REVIEW
An Updated Review of Rubus ellipticus (an Edible Shrub), Its Bioactive Constituents and Functional Properties
Academic Editor: James H. Zothantluanga
Sciences of Phytochemistry|Vol. 1, Issue 2, pp. 76-86 (2022)
Received
Oct 25, 2022Revised
Nov 12, 2022Accepted
Nov 12, 2022Published
Nov 22, 2022
Abstract
Introduction
Medicinal plants as natural alternatives in modern civilization are expanding daily and gaining popularity worldwide (1). Modernized traditional health care is currently hampered by significant scientific advancements, chronic disease, resistant infections, degenerative disorders of aging, autoimmune disorders, and huge problems from risky medicine. Additionally, these non-allopathic medical approaches are still used by 70% of India's 1.1 billion people (2). It is inevitably necessary to continue looking for newer traditional medicine sources and to check existing ones for new therapeutic uses (3).
Nobel Prize winner Ernst Boris Chain, who developed the powerful natural antibiotic penicillin, published a motivational piece titled "The quest for new biodynamic substances". In 1967, he wrote, "In China and India, there has been an extensive drive aimed at the systemic study of medicinal plants traditionally used in these countries in folklore medicine; this has failed, so far, to bring to light new classes of compounds with interesting pharmacologic activities. As far as drug research is concerned, therefore, we cannot expect many major surprises to come from the study of plant constituents" (2).
India is home to the Himalayas, the Western Ghats, the Nicobar Islands, and the North-Eastern rain forests, which together make up four of the world's total 34 biodiversity hotspots (4). With about 1,350 species, Rubus is a vast and diversified genus of flowering plants in the rose family, Rosaceae, subfamily Rosoideae, and it is extensively dispersed in forests at high elevations, including those in the Himalayas and the Nilgiris area (5). A huge evergreen shrub that flourishes in the wild, Rubus ellipticus Smith (also known as R. ellipticus), a member of the Rosacea is a significant ethnomedicinal plant. The berries are also consumed globally for their flavour or as a reservoir of phenolics, tannins, and flavonoids, which are natural remedies (6). It has traditionally been used to treat gastralgia, ulcer, diarrhea wound healing, antifertility, antimicrobials, analgesics, and epilepsy (7, 8). Numerous bioactive components from the species have been shown to have a wide range of pharmacological properties that are health-promoting (9). This review will serve as a springboard for subsequent research and focuses on the species' potential to produce pharmaceutical, health-improving, and nutraceutical products.
Taxonomical Classification
- Domain: Eukaryota
- Kingdom: Plantae
- Subkingdom: Tracheobionta
- Phylum: Spermatophyta
- Subphylum: Angiospermae
- Class: Dicotyledonae
- Subclass: Rosidae
- Order: Rosales
- Family: Rosaceae
- Genus: Rubus
- Species: Rubus ellipticus
Geographical Distribution
R. ellipticus is a weedy wild raspberry that thrives in moist forests and spreads quickly in both bright open spaces and dense rainforests. As a result, it is common in forests at high elevations, including those in the Himalayas and the Nilgiris region. In addition to this, plants are extremely resilient and can build a natural defense mechanism that allows them to flourish while being exposed to both biotic and abiotic environmental stimuli (5). It is one of the top 100 most invasive species in the world (10). It grows between 300 and 2600 meters above sea level along highways, hillsides, thickets, slopes, mountain valleys, and sparsely populated woodlands (11). The plant is found in an area with a 2000–6500 mm per annum rainfall range. The plant was initially allowed to grow naturally in Queensland and is indigenous to Southern Asia, South East Asia, South Western China, Sikkim, Myanmar, Bhutan, Laos, Pakistan, Sri Lanka, Nepal, Philippines, Vietnam, Thailand, and India (distributed in the Himalayan foothills, Nilgiri, and Peninsular Hills region) (12). The plant is just found in a limited region of the Central coastal region of New South Wales and Southern Queensland in Australia (11). In China, it grows in frost-tolerant habitats such as wet lowland regions next to pond banks and heavily forested to drier montane habitats at elevations of 1000–2500 m up to 3000 m. In India, it is found in Assam, Sikkim, Tamil Nadu, Kerala, and Maharashtra state. The plant's blooming season lasts from March to April, while the fruiting season, which produces golden yellow fruits, lasts from April to May (13).
Morphological Studies
R. ellipticus is a thorny, 1-3-meter-tall shrub. Stems are pubescence consisting of sparse, curved prickles, dense, purplish-brown glandular hairs, and pubescence that is brown or purplish-brown in colour. Stipules are linear, 7–11 mm long, pubescent, and covered in intermixed glandular hairs. The leaves are imparipinnate and trifoliate. The petiole is 2-6 cm long, 2-3 cm long make up the petiolule of the terminal leaflet, and the lateral leaflets are subsessile. Leaflets' blades are obovate or elliptic. In compared to the lateral leaflets, the terminal leaflet is larger, has a base that is rounded, a margin that is unevenly minutely sharply serrated, and an apex that is acute, abruptly pointed, shallowly cordate, or sub-truncate. Along the midvein, the veins are pubescent and impressed adaxially. Purplish red pubescent bristles cover the rachis and petiolule. Terminal inflorescences with thick glomerate racemes. Sepals that are erect, oblong, and abaxially densely covered in yellowish grey tomentose; flowers with a calyx that is abaxially pubescent, intermixed with yellowish tomentose, and sparsely bristly. Petals those are pink or white, spatulate, longer than the sepals, with a densely hairy base and a premorse edge. The ovary is hairy, and the styles are glabrous and a little bit longer than the stamens. Golden-yellow, sub-globose aggregate fruit with triangular-ovoid pyrenes and pubescent drupelets at the apex. The thalamus of the R. ellipticus fruit is nipple-shaped and measures 6 mm long and 7 mm diameter at the base. The fruit weighs 444 mg, has a volume of 567 μL, is yellow, and is very easily detachable from the thalamus and falls when it reaches maturity (13, 14).
Folklore Medicinal Uses
R. ellipticus plant possesses great importance in Tibetan traditional medicine. The fruit has a laxative effect, and the inner bark is used for a variety of purposes including as a renal tonic and an anti-diuretic. Typhoid fever is traditionally treated using the entire plant since it has astringent properties. Extracting root juice has been used to treat a variety of ailments, including fever, gastrointestinal problems (including infant colic when the young shoots are also used), diarrhoea, and dysentery, and applying root paste to wounds speeds up healing (8). Additionally, the fruit juice is used to cure colds, sore throats, colic, and fevers. When the senses are impaired and a person has vaginal or seminal discharge, the inner bark is said to be beneficial (15). As the fruit is fibrous, it also aids in digestion. It is used as a diuretic and a diaphoretic in the summer to stimulate sweating. R. ellipticus roots and young shoots are used to relieve colic discomfort. It has traditionally been used to treat ulcers, gastralgia, wound healing, diarrhoea, antifertility, antibacterial, and analgesic (7). There have been claims that various portions of the plant are helpful for illnesses like diabetes, diarrhoea, gastritis, dysentery, epilepsy, wound healing, anti-fertility, antibacterial, analgesic, and renal tonic. (7, 8, 13).
As one of Manipur's significant ethnomedicinal plants, R. ellipticus root bark has long been used by the Naga tribe in Manipur to treat fever. For fever treatment, the Nagas advise drinking a root bark decoction twice daily. In addition, the root bark is utilized for severe headaches, shattered bones, emmenagogues, abortifacients, and dysentery (16, 17). The Lepcha tribe in North Sikkim, India, uses R. ellipticus to treat a variety of illnesses. Unexpected stomach ache is relieved by chewing raw fresh shoots. Children were given a root decoction to relieve their heated stomachs. Young fruit paste is eaten in cases of gastritis, diarrhea, and dysentery, whereas ripe fruits are used in cases of constipation as they posses laxative action (8). In the Tanahun District of Western Nepal, the root juice used to treat urinary tract infections and its fruits, which are tasty, were listed among the top ten wild edible medicinal plants (18, 19). In different parts of Nepal, it is used to treat colds, typhoid fever, and several other alignments (19). It is normally advised to use it for the full nine months of pregnancy and can be used in the last stages of pregnancy to aid with delivering. The muscles in the pelvis and uterus seem to be toned by raspberry leaf tea (20).
Phytochemical Studies
Fruits
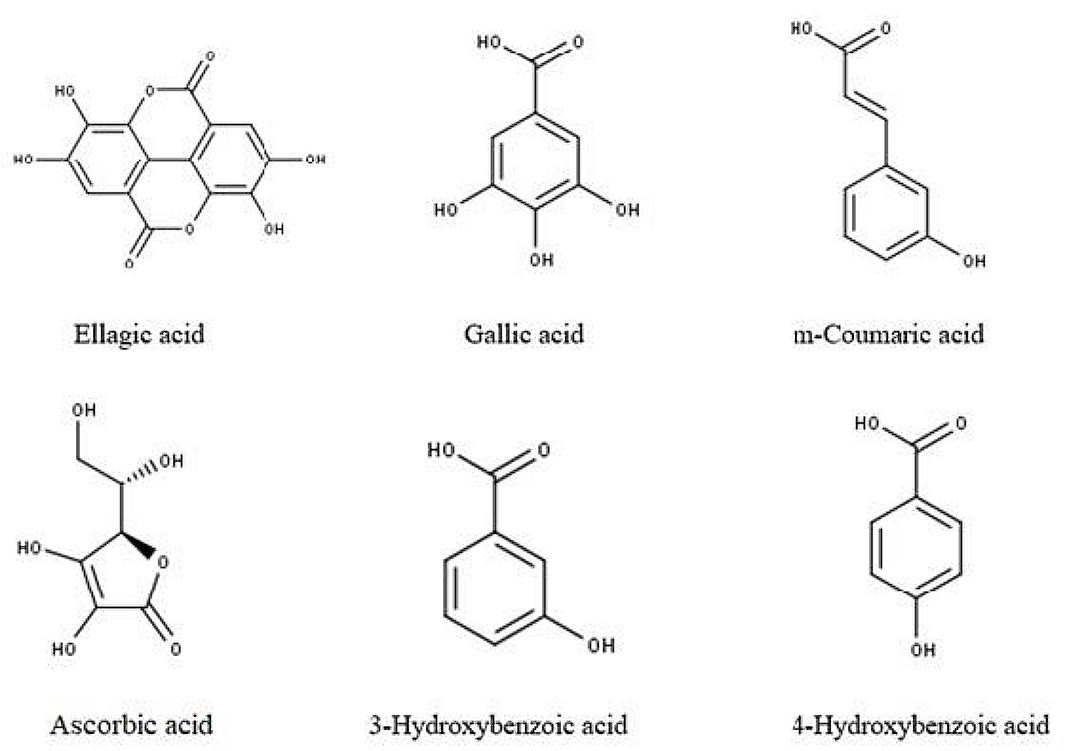
In various studies, the phytoconstituents from the fruit extract of R. ellipticus were reported;; the extract components included - ellagic acid, 3-Hydroxybenzoic acid, 4-Hydroxybenzoic acid, m-Coumaric acid, ferulic acid, vanillic acid, trans-Cinnamic acid, phloridzin, cyanin, delphinidin (21), chlorogenic acid, gallic acid, catechin, caffeic acid, β-carotene, ascorbic acid (22, 23), 4-Dimethylamino-2,2,6,6-tetramethylpiperidinde; 3-piperidinecarboxamide, N,N-diethyl-; 1-Octacosanol; 3,3-Diethoxypropylamine; (e)-9,11-dodecadien-1-ol; (2-(4-Hydroxy-phenyl)-ethyl)-carbamic acid ethyl ester; 2,4-Bis(1,1-dimethylethyl), Benzenepropanoic acid, 3,5-bis(1,1-dimethylethyl)-4-hydroxmethyl Ester; n-Hexadecanoic acid; 2-Bromopropionic acid, tridecyl ester; glucurolactone; Carbamic acid, hydroxy-, ethyl ester; Acetamide, N-(3-(3-dimethylaminopropylamino)propyl)-2-hydroxyimino-2-phenyl; 1,3-Propanediamine, Nʹ-(3-(dimethylamino) propyl)-NN dimethyl; 2-Propanamine, N-methyl-1-(4-(2-(1-piperidyl)ethoxyphenyl); 3-Piperidinamine, 1-ethyl-; 1-(Diethylamino)ethylidenimino)sulfur pentafluride; 7,9-Dimethyl-1,4-dioxa-7,9-diazacycloundecan-8-one; 4-Fluoro-n-(2-(4-methyl-piperazine-1-carbonyl phenyl benzamide); 1,1-(Diethylcarbamoyl)succinimide); 4(Equat)-N-butyl-1,2(axial)-dimethyl- ransdecahydroquinol-4-ol (24-26). Figure 1 shows the structures of some of the aforementioned chemical components.
Leaf
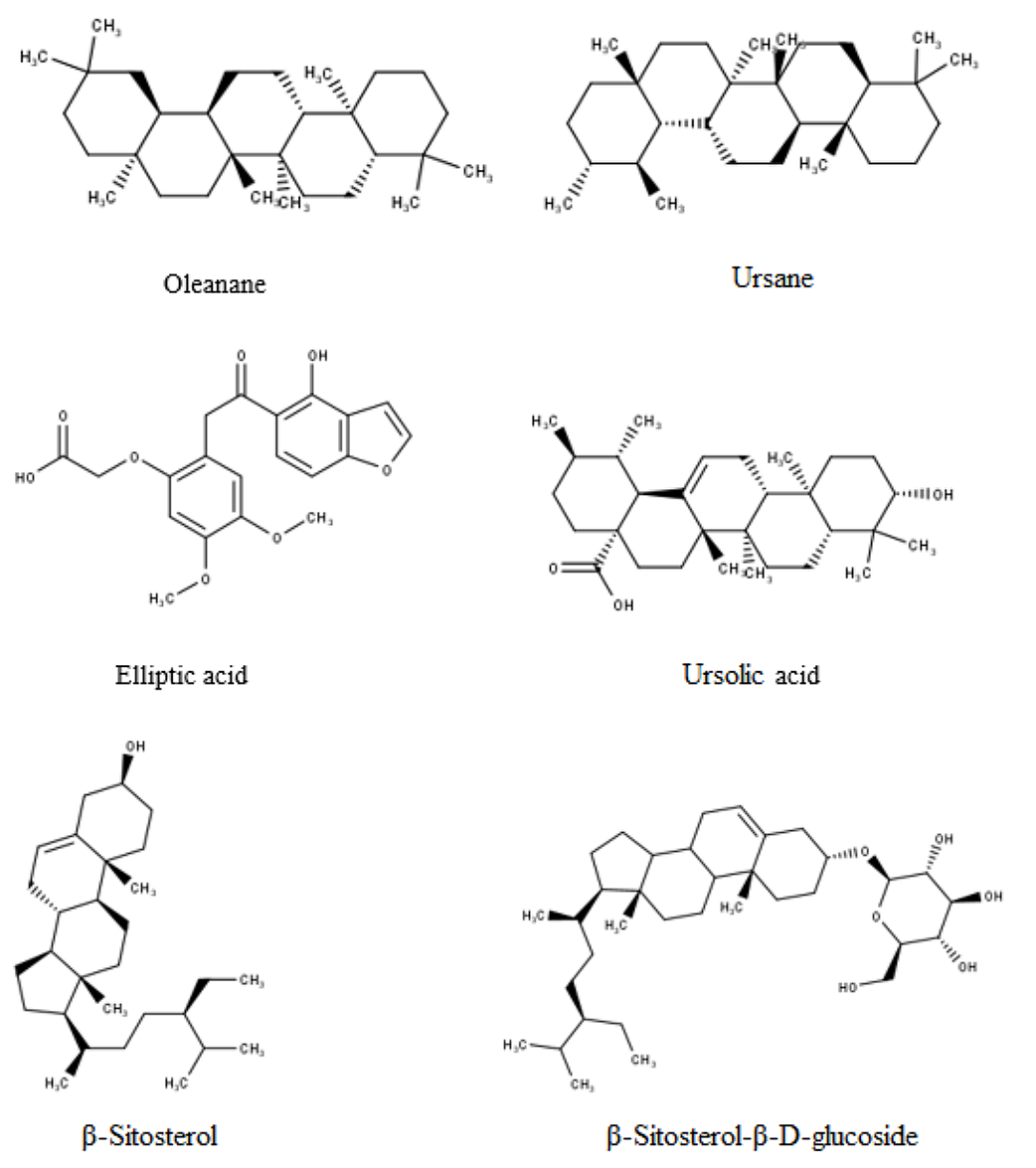
The leaf extract of R. ellipticus has been shown to contain several, different compounds, including, ursane, oleanane (27), elliptic acid (28), ursolic acid, 1-Octacosanol, β-Sitosterol, β-Sitosterol-β-D-glucoside, octacosanic acid (29). The structure of some of the chemical components is seen in Figure 2.
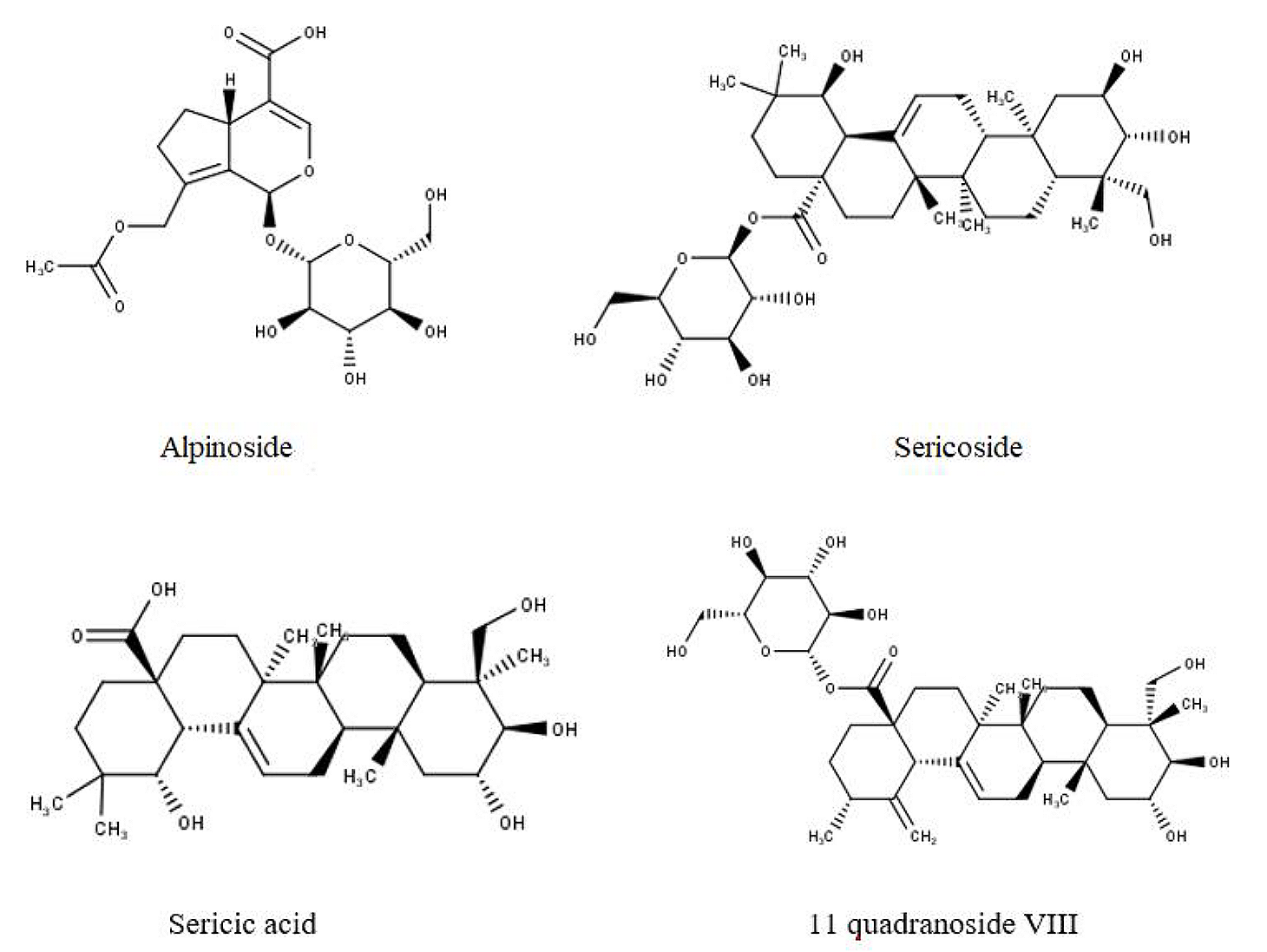
Li et al. (30), conducted a study on the chemical elements obtained from R. ellipticus' roots and detected several chemical constituents such as - 2R,3,23-trihydroxyurs-12,19-dien-28-oic acid 28-O-d-glucopyranoside; 2R,3,23-trihydrox-yurs-12,18-dien-28-oic acid 28-O-dglucopyranoside; alpinoside; 11 quadranoside VIII; sericoside; sericic acid; buergericic acid; pinfaensin; rosamutin; kaji-ichigoside F1; trachelosperoside A1; nigaichigoside F1 and F2; pedunculoside; sauvissimoside R1; 4-Epinigaichigoside F1; ziyuglycoside; euscaphic acid; 1R,2R,3,19R-tetrahydroxyurs-12-en-28-oic acid; 19R-hydroxyasiatic acid; and 2R,3,19R-trihydroxyurs-12-en-23,28-dioic. Figure 3 depicts some of the chemical ingredients together with their structures.
Whole Aerial Parts
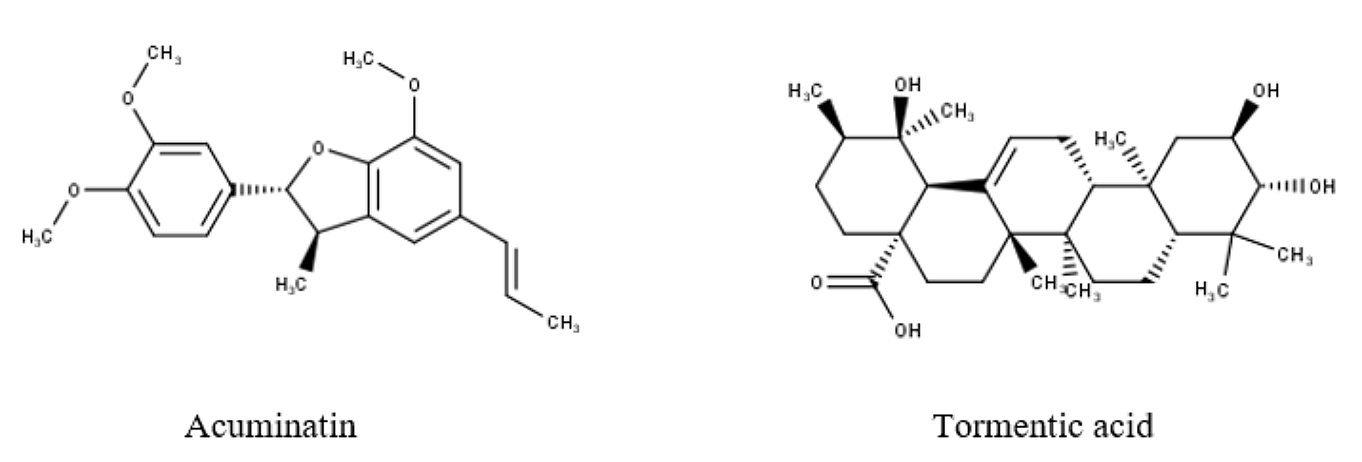
Several studies documented the phytoconstituents from R. ellipticus fruit extract; according to studies conducted by numerous researchers, include - 3-β-Hydroxy-urs-12; acuminatic acid (acuminatin); 18-Dien-28-oic acid-3-0(β-d-glucopyranosyl) (1→4)-α-larabinopyranoside; tormentic acid; and β-Sitosterol-β-d-glucoside (29, 31). In Figure 4, certain chemical components are listed together with their structures.
Nutritional Value of R. ellipticus
Understanding the nutritional composition of R. ellipticus is fascinating and important to understand its function in the human diet. The nutritional composition of the fruits of R. ellipticus is listed in Table 1.
| Nutrient composition | % | References |
|---|---|---|
| Fibres | 2.35 ± 0.05 | (15) |
| Protein | 4.37 ± 0.52 | (6, 15) |
| Lipids | 2.73 ± 0.06 | (6) |
| Fat | 0.96 ± 0.20 | (15) |
| Carbohydrates | 86.4 ± 0.38 | (6) |
| Moisture (fresh weight) | 66.36 ± 0.58 | (6) |
| Ash | 2.97 ± 0.01 | (6, 15) |
| Energy (Kcal) | 374.0 ± 1.56 | (6) |
| Sodium | 89.43 ± 0.01 | (6) |
| Potassium | 680.16 ± 1.27 | (6, 15) |
| Calcium | 450.1 ± 0.22 | (6, 15) |
| Nitrogen | 700 ± 0.08 | (6, 15) |
| Magnesium | 450.1 ± 0.22 | (6) |
| Phosphorus | 1.26 ± 0.001 | (6) |
| Zinc | 12.77 ± 0.05 | (6) |
| Iron | 4.249 ± 0.15 | (6) |
| Copper | 0.020 ± 0.01 | (6) |
| Manganese | 1.948 ± 0.03 | (6) |
| Lead | 0.02 ± 0.18 | (6) |
| Chromium | 0.47 ± 0.19 | (6) |
Pharmacological Studies
R. ellipticus has been used is used to treat various ailments since ancient times. Various part of the plant extracts has been reported for the presence of flavonoids, phenolic compounds, steroids, saponin, and tannins. These various compounds are used as an analgesic, antipyretic, anti-inflammatory (5), antimicrobial (32), antitumor, wound healing (33), antidiabetic (34), nephroprotective (35), antioxidant, and antiproliferative (23).
Antioxidants
Antioxidants are chemicals that have the potential to donate electrons to reactive species, so helping to rid the body of oxidative species that cause chronic diseases in humans (23). And it was observed that decreased plasma antioxidant count contributed to the rising prevalence of cancer death (36).
Meda et al. (37) studied the ability of R. ellipticus to scavenge reactive oxygen species, prevent ß-carotene bleaching, and decrease ferric activity, which was examined concerning the plant's antioxidant characteristics. The acetone extract had the highest levels of ABTS cationic radical scavenging and DPPH free radical scavenging activity with value of 1072.6 mg BHAE/100 g FW and 619.3 mg CE/100 g FW respectively, while the methanol had 502.2 mg CE/100 g and that of acidic methanol had 521 mg CE/100 g of antioxidant activity. The presence of phenolics is required for the ferric reduction action (Fe3+ to Fe2+) of plant extracts, which was highest with acid acetone extracts (1389.82 mg AAE/100 g FW) and lowest with methanolic extracts (695.7 mg AAE/100 g FW) (37). Vadivelan et al. (7) carried out a study to find the presence of total phenolic compounds in Himalayan berry root extract that adds to its antioxidant activity. Also, they prepared different samples using different solvents to detect various forms of phenolic contents that result in between 21 - 225 mg/g of gallic acid. Study results revealed that the highest and lowest concentration of total phenols found in methanol and petroleum ether extract. The root extract prepared in methanol with a high total phenol content demonstrated the strongest free radical scavenging activity against DPPH and ABTS free radicals when compared to ascorbic acid. The methanolic leaves extract has better DDPH radical scavenging activity (6.96 g/mL) than BHA (4.88 g/mL) and quercetin (4.12 g/mL) but less than BHT (13.18 g/mL). George et al. (33), reported 71.08% superoxide and 66.08% nitric oxide free radicals inhibitory capacity. At two different levels of dosages of leaf extract (100 and 250 mg/kg), the antioxidant activity was dramatically boosted as measured by Glutathione activity, glutathione peroxidase, catalase activity, and superoxide dismutase (38). According to the results of the DPPH experiment conducted by Ahmad et al. (6), R. ellipticus medicinal extract was found to have the highest level of antioxidant activity i.e., 29.22 ± 0.88 mM AAE/100g fresh weight. Likewise, the study conducted by Muniyandi et al. (26) revealed that the methanolic extract of R. ellipticus had improved hydrogen donating ability (11.01 µg/mL) against stable DPPH and also showed better results (18,804.10 µM/TE) in ABTS radical scavenging activity. From various studies, alcoholic extracts of R. ellipticus were found to have better antioxidant activity than other extracts (7, 39).
Analgesic and Antipyretic
George et al. (5) compared the doses of 200 mg/kg and 400 mg/kg of methanolic leaf extract of R. ellipticus with the standard treatment Aspirin (100 mg/kg) (73.13% inhibition), and it was found that the fruit extract considerably decreased the frequency of writhing to 19.40% and 32.84%, respectively. They also measured pain reaction by Eddy's hot plate method at the dose of 400 mg/kg. At 30, 60, and 120 min of reaction time, the animal was shown to be able to resist the hot plate for 11.2, 13.6, and 7.7 s, respectively. This is comparable to the reference drug morphine (10 mg/kg), which was used as a control (7.8, 9.6, and 12.4 s). At the same dose, the extract was found to have potent antipyretic effects against Brewer's yeast-induced hyperpyrexia in rats. From the third to the seventh h after delivering the dose in yeast-induced rats, body temperatures decreased significantly, and the activity was comparable to that of the common medicine paracetamol (100 mg/kg).
Anti-Inflammatory
Triterpenoid saponins were found in the Rubus species by Li et al. (30) who also reported nine distinct categories of triterpenoids and 21 distinct categories of saponins that were further subdivided into seven categories (30). Tannins, triterpenoids, and flavonoids were discovered in the phytochemical tests conducted by Vadivelan et al. (7) on the ethanol root extract of Rubus species. The availability of these substances showed that edoema in rats generated by carrageenan at two different doses of 250 mg and 500 mg/kg had an anti-inflammatory impact whereas no impact was shown with 125 mg/kg even after 6 h and was strong after 2 h of consumption and lasted for 2-3 h. Methanolic leaf extract was found to be effective at preventing the carrageenan-induced (45.43% and 66.47%) thickening of the paws of edoema in rats at doses of 200 mg and 400 mg/kg, respectively. Similarly, when George et al. (5) compared the control Indomethacin at 10 mg/kg dose (76.52%) in croton oil-induced ear inflammation with the methanolic extract. It was seen that the ear inflammation reduced dramatically from 36.66% (at dose 200 mg/kg) to 45.78% (at dose 400 mg/kg).
Wound Healing
Research indicates that the extract should be incorporated at an optimum dose of 2 g/kg for wound healing. In one of the investigations conducted by George et al. (33) two different extract doses (100 mg/kg and 200 mg/kg) were used. The wound area's findings for contraction of excision (1.5 cm in diameter, 0.2 cm in depth) were noted down after a 3-day interval, from the third day to the twenty-first day. It was shown that the wound area's % contraction was significantly improved. The contraction % with 2% acetone R. ellipticus extract was found to be close to betadine (100% on day 12) (94.23% on day 12). According to the study, using a natural extract from the Rubus species made healing wounds far more efficient.
Antidiabetic
Sharma and Kumar (34)found that R. ellipticus had an impact on the alloxan-induced rats' glucose tolerance levels. They found that taking 200 mg/kg of fruit extract orally for 15 days had a substantial influence on diabetic activity. Fruit juice was extracted using three distinct solvents: ethanol, petroleum ether, and aqueous solutions, with better results achieved for ethanolic and aqueous extracts than petroleum extracts. According to reports, plant matter exhibits an antihyperglycemic effect by preserving the ability of beta cells in the pancreas to absorb the most glucose and assisting in lowering glucose load (40). Subba et al. (19) found that the methanolic extract of R. ellipticus leaves inhibits α-amylase significantly, with an IC50 of 269.94 ± 0.11 g/mL. Euscaphic acid present in R. ellipticus was found to have the most effective α-amylase inhibitor, with IC50 values of 0.65 mM (30).
Nephroprotective
The nephroprotective effect of R. ellipticus fruit extracts on gentamicin and cisplatin-induced nephrotoxicity in rats was evaluated by Sharma et al. (41). Nephrotoxicity was induced in Wistar rats by intraperitoneal administration of gentamicin (100 mg/kg/day) for eight and cisplatin (7.5 mg/kg/day) for 10 days. The nephroprotective effect of concurrent administration of petroleum ether, ethanolic and aqueous extracts of R. ellipticus fruits at a dose of 200 mg/kg/day given by oral route was determined. And it was concluded that the pet. ether and aqueous extracts of the fruits are less significant in comparison to ethanolic extract. On acetaminophen-induced nephrotoxicity in male albino rats, the pet. ether, ethanolic, and aqueous extract had significant nephroprotective and was observed to protect against necrotic damage of renal tissue.
Antiproliferative and Antitumor
Since cancer has become the most sensitive disease in today's age, the ability to limit cancer cell multiplication is more crucial than ever. Using natural goods to combat this type of issue is the best option. Fruit extracts have been tested for their ability to inhibit the proliferation of two different Human Cervical Cancer Cell lines, C33A cells, and HeLa cells, and fruit extracts (at varying concentrations) were found to be more efficient against C33A cells (23). In solid tumours generated by carcinoma cells such Dalton lymphoma ascites (DLA) cells and Ehrlich ascites carcinoma, R. ellipticus has also been investigated for its anticancer properties by George et al. (33). From the study, it was observed that the methanolic leaf extract lowers the impact of tumor cells. Fruit extract therapy at various doses reduces the size of DLA-induced tumours in mice from 3.07 cm3 to 2.56 cm3, while increasing the amount of extract from 50 to 250 mg/kg body weight. They also found that at the same concentration (i.e., 50 to 250 mg/kg body wt.), the extracts were effective against EAC cell-induced tumors and also efficient in prolonging the life span of animals. Furthermore, Muniyandi et al. (26) reported that the methanolic extract significantly and dose-dependently reduced the viability of the tested cell lines. Only half of the Caco-2 cell lines were viable at a concentration of 10 g/l.
Antifertility
As per the Ayurvedic and Unani systems of traditional medicine, R. ellipticus proposed to have antifertility activity. According to the study of Dhanbal et al. (42), leaves extract of R. ellipticus at 200 mg/mL showed 54.33% anti-implantation, 37.10% early abortifacient, and 91.43% of total anti-fertility activity in rats. And also concluded that the extract significantly decreased implantation sites and increased reabsorption in female albino rats. According to the study of Sharma et al. (43), the aerial portion of the entire plant without roots has 100% activity even at the lower dose (50 mg/kg body weight) during 1-3 days of pregnancy, the roots of R. ellipticus (250 mg/kg) found to exhibit considerable anti-implantation action during 1–7 days of pregnancy. Prakash et al. (44)prepared extract using 90% ethanol from the entire part of the plant except for the roots and was used for studying antifertility properties. And it was concluded that at a dose of 250 mL/kg, it showed 100% antifertility activity during early pregnancy and also had potent estrogenic properties.
Antimicrobial, Antibacterial, and Antifungal Activities
The methanolic extract of R. ellipticus root bark was tested by Khanal et al. (32) for antibacterial activity against g-positive S. aureus, g-negative Salmonella typhi, and Klebsiella pneumoniae using Disc diffusion and the Resazurin microtiter assay technique.
The methanolic extract of R. ellipticus root bark had significant antibacterial activity using the disc diffusion method against S. aureus with a 17 mm zone of inhibition but had no effect on g-negative organisms. Resazurin microtiter assay results showed that the MBC (minimum bactericidal concentration) and MIC (minimum inhibitory concentration) values were 12.5 mg/mL and 3.125 mg/mL, respectively. Valivelan et al. (45) compared the antimicrobial activity of reference medication gentamycin (10–20 g/mL) with the ethanolic extract of R. ellipticus roots. The agar diffusion method was used in the study to demonstrate a limited amount of antibacterial activity at doses ranging from 250 to 1000 g/mL. It was observed that S. aureus, Bacillus subtilis, Escherichia coli, and Shigella had substantial activity at 1000 g/mL, however, there was very little antifungal activity against Saccharomyces cerevisiae, Rhizopus nigricans, Aspergillus niger, and Candida albicans. Panda et al. (46), used the broth microdilution method to assess the antibacterial activity of R. ellipticus leaf extracts against E. coli and S. aureus bacterial, and C. albicans fungal strains. It was found that the ethanolic extract had 100% growth inhibition activity against the S. aureus strain. Of, the acetone extract showed 100% growth inhibition and the highest anti-fungal effectiveness. The aqueous extract of R. ellipticus showed the highest level of antibacterial activity in E. coli (MIC50= 450 g/mL). The best solvent for anti-fungal action against C. albicans was acetone (MIC50 = 240 g/mL). They also found that the extracts were effective against various helminths and enterovirus (46). The same method is used to evaluate the antibacterial property of leaf extracts against eight common food-borne pathogens. In the study by Panda et al. (47), it was found that the maximum growth inhibition was with ethanolic extract against E. coli and to the greatest extent possible, B. cereus, L. innocua, and M. luteus were inhibited by the aqueous extract. The MIC50 value of aqueous leaf extract against E. coli, B. cereus was found as 559 μg/mL and 560 μg/mL respectively. With ethanolic extract, the MIC50 value was 273 μg/mL and 527 μg/mL against Bacillus cereus, and E. coli respectively.
Toxicological Activity
In a work by George et al. (5), rats and mice were given various dosages of methanol extracts to assess acute toxicity (i.e., 0.1 g, 0.5 g, 1 g, and 2 g/kg), followed by a three-h fast. The toxicity testing results showed no evidence of mortality. According to Sharma and Kumar's study (34) on Swiss albino mice and Wistar albino rats, R. ellipticus fruit extracts in petroleum ether, ethanolic, and aqueous form did not cause any harmful neurological or behavioural effects up to a dose of 2000 mg/kg. Furthermore, Swiss albino mice and Wistar albino rats at a dose of 2000 mg/kg of R. ellipticus leaf methanolic extract did not exhibit any behavioral changes and did not die. In another study, rats were used in in vivo tests for acute toxicity, which were carried out following OECD (Organization for Economic Co-operation and Development) recommendations. No clinical abnormalities were noticed from a few h to several days at an oral dose of 2 g/kg (7). The methanolic extract of R. ellipticus leaves was tested for cytotoxicity in the HEK293 cell line by Sachdeva et al. (24), who found that the extract had a TC50 value of 90 g/mL and had no harmful effects on HEK293.
Other Activities
According to reports, the leaves of R. ellipticus have hypnotic effects that are enhanced by pentobarbitone sodium, anticonvulsant properties against electrically induced convulsions, and positive inotropic and chronotropic effects (13). According to the experiment carried out by Alqhtani et al. (48), 1 mL of aqueous leaf extract in combination with 249 mL of dechlorinated water was found to exhibit ovicidal and larvicidal activity against Culex quinquefasciatus, Aedes aegypti, and Anopheles stephensi. And the root was found to have antiprotozoal activity against Entamoeba histolytica and hypoglycemic activity (49).
Conclusion
Rubus fruits or berries are high in nutrients and contain a diverse spectrum of phytochemicals. One of the significant ethnomedicinal plants among them, R. ellipticus, serves a variety of functions, including being utilized to produce processed products, edible fruits, and traditional remedies. Its intake may be crucial in the fight against several illnesses due to its anti-inflammatory, nephroprotective, anti-proliferative, antipyretic, cytotoxic, analgesic, anti-cancer, anti-fertility, wound-healing, anti-microbial, antioxidant, and anti-plasmodial characteristics. In light of the aforementioned, one of the probable mechanisms of action of the extract might be its free radical scavenging and antioxidant properties. This hypothesis, however, needs to be validated. We may infer from the facts above that R. ellipticus can be employed as a raw material in the formulation of nutraceuticals.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
Not applicable.
Ethics Statement
Not applicable.
Funding Information
Not applicable.
References
- Wyk V, Ben-Erik, Wink M. Phytomedicines, Herbal Drugs, and Poisons. Chicago: University of Chicago Press; 2016. p. 304.
- Vaidya ADB, Devasagayam TPA. Current status of herbal drugs in India: An overview. J Clin Biochem Nutr. 2007;41(1):1–11.
- Abu Ahmed AM, Sharmen F, Mannan A, Rahman MA. Phytochemical, analgesic, antibacterial, and cytotoxic effects of Alpinia nigra (Gaertn.) Burtt leaf extract. J Tradit Complement Med. 2015;5(4):248–52.
- Ravi S, Bharadvaja N. Market Analysis Of Medicinal Plants In India: Review. Curr Pharm Biotechnol. 2019;20:1–22.
- George BP, Parimelazhagan T, Saravanan S. Anti-inflammatory, analgesic and antipyretic activities of Rubus Ellipticus smith. leaf methanol extract. Int J Pharm Pharm Sci. 2013;5(Suppl 2):220–4.
- Ahmad M, Masood S, Sultana S, Hadda T Ben, Bader A, Zafar M. Antioxidant and nutraceutical value of wild medicinal Rubus berries. Pak J Pharm Sci. 2015;28(1):241–7.
- Vadivelan R, Bhadra S, Ravi AV, Shanish KS, Elango KSB. Evaluation of anti-inflammatory and membrane stabilizing property of ethanol root extract of Rubus ellipticus Smith in Albino rats. J Nat Remedies. 2009;9(1):74–8.
- Maity D, Pradhan N, Chauhan AS. Folk uses of some medicinal plants from North Sikkim. Indian J Tradit Knowl. 2004;3(1):66–71.
- Badhani A, Rawat S, Bhatt ID, Rawal RS. Variation in Chemical Constituents and Antioxidant Activity in Yellow Himalayan (Rubus ellipticus Smith) and Hill Raspberry (Rubus niveus Thunb.). J Food Biochem. 2015;39(6):663–72. Available from: https://doi.org/10.1111/jfbc.12172
- Lowe S, Browne M, Boudjelas S, De Poorter M. 100 of the World’s Worst Invasive Alien Species: A selection from the Global Invasive Species Database. Auckland: ISSG; 2000. 7 p.
- Wu K, Zhang J, Zhang G, Ding J. Epiblema tetragonana and Epinotia ustulana (Lepidoptera: Tortricidae), two potential biological control agents for the invasive plant, Rubus ellipticus. Biol Control. 2014;77:51–8.
- Wu K, Center TD, Yang C, Zhang J, Zhang J, Ding J. Potential classical biological control of invasive Himalayan yellow raspberry, Rubus ellipticus (Rosaceae). Pac Sci. 2013;67(1):59–80.
- Pandey Y, Bhatt SS. Overview of Himalayan yellow raspberry (Rubus ellipticus Smith.): A nutraceutical plant. J Appl Nat Sci. 2016;8(1):494–9.
- Upreti K, Tewari LM, Jalal JS. Diversity and Distribution of Wild Edible Fruit Plants of Uttarakhand. In: Biodiversity Potentials of the Himalaya. 2010. p. 157–96.
- Saklani S, Chandra S, Badoni P, Dogra S. Antimicrobial activity, nutritional profile and phytochemical screening of wild edible fruit of Rubus ellipticus. Int J Med Aromat Plants. 2012;2(2):269–74.
- Ringmichon CL, Gopalkrishnan B, Dixit AP. Ethno-pharmacognostical Studies on Root Bark of Rubus ellipticus Smith . from Manipur. J Pharmacogn Phytochem. 2013;2(2):223–8.
- Pradhan BK, Badola HK. Ethnomedicinal plant use by Lepcha tribe of Dzongu valley, bordering Khangchendzonga Biosphere Reserve, in North Sikkim, India. J Ethnobiol Ethnomed. 2008;4:22.
- Uprety Y, Poudel RC, Asselin H, Boon E. Plant biodiversity and ethnobotany inside the projected impact area of the Upper Seti Hydropower Project, Western Nepal. Environ Dev Sustain. 2011;13(3):463–92.
- Subba B, Gaire S, Sharma KR. Analysis of Phyto-Constituents, Antioxidant, and Alpha Amylase Inhibitory Activities of Persea Americana Mill., Rhododendron Arboretum Sm. Rubus Ellipticus Sm. From Arghakhanchi District Nepal. Asian J Pharm Clin Res. 2019;12(1):301.
- Saklani S, Chandra SMA. Evaluation of Nutritional profile, medicinal value and quantitative estimation in different parts of Pyrus pashia, Ficus palmata and Pyracantha crenulata. J Glob Trends Pharm Sci. 2011;2(3):350–4.
- Belwal T, Pandey A, Bhatt ID, Rawal RS, Luo Z. Trends of polyphenolics and anthocyanins accumulation along ripening stages of wild edible fruits of Indian Himalayan region. Sci Rep. 2019;9:1–11. Available from: http://dx.doi.org/10.1038/s41598-019-42270-2
- Badhani A, Rawat S, Bhatt ID, Rawal RS. Variation in Chemical Constituents and Antioxidant Activity in Yellow Himalayan (Rubus ellipticus Smith) and Hill Raspberry (Rubus Niveus Thunb.). J Food Biochem. 2015;39(6):663–72.
- Saini R, Dangwal K, Singh H, Garg V. Antioxidant and antiproliferative activities of phenolics isolated from fruits of Himalayan yellow raspberry (Rubus ellipticus). J Food Sci Technol. 2014;51(11):3369–75.
- Sachdeva C, Mohanakrishnan D, Kumar S, Kaushik NK. Assessment of in vitro and in vivo antimalarial efficacy and GC-fingerprints of selected medicinal plant extracts. Exp Parasitol. 2020;219:107994.
- George E, George B, Sajeesh T, Thangaraj P, Muniyandi K, Sathyanarayanan S. GC-MS Analysis of Methanolic Extract of Rubus ellipticus. In: Phytomedicine, Research and Development. 2020. p. 37–43.
- Muniyandi K, George E, Sathyanarayanan S, George BP, Abrahamse H, Thamburaj S, et al. Phenolics, tannins, flavonoids and anthocyanins contents influenced antioxidant and anticancer activities of Rubus fruits from Western Ghats, India. Food Sci Hum Wellness. 2019;8(1):73–81. Available from: https://doi.org/10.1016/j.fshw.2019.03.005
- Smith R, Levl R. Oleanane and Ursane Glucosides From Rubus Species. Phytochemistry. 1992;31(10):3642–4.
- Dutta SP, Ghatak KL, Ganguly SN. Isolation and Structure Elucidation of New Pentacyclic Triterpene Acid from the Leaves of Rubus ellipticus. Nat Prod Sci. 1997;3:108–10.
- Aswal BS, Goel AK, Kulshrestha DK, Mehrotra BN, Patnaik GK. Screening of Indian plants for biological activity: Part XV. Indian J Exp Biol. 1996;34(5):444–67.
- Li W, Fu H, Bai H, Sasaki T, Kato H, Koike K. Triterpenoid saponins from Rubus ellipticus var. obcordatus. J Nat Prod. 2009;72(10):1755–60.
- Talapatra S, Kamacharya B, De S, Talapatra B. Chemical investigation of some medicinal plants of Nepal. Indian J Chem. 1989;28(4):356–7. Available from: https://www.sciencedirect.com/science/article/pii/0378874195012426
- Khanal LN, Sharma KR, Pokharel YR, Kalauni SK. Assessment of Phytochemical, Antioxidant and Antimicrobial Activities of Some Medicinal Plants from Kaski District of Nepal. Am J Plant Sci. 2020;11(09):1383–97.
- George BP, Parimelazhagan T, Kumar YT, Sajeesh T. Antitumor and Wound Healing Properties of Rubus ellipticus Smith. JAMS J Acupunct Meridian Stud. 2015;8(3):134–41.
- Sharma US, Kumar A. Anti-diabetic effect of Rubus ellipticus fruit extracts in alloxan induced diabetic rats. J Diabetol. 2011;2(4):2–7.
- Sharma US. Nephroprotective evaluation of Rubus ellipticus (smith) fruits extracts against cisplatin and gentamicin induced renal-toxicity in rats. J Pharm Res. 2010;4(1):285–7.
- Deighton N, Brennan R, Finn C, Davies HV. Antioxidant properties of domesticated and wild Rubus species. J Sci Food Agric. 2000;80(9):1307–13.
- Lamien-Meda A, Lamien CE, Compaoré MMY, Meda RNT, Kiendrebeogo M, Zeba B, et al. Polyphenol content and antioxidant activity of fourteen wild edible fruits from Burkina Faso. Molecules. 2008;13(3):581–94.
- Sharma M, Neerarani G, Kumar DB. Evalution of Total Phenolic Content, Antioxidant Properties of Different Leaf Extracts of Rubus ellipticus. Ethiop Int J Multidiscip Res. 2014;2(2):14–7.
- Sharma US, Kumar A. In vitro antioxidant activity of Rubus ellipticus fruits. J Adv Pharm Technol Res. 2011;2(1):47–50.
- Jadhav JK, Masirkar VJDV. Antihyperglycemic effect of Diospyros melanoxylon (Roxb.) bark against alloxan-induced diabetic rats. Int J Pharmtech Res. 2009;1(2):196–200.
- Sharma US, Kumar A, Hauz A, Road A, Pradesh U, Road J, et al. Therapeutic Efficacy of Rubus Ellpticus (Smth) Fruits Extracts in Acute Acetaminophen Induced Nephrotoxicity in Rats. Pharmacologyonline. 2010;3:514–24.
- Dhanabal SP, Prasanth S, Ramanathan, Elango K, Suresh B. Validation of Antifertility Activity of Various Rubus Specis in Female Albino Rats. Indian J Pharm Sci. 2000;62(1):58–60.
- Sharma BB, Varshney MD, Gupta DN, Prakash AO. Antifertility screening of plants. Part I. Effect of ten indigenous plants on early pregnancy in albino rats. Pharm Biol. 1983;21(4):183–7.
- Prakash AO. Biological evaluation of some medicinal plant extracts for contraceptive efficacy in females. Future Aspects Contracept. 1985;13(4):115–28.
- Vadivelan R, Kumar R, Bhadra S, Raghuram A, Shanish A, Elango K, et al. Antimicrobial evaluation of the ethanolic root extracts of Rubus ellipticus (Smith). Pharmacist. 2008;3(1):19–21.
- Panda SK, Padhi L, Leyssen P, Liu M, Neyts J, Luyten W. Antimicrobial, anthelmintic, and antiviral activity of plants traditionally used for treating infectious disease in the Similipal Biosphere Reserve, Odisha, India. Front Pharmacol. 2017;8:658.
- Panda SK, Mohanta YK, Padhi L, Luyten W. Antimicrobial activity of select edible plants from Odisha, India against food-borne pathogens. LWT. 2019;113:108246.
- AlQahtani FS, AlShebly MM, Govindarajan M, Senthilmurugan S, Vijayan P, Benelli G. Green and facile biosynthesis of silver nanocomposites using the aqueous extract of Rubus ellipticus leaves: Toxicity and oviposition deterrent activity against Zika virus, malaria and filariasis mosquito vectors. J Asia-Pac Entomol. 2017;20(1):157–64.
- Patel AV, Rojas-Vera JDC. Therapeutic constituents and actions of Rubus species. Curr Med Chem. 2004;11(11):1501–12.
