Immunological and hematological effects of I. gabonensis stem bark in sodium arsenite-exposed rats, the authors concluded that ethanol stem bark extract of I. gabonensis may be protective against sodium arsenite-induced inflammation and hematological derangements in Wistar rats (10). A similar study observed that I. gabonensis stem bark extract reversed changes in hematological parameters of cadmium-induced hematotoxicity rats (11). Powdered kernels from the plant act as an astringent and are applied to treat burns (12). Additionally, the stem of the tree has served as chewing sticks for dental hygiene, aiding in teeth cleaning (13). In Nigeria and Cameroon, the seeds of this plant are used as a condiment in soup (14). Yet, there is a lack of information available regarding the impact of methanolic leaf extract and fractions from I. gabonensis on the hematological parameters in splenomegaly-induced Wistar rats. This study aimed to explore the effects of I. gabonensis leaf extract and fractions on the Blood Parameters of Splenomegaly Wistar Rats.
Experimental Section
Collection and Authentication of Plant Materials
Fresh leaves of I. gabonensis were collected from Awka, Anambra State, Nigeria, in the early hours of the day. The plant samples were authenticated by Mr A. O. Ozioko at the International Centre for Ethnomedicine and Drug Development, Nsukka, Enugu State, Nigeria. The voucher specimen was deposited in the herbalism at the Department of Pharmacognosy and Traditional Medicine Faculty of Pharmaceutical Sciences, Agulu campus, Nnamdi Azikiwe University.
Preparation of Plant Materials for Extraction
The leaves were air-dried at room temperature for about two weeks. After drying, the leaves were subjected to pulverization using a manual blender. The blender was properly cleaned to avoid the influx of any adulterant or foreign particles. After blending, the powdered particles were properly stored in a refrigerator until they were needed for extraction.
Extraction of Powdered Plant Materials
The extraction was done using the Soxhlet extraction method with methanol at 70oC. One thousand grams of the powdered plant material was used for the extraction. The extraction was done until the colour of the solvent returning to the heated round bottom flask became colourless. At this point, the extraction was carried out further for three more cycles to ensure complete extraction. After the extraction, the sample was concentrated with a rotary evaporator at 40oC. The concentrated extract was stored in a refrigerator.
Phytochemical Analysis
Phytochemical analysis of the powdered I. gabonensis leaves involved testing for alkaloids, flavonoids, glycosides, tannins, saponins, proteins, and starch. These tests were performed using established methods, as documented by Trease and Evans (15).
Fractionation
The fractionation of the crude methanol extract was carried out according to the method of Onyegbule et al. , 2014 (16). The methanolic extract was solubilized in methanol and made up to 300 ml. The 300 ml sample was divided into 3 of 100 ml each. A 100 ml methanol extract was evaporated until it reached 60% of the total volume. Subsequently, 50 mL of this sample was vigorously shaken with 150 mL hexane in a separation funnel. The mixture was then left undisturbed for one hour to facilitate the separation of components based on their polarity. Following this, the ethyl acetate fraction of the extract was gathered and concentrated using a water bath at a temperature of 50°C. This same method was utilized to obtain Butanol and ethyl acetate fractions.
Animals for the Study
Healthy adult male and female Albino Wistar rats weighing 150-200 g were used for the experiments. The animals were obtained from the animal house of the Department of Pharmacology and Toxicology, Faculty of Pharmaceutical Sciences, Nnamdi Azikiwe University, Awka, Anambra state, Nigeria. They were maintained under standard environmental conditions with free access to a normal diet and clean water. Ethical approval for the study was obtained from the Enugu State University of Science and Technology Animal Ethics Committee, with the approval number ESUT/AEC/0113/AP097.
Acute Toxicity
The acute toxicity test for the extract and fractions was conducted following Lorke’s method (17). A total of 52 female rats were utilized for this study. In the initial phase of the test, there were 9 rats for both the extract and the three fractions. These 9 rats were divided into 3 groups of 3 rats each. One group received a dosage of 10 mg/kg of the body weight of the sample tested, another group received 100 mg/kg, and the remaining group received 1000 mg/kg. The rats were observed for 24 h.
In the second phase, four doses (2000, 3000, 4000, and 5000 mg/kg body weight) of the test samples were administered. For the extract, 4 rats were used, each receiving one of the specified test doses. Similarly, for each of the 3 fractions, 4 rats were used, with each rat receiving one of the test doses, as was done with the extract. The rats were observed for 24 h.
Haematological Studies
The animals used for the study were grouped into sets of 17, each containing 5 animals. They were injected with 2.5 mg/kg of Oxaliplatin intraperitoneally for 3 weeks, ensuring that the cumulative dose for each animal did not exceed 30mg/kg. Oxaliplatin, a spleen toxicant, was used to induce splenomegaly. The animals underwent a 28-day treatment period with the samples. Hematological parameters such as Packed Cell Volume, Red Blood Cell count, and Haemoglobin were analyzed pre-induction, post-induction, and post-treatment using clinical laboratory standard procedures. Pre-induction blood analysis was conducted before the administration of Oxaliplatin. Post-induction blood analysis was performed after the induction of splenomegaly using Oxaliplatin. Post-treatment blood analysis took place after 28 days of treatment with the test samples. The animal was treated as follows for 28 days:
- Group 1 received distilled water.
- Group 2 received chemiron (standard drug) (5 mg/kg body weight)
- Group 3 received a low dose of the extract (100 mg/kg body weight)
- Group 4 received a medium dose of the extract (250 mg/kg body weight)
- Group 5 received a high dose of the extract (500 mg/kg body weight)
- Group 6 received a low dose of the n-hexane fraction (100 mg/kg body weight)
- Group 7 received a medium dose of the n-hexane fraction (250 mg/kg body weight)
- Group 8 received a high dose of the n-hexane fraction (500 mg/kg body weight)
- Group 9 received a low dose of the butanol fraction (100 mg/kg body weight)
- Group 10 received a medium dose of the butanol fraction (250 mg/kg body weight)
- Group 11 received a high dose of the butanol fraction (500 mg/kg body weight)
- Group 12 received a low dose of the ethyl acetate fraction (100 mg/kg body weight)
- Group 13 received a medium dose of the ethyl acetate fraction (250 mg/kg body weight)
- Group 14 received a high dose of the ethyl acetate fraction (500 mg/kg body weight)
Statistical Analysis
Data obtained from the study were analyzed using Statistical Packing for Social Sciences (SPSS-25). Results were expressed as mean ± SEM. Raw data were subjected to one-way analyses of variances (ANOVA) followed by post hoc Tukey’s test. p< 0.05 was considered statistically significant.
Result
Phytochemical Analysis
The result of the qualitative phytochemical analysis is presented in Table 1. The phytochemical present was indicated with a positive (+) sign, while the ones absent were indicated with a negative (-) sign.
| S/N | Phytoconstituents | Result |
|---|---|---|
| 1 | Tannins | + |
| 2 | Saponins | + |
| 3 | Flavonoids | + |
| 4 | Alkaloids | + |
| 5 | Terpenoids | + |
| 6 | Steroids | + |
| 7 | Quinones | + |
| 8 | Cardiac glycosides | + |
| 9 | Anthraquinone glycoside | _ |
| Note: (+) means present and (-) means absent. | ||
Acute Toxicity
No death was recorded after 24 h of administration of the various doses of the extract and fractions in both phase 1 and phase 2 of the study.
Pharmacological Assay
Packed Cell Volume
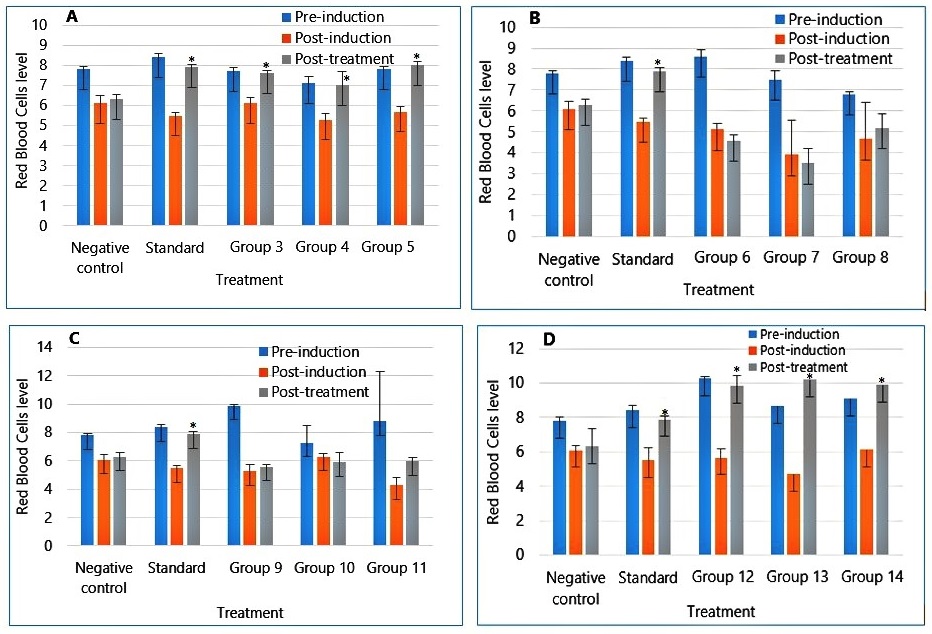
Figure 1 represents the Packed Cell Volume assay results. The result shows the packed cell volume levels in the treated rats, pre-induction, post-induction, and post-treatment at different concentrations of the extract and fractions. It can be observed that the extract and the ethylacetate fraction caused the PCV to return to the level it was prior to induction.
Red Blood Cell
Figure 2 displays the Red Blood Cell assay results for treated rats at various concentrations of the extract and fractions. Both the extract and ethylacetate fraction led to a return of Red Blood Cell levels to pre-induction values.
Haemoglobin
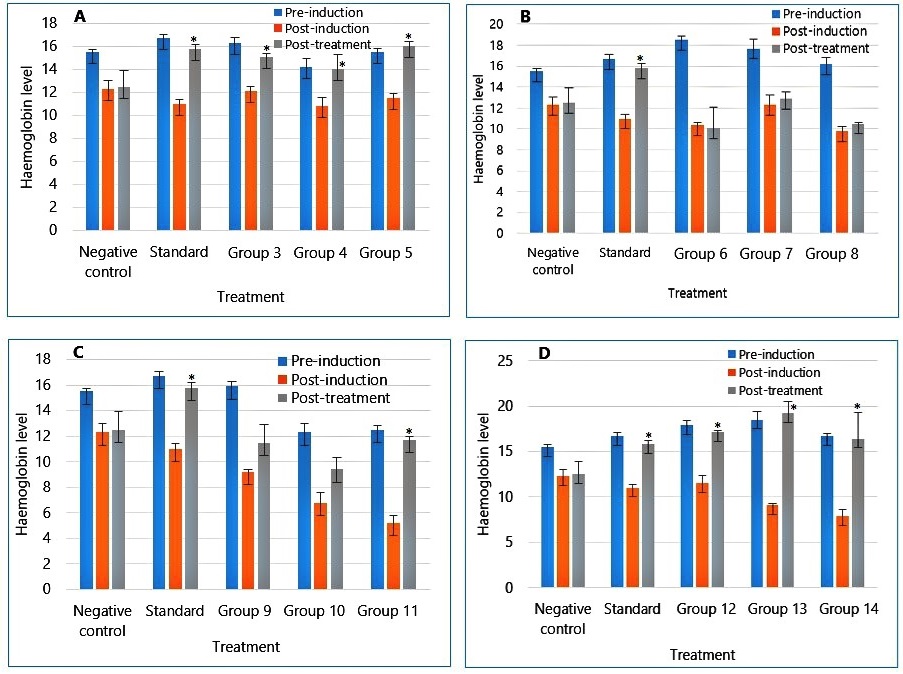
Figure 3 presents Haemoglobin assay results for treated rats at various concentrations of the extract and fractions. Both the extract and ethylacetate fraction restored Haemoglobin levels to pre-induction values.

Discussion
The phytochemical analysis of the methanolic extract of I. gabonensis unveiled the presence of numerous bioactive compounds. Identified compounds include tannins, saponins, flavonoids, alkaloids, terpenoids, steroids, quinones, and cardiac glycosides. Tannins, recognized as polyphenolic compounds, are esteemed for their antioxidant and antimicrobial properties (18). Saponins exhibit diverse biological activities encompassing anti-inflammatory, anticancer, and antimicrobial effects (19). Flavonoids, a diverse compound group, hold antioxidant, anti-inflammatory, and anticancer properties (19). Alkaloids, nitrogen-containing compounds, commonly demonstrate pharmacological activities like analgesic and antimicrobial effects (19). Terpenoids, a vast class of compounds, showcase diverse biological activities, including antimicrobial, anti-inflammatory, and anticancer properties (18). Steroids, classified as lipids, play crucial roles in various physiological processes (19). Quinones, aromatic compounds, have been recognized for their antimicrobial and anticancer activities (18). Finally, cardiac glycosides, natural compounds, have historical use in the treatment of heart diseases (19)
The phytochemicals found in I. gabonensis have been linked to diverse health benefits. For instance, supplementation with I. gabonensis seed extract has demonstrated positive effects on anthropometric and cardiovascular outcomes. These include reductions in body weight, waist circumference, total cholesterol, LDL cholesterol, and triglycerides (20). Moreover, I. gabonensis has exhibited antimicrobial activity against various bacteria and fungi (18, 21) and has shown potential as an analgesic agent (21). Studies have hinted at the anti-obesity effects of I. gabonensis through the modulation of adipogenesis and metabolic parameters (22). Additionally, the presence of tannins in Irvingia gabonensis may contribute to its antioxidant properties (18).
The findings of the acute toxicity study are consistent with previous studies that have also demonstrated the safety of I. gabonensis. For example, a study investigated the anti-diarrheal and anti-ulcer properties of Irvingia gabonensis in rats. The study found no deaths after administering the extract up to 1600 mg/kg (23). The study on Wistar rats exposed to sodium fluoride-induced toxicity found that Irvingia gabonensis juice exhibited renal and hepato-protective effects, with no recorded deaths among the observed rats (24). These studies support the conclusion that I. gabonensis has low acute toxicity and is safe for administration at the tested doses. The absence of deaths in the acute toxicity study suggests that I. gabonensis does not pose immediate lethal effects within the observed time frame. It is important to note that these findings are specific to the tested doses and the duration of observation in the acute toxicity study. Further research may be needed to assess the long-term effects and potential toxicity of I. gabonensis at higher doses or with prolonged exposure.
In the Pharmacological assay, the administration of Oxaliplatin caused a hematological toxicity that decreased the level of packed cell volume. Red blood cells and Haemoglobin. Oxaliplatin, a third-generation platinum-based chemotherapy, is commonly administered in combination with leucovorin and fluorouracil (FOLFOX) for the neoadjuvant and adjuvant treatment of colorectal cancer (CRC) (25). Oxaliplatin is associated with a range of side effects, including fatigue, nausea, peripheral neuropathy, and hematological toxicity (26). When Oxaliplatin is added to fluorouracil and leucovorin for the treatment of CRC patients, there is a significant increase in all-grade anaemia compared to treatment with fluorouracil and leucovorin alone (25).
Furthermore, CRC patients often present with pre-operative cancer-related anaemia (27), which has been identified as a contributing factor to poor disease progression and recovery (28). Generally, the anaemia observed in these patients is mild, with most experiencing grade 1-2 anaemia, and only a small percentage experiencing grade 3-4 anaemia (25). Additionally, there have been reported case reports of oxaliplatin-induced immune-mediated haemolytic anaemia (29). In more recent studies, it has been shown that around 90% of patients experience an increase in the size of their spleens [6], and approximately 25% of those receiving oxaliplatin exhibit splenomegaly, defined as a spleen size increase of 50% or more (30).
The administration of the extract and fractions caused the reversal of the haematological toxicities. The result of the packed cell volume presented in Figure 1 shows that the administration of the extract and the ethylacetate fraction at different concentrations caused a significant concentration-dependent increase in the packed cell volume compared to the standard drug.
The extract and fractions of ethylacetate fraction caused an increase in the red blood cell level post-treatment, similar to that of the standard drug used. This can be observed in Figure 2.
Haemoglobin level also increased significantly in level on the extract and ethylacetate fractions administration, as seen in Figure 3. Butanol fraction also led to a slight increase in the haematological parameters. The Hexane fraction treated groups showed no significant effect on the haematological parameters, as shown in Figure 1B, 2B, and 3B.
A previous study titled 'Toxicological Assessment of I. gabonensis Leaf Extracts in Albino Rats: A Comparative Study between Aqueous and Ethanol Extraction Methods' revealed that the aqueous and ethanol extracts of I. gabonensis had no significant impact on the RBC and PLT counts in female albino rats. However, increasing doses of the extract correlated with elevated PCV, Hb concentration, MCV, and MCHC values. Remarkably, MCV values were notably higher in the control group compared to the treatment groups, suggesting a potential protective effect of the extract against anaemia in female albino rats. These findings indicate a potentially positive influence of Irvingia gabonensis leaf extracts on erythrocyte and hemoglobin levels in both male and female albino rats (31). The use of methanol in the extraction for this present study may have caused the extraction of phytochemicals that significantly impacted the RBC.
Some other studies have also demonstrated the ability of natural products to significantly cause an increase in hematological parameters. In a study examining the impact of the aqueous leaf extract of Ocimum gratissimum on hematological parameters in rats, the authors noted that the oral administration of O. gratissimum resulted in elevated levels of RBC, PCV, Hb, platelet count, and neutrophils (32). Another study focused on the effect of Solanum villosum (mill. ) extract and its silver nanoparticles on the hematopoietic system of rats with diethylnitrosamine-induced hepatocellular carcinoma. We observed that the oral administration of ethanolic Solanum villosum and its silver nanoparticles led to increased concentrations of Hb, PCV, RBC, MCV, MCH, MCHC, and PLTS while reducing WBC levels in diethylnitrosamine-induced HCC (33). Additionally, in a study centered on the erythropoietic potential of Parquetina nigrescens in a cephalosporin-induced anaemia model, it was concluded that the treatment of anaemic animals with P. nigrescens, particularly at a dosage of 250 mg/kg BW, significantly boosted RBC levels by 35.8%, Hb by 25.2%, PCV by 39.4%, cobalt by 70.9%, and vitamin C by 82.6% (p< 0.05) (34). These and numerous other studies have collectively provided evidence supporting the effectiveness of natural products in positively impacting hematological health.
The phytochemicals detected in the methanolic extract of I. gabonensis, as shown in Table 1, have also been seen in plants that possess anti anaemic effects (35, 36). This provides a correlation between this study and previous studies.
Conclusion
The results of this study indicate that methanolic leaf extract and ethyl acetate fraction of I. gabonensis may be beneficial in splenomegaly-induced anaemia by boosting PCV level by 92.6 – 103.7%, RBC level by 96.0 – 117.9%, and HB level by 92.6 – 103.7%. The phytochemicals present are similar to those reported in previous studies in plants with anti-anaemic effects. Further investigations are being conducted to isolate the phytochemical compounds found in the ethyl acetate fraction of the methanolic leaf extract of I. gabonensis.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
Ethical approval for the study was obtained from the Enugu State University of Science and Technology Animal Ethics Committee, with the approval number of ESUT/AEC/0113/AP097.
Funding Information
Not applicable.
References
- Nguyen Y, Stirnemann J, Belmatoug N. La maladie de Gaucher : quand y penser ? [Gaucher disease: A review]. Rev Med Interne. 2019 May; 40(5):313-322.
- Kang DW, Kim SH. Clinical aspects of splenomegaly as a possible predictive factor of coronary artery changes in Kawasaki disease. Cardiol Young. 2019 Mar;29(3):297-302
- Gala AR, Surapaneni T, Aziz N, Kallur SD. A Review of Outcomes in Pregnant Women with Portal Hypertension. J Obstet Gynaecol India. 2018 Dec;68(6):447-451
- Chapman J, Goyal A, Azevedo AM. Splenomegaly. [Updated 2023 Jun 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430907/
- Spivak J.. Polycythemia vera: myths, mechanisms, and management. Blood 2002;100(13):4272-4290.
- Bolognesi M., Merkel C., Sacerdoti D., Nava V., & Gatta A.. Role of spleen enlargement in cirrhosis with portal hypertension. Digestive and Liver Disease 2002;34(2):144-150
- Abubakar AM, Rukayyah BI. Review of Medicinal Plants with Antianaemic Activity Found in Nigeria. Scholars International Journal of Biochemistry 2019; 2(8): 225-229
- Sun J, Chen P. UHPLC-MS/MS analysis of polyphenols in Irvingia gabonensis (African wild mango) seeds and their potential antioxidant properties. J Agric Food Chem. 2012;60(41):10456-10465.
- Lowe J, Pokorny M, Dykes L. Irvingia gabonensis: a review of its traditional uses, phytochemistry, and pharmacology. Pharmaceutical Biology 2000;38(1):5-12
- Okolo CO, Ewere EG, Okolie NP, Ndem JI, Oyebadejo SA. Immunological and hematological effects of Irvingia gabonensis stem bark in sodium arsenite-exposed rats. GSC Biological and Pharmaceutical Sciences 2021; 15(01): 027–037
- Oluwafemi AO, Basiru OA, Babatunji EO, Adebola BO. Hematological Properties of Irvingia Gabonensis in Male Adult Rats. Journal of Pharmaceutical and Scientific Innovation 2014; 3:434-436
- Ngondi JL, Oben JE, Minka SR. The effect of Irvingia gabonensis seeds on body weight and blood lipids of obese subjects in Cameroon. Lipids Health Dis. 2005 May 25;4:12
- Nosiri I, Abdu-Aguye I, Hussaini MI, Abdurahaman E. Leaf Extracts Of Irvingia gabonensis Increase Urine Output And Electrolytes In Rats. The Internet Journal of Alternative Medicine. 2011 8;2
- Fajimi O, Sarumi MB, Olayode MN, Gamra EO, Sanusi SI. In vitro propagation of Irvingia gabonensis. African journal of biotechnology. 2007; 6(8): 976-978
- Evans WC. Trease and Evans Pharmacognosy. 15th Edition. London: Elsevier Science Ltd 2002.
- Onyegbule FA, Ilouno IO, Eze PM, Abba CC, Chigozie VU. Evaluation of the Analgesic, Anti-Inflammatory and Antimicrobial Activities of Leaf Extracts of Breynia nivosa. Chem Sci Rev Lett. 2014; 3(12):1126-1134
- Erhirhie EO, Ihekwereme CP, Ilodigwe EE. Advances in acute toxicity testing: strengths, weaknesses and regulatory acceptance. Interdiscip Toxicol 2018; 11(1): 5–12
- Kuete V, Wabo G, Ngameni B, Mbaveng A, Metuno R, Etoa F. et al.. Antimicrobial activity of the methanolic extract, fractions, and compounds from the stem bark of irvingia gabonensis (ixonanthaceae). Journal of Ethnopharmacology 2007;114(1):54-60
- Odion E, Obarisiagbon P, Akpofure H, Odiete E. Phytochemical identification and analgesic potential of the seed extract of Irvingia gabonensis. Asian Journal of Applied Chemistry Research 2022:34-42
- Lee J, Chung M, Fu Z, Choi J, Lee H. The effects of Irvingia gabonensis seed extract supplementation on anthropometric and cardiovascular outcomes: a systematic review and meta-analysis. Journal of the American College of Nutrition 2019;39(5):388-396
- Olanrewaju I, Mordi R, Echeme J. Antibacterial, antifungal and anti-tubercular activities of chloroform fraction of the leaf extract of Irvingia gabonensis (African bush mango). Anti-Infective Agents 2020;18(2):109-114
- Ngondi J, Etoundi B, Nyangono C, Mbofung C, Oben J. Igob131, a novel seed extract of the West African plant Irvingia gabonensis, significantly reduces body weight and improves metabolic parameters in overweight humans in a randomized double-blind placebo-controlled investigation. Lipids in Health and Disease 2009;8(1)
- Raji Y, Ogunwande I, Adesola J, Bolarinwa A. Anti-diarrhegenic and anti-ulcer properties of Irvingia gabonensis in rats. Pharmaceutical Biology 2001;39(5):340-345
- Emejulu A, Alisi C, Asiwe E, Igwe C, Nwogu L, Onwuliri V. Renal and hepato-protective effects of Irvingia gabonensis juice on sodium fluoride-induced toxicity in Wistar rats. Journal of Clinical Toxicology 2016;06(02)
- André T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, Topham C, Zaninelli M, Clingan P, Bridgewater J, Tabah-Fisch I, de Gramont A; Multicenter International Study of Oxaliplatin/5-Fluorouracil/Leucovorin in the Adjuvant Treatment of Colon Cancer (MOSAIC) Investigators. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004 Jun 3;350(23):2343-51
- Hong YS, Nam BH, Kim KP, Kim JE, Park SJ, Park YS, Park JO, Kim SY, Kim TY, Kim JH, Ahn JB, Lim SB, Yu CS, Kim JC, Yun SH, Kim JH, Park JH, Park HC, Jung KH, Kim TW. Oxaliplatin, fluorouracil, and leucovorin versus fluorouracil and leucovorin as adjuvant chemotherapy for locally advanced rectal cancer after pre-operative chemoradiotherapy (ADORE): an open-label, multicentre, phase 2, randomized controlled trial. Lancet Oncol. 2014 Oct;15(11):1245-53.
- Väyrynen JP, Tuomisto A, Väyrynen SA, Klintrup K, Karhu T, Mäkelä J, Herzig KH, Karttunen TJ, Mäkinen MJ. Pre-operative anemia in colorectal cancer: relationships with tumor characteristics, systemic inflammation, and survival. Sci Rep. 2018 Jan 18;8(1):1126
- Tokunaga R, Nakagawa S, Miyamoto Y, Ohuchi M, Izumi D, Kosumi K, et al. The impact of pre-operative anaemia and anaemic subtype on patient outcome in colorectal cancer. Colorectal disease: the official journal of the Association of Coloproctology of Great Britain and Ireland. 2019;21(1):100–9
- Kim MJ, Han SW, Lee DW, Cha Y, Lee KH, Kim TY, et al. Splenomegaly and Its Associations with Genetic Polymorphisms and Treatment Outcome in Colorectal Cancer Patients Treated with Adjuvant FOLFOX. Cancer research and treatment: official journal of Korean Cancer Association. 2016;48(3):990–7
- Overman MJ, Maru DM, Charnsangavej C, Loyer EM, Wang H, Pathak P, Eng C, Hoff PM, Vauthey JN, Wolff RA, Kopetz S. Oxaliplatin-mediated increase in spleen size as a biomarker for the development of hepatic sinusoidal injury. J Clin Oncol. 2010 May 20;28(15):2549-55
- Okereke S, Edom V, Nwaogwugwu C, Aaron C, Oko I, Obinna I, Ekechukwu G, Alugbuo U, James U. Toxicological Assessment of Irvingia gabonensis Leaf Extracts in Albino Rats: A Comparative Study between Aqueous and Ethanol Extraction Methods. Advances in Biological Chemistry 2023; 13: 143-170
- Ofem O, Ani E, Eno A. Effect of aqueous leaves extract of Ocimum gratissimum on hematological parameters in rats. Int J Appl Basic Med Res. 2012 Jan;2(1):38-42.
- Venkatesh R, Kalaivani K. Effect of Solanum Villosum (Mill.) Extract and its Silver Nanoparticles on Hematopoietic System of Diethylnitrosamine-induced Hepatocellular Carcinoma in Rats. Innovare Journal of Health Sciences. 2017; 5(1):13-16
- Ighodaro OM, Asejeje FO, Adeosun AM, Ujomu TS, Adesina FC, Bolaji KT. Erythropoietic potential of Parquetina nigrescens in cephalosporin-induced anaemia model. Metabol Open. 2020 Oct 10;8:100064
- Fagbohoun L, Nonvidé G, Orou A, Houngbèmè A, Sakirigui A, Gunin F, Gbénou J. Anti-Anaemic Activity and Potential Toxicity of Extracts of Four Tinctorial Plants Used in the Treatment of Anemia in Benin: Gossypium barbadense, Sorghum bicolor, Hibiscus sabdariffa and Justicia secunda. American Journal of Plant Sciences 2022; 13: 1460-1477
- Adeyemi SB, Abidakun OM, Azeez R, Chijindu PCI, Oyebanji O. Phytochemical and Nutritional Composition of Commonly used Medicinal Plants for the Treatment of Anaemia in Kwara State, Nigeria. Annals of West University of Timişoara, ser. Biology 2018; 21 (2):165-174
