RESEARCH ARTICLE
Viability of Lactobacillus acidophilus in Effervescent Granules Prepared via Wet Granulation Method: In Vitro Study
Sciences of Pharmacy|Vol. 2, Issue 4, pp. 232-242 (2023)
Received
Aug 29, 2023Revised
Nov 22, 2023Accepted
Nov 24, 2023Published
Nov 25, 2023
Abstract
Keywords:
Introduction
Microorganisms play a crucial role in fulfilling human life needs. The use of probiotics as a therapeutic tool can be developed, among other things, as an immunomodulator and antimicrobial agent. The diverse functions of probiotic bacteria are considered an alternative solution to various issues related to bacterial antibiotic resistance, irrational antibiotic use, and its increasing use in recent decades (1). The potential of probiotics as an alternative to antibiotics is because research by Halder (2017) states that probiotics have the potential to act against various infectious agents, one of which is Lactobacillus (2). Lactobacillus can fight pathogens by producing different antimicrobial components, lowering pH, competing with pathogens for adhesion and nutrients in the intestine, and suppressing the growth of pathogenic bacteria through direct coaggregation with bacteria (3). Sekendar (2017), states that Lactobacillus produces bacteriocins, which have strong antibacterial properties and act bactericidally or bacteriostatically against various food pathogens (4). Bacteriocin synthesis may also contribute to the probiotic activity of intestinal Lactobacillus and, in some cases, may be directly responsible for beneficially altering the intestinal microbiota or suppressing some gastrointestinal pathogenic bacteria (5). Many benefits of using probiotics come from their ability to combat pathogenic organisms through various mechanisms, including their immunomodulatory effects and their ability to restore the balance of normal gut flora (6).
Probiotics are beneficial live bacteria that promote digestive health and protect against pathogenic bacteria. Typically, probiotics are found in foods and beverages. Probiotics are live microorganisms that can provide positive health effects when consumed in sufficient amounts. Probiotic products available in the market today use bacteria from the Lactobacillus and Bifidobacterium species. The development of drinks and supplements containing probiotics is in accordance with research by Shabbir (2023), who carried out the development of Fermented Skim Milk drinks equipped with Probiotics with a combination of Lactobacillus casei and Lactobacillus rhamnosus, concluding that Probiotics can be used in milk fermentation to make fermented milk drinks and beverages. Yakult can be used to market innovative beverages to specific consumer groups, manufacturers and industries, as well as large-scale retail distribution (7). Researcher Badgeley (2021) said that in recent years, researchers have investigated various health benefits associated with probiotic supplements, which primarily contain live microorganisms from Bifidobacterium and Lactobacillus species. The main areas of focus of his research are the effects of probiotic supplements on gut microbiota and cancer prevention, as well as on the efficacy and toxicity of chemotherapy (8). Commonly used Lactobacillus species include Lactobacillus acidophilus (L. acidophilus), Lactobacillus johnsonii, L. casei, L. rhamnosus, Lactobacillus gasseri, and Lactobacillus reuteri, while Bifidobacterium species include Bifidobacterium bifidum, Bifidobacterium longum, Bifidobacterium breve, and Bifidobacterium infantis (9). Both the Lactobacillus and Bifidobacterium have different roles in human digestion. L. acidophilus aids in intestinal lactose digestion, stimulates the immune response against unwanted microorganisms, and helps control blood cholesterol levels. L. acidophilus is one of the lactic acid bacteria that can be utilized as a probiotic microbe and is currently one of the most widely developed probiotic strains (9).
Lactic acid bacteria (LAB) have long been used in the food industry for their ability to convert sugars, including lactose and other carbohydrates, into lactic acid (10). L. acidophilus bacteria can impart the characteristic sour taste to fermented dairy products such as yogurt and also produce a low pH, which can inhibit the growth of other microbes and survive in the stomach alongside thousands of other bacteria (11). For the fermentation process, the raw material used (milk) must undergo initial heating (pasteurization), cooling, adding lactic acid bacteria and an incubation process. After the incubation process, the process of storing the fermented yogurt product is by storing it in the refrigerator (11). The role of lactic acid bacteria in making yoghurt and similar products is in producing acids and flavor compounds such as acetaldehyde. According to Abedi (2020), lactic acid produced by the metabolism of L. acidophilus can lower the pH thereby inhibiting the growth and reproduction of pathogenic bacteria (12).
Given the importance of L. acidophilus bacteria, one way to utilize them is by granulating the bacteria into granules. In a study using L. casei (13), it was found that drying at 45°C for 2 hours was the optimal condition to achieve good viability and quality of probiotic granules, with a viability result of 7.43 cfu/gram (meeting the requirements). According to the FAO and WHO, probiotic products should contain at least 106 – 107 cfu of live bacteria per gram of the probiotic product (14).
In the study conducted by Gantini (2016), probiotic products were made into instant granules using the wet granulation method. In this research, probiotic products will be formulated into effervescent granules using the wet granulation method. In addition to the active ingredient, the formulation of effervescent granulation also includes several additives, such as carbonate additives to enhance taste and acid source additives (15). The acid sources referred to here are citric acid and tartaric acid. Citric acid and tartaric acid have their respective properties that, when combined, will affect the physical properties of the resulting granules (16). The wet granulation method is a process of transforming fine powder into granules using a suitable binder solution. The advantages of the wet granulation method include achieving good flowability, improving compressibility, controlling release, preventing component separation during the process, ensuring even granule distribution, and increasing dissolution rate (13).
Based on the above, we formulated effervescent granules containing L. acidophilus bacteria using the wet granulation method as a probiotics supplement. Therefore, physical properties of effervescent granules were evaluated including the organoleptic, flow rate, angle of repose, bulk density, tapped density, compressibility index, water content, dissolution time, particle size distribution, sedimentation, foam height, and viability of lactic acid bacteria using the Total Plate Count (TPC) method.
Methods
Materials
Lactobacillus acidophilus was obtained from Agritama Sinergi Inovasi (AGAVI) Co. , Ltd (Indonesia). Excipients used were polyvinylpyrrolidone (Anhui Sunhere Pharmaceutical Excipients Co. , Ltd, China), lactose (DMV-Fonterra Excipients GmbH & Co. , KG, Germany), tartaric acid (Justus Kimiaraya Co. , Ltd, Indonesia), citric acid (Shandong Ensign Industry Co. Ltd, China), sodium bicarbonate from (AGC Inc. Chemical Co. , Ltd, Japan), De Man Rogosa and Sharpe Broth (Merck KGaA, Darmstadt, Germany).
Tools
The tools used in this research include sieves No. 12 and 18, glassware (Pyrex), mortar and stamper, micropipettes, test tubes, 10 mL centrifuge tubes, tube racks, stopwatches, analytical scales (Mettler Toledo), hot plates (IKA®C-MAG HS-4), autoclave (ALL AMERICAN®), incubator (Memmert®), centrifuge (DLAB®), oven, granule flow tester, tap density tester (TDT-3-H), moisture balance (Radwag®), pH meter (Mettler Toledo), orbital shaker (IKA® KS 4000-i), microscope, colony counter (Funke Gerber®), vortex mixer H-VM-300 (HEALTH®).
Morphological Identification of Lactobacillus Acidophilus Isolate
Confirmation Test for Bacterial Growth
The deMan Rose Sharp Agar (MRSA) media and test tubes underwent sterilization by autoclaving at 121°C for 15 min. The working table was sanitized using a cloth/tissue dampened with 70% alcohol. Subsequently, 15 mL of sterile MRSA media was dispensed into each test tube and allowed to solidify with the tube positioned at an angle between two Bunsen burners. Once solid, a single dose of pure L. acidophilus bacteria was streaked onto the solidified media in a zig-zag pattern. Incubation was carried out for 24 h at 37°C (17).
Bacterial Gram Staining Test
The bacterial suspension was streaked onto the slide using a loop and fixed to the bottom of the slide. Following this, 1-2 drops of methylene blue were dripped, allowed to stand for 1 min, then cleaned with distilled water and air-dried. Subsequently, 1-2 drops of iodine were added, left for 1 min, rinsed with distilled water, and dried. Once fully dried, 70% alcohol was applied, left for 30 min, followed by rinsing with distilled water and drying. After complete drying, 1-2 drops of fuchsin were added, left for 1 min, then rinsed using distilled water and dried. Observations were conducted using a microscope at a magnification of 100x.
Preparation of Lactobacillus acidophilus Effervescent Granules
Sucrose was initially dissolved in 300 mL of distilled water and subsequently sterilized through autoclaving at 121°C for 15 min. A 10% v/v of L. acidophilus bacterial culture suspension (inoculum) was introduced into 30 mL of sterile MRSB medium within a 100 mL Erlenmeyer flask. The mixture was incubated at room temperature with agitation at 125 rpm for a duration of 48 h. To achieve biomass production of L. acidophilus bacteria, the inoculum was integrated into the cultivation medium (sucrose medium) and subjected to incubation for 24 h. This incubation occurred with continuous agitation at 125 rpm at room temperature. Harvesting and subsequent separation of the biomass were achieved through centrifugation at 5000 rpm for 20 min. The obtained mass of L. acidophilus were subsequently subjected to a drying process within an oven set at 40°C for a duration of 24 h.
| Material | Function | F1 (%) | F2 (%) | F3 (%) |
|---|---|---|---|---|
| L. acidophilus | Active Substance | 3 | 3 | 3 |
| Polyvinylpyrrolidone | Binder | 3 | 3 | 3 |
| Citric Acid | Acid Component | 0.3 | 1.57 | 1.15 |
| Tartaric Acid | Acid Component | 49.7 | 48.42 | 48.85 |
| Sodium Bicarbonate | Base Component | 25 | 25 | 25 |
| Lactose | Filler | Ad 100 | Ad 100 | Ad 100 |
Determination of the concentration of bacteria used is in accordance with research by Gantini (2016) which formulated Lactobacillus casei bacteria which were made into instant granules. Where the concentration of bacteria used is 3% (13). When designing the formula to be used, formula optimization is first carried out to obtain the optimum formula. In this optimization, Design Expert Software was used to optimize the ingredients, citric acid and tartaric acid were chosen as independent variables by setting citric acid with a lower limit value of 0.3 and upper limit 2.0, while tartaric acid had a lower limit value of 0.5 and an upper limit value of 50. PVP binder is used in the form of an alcohol solution with a concentration of 3-15%. Granulation using this material will produce good granules, dry quickly, and have good compressive strength (18). So the presentation used is a minimum PVP presentation of 3%.
The formulation of effervescent granules was achieved using the wet granulation technique (19). Initially, the base portion comprising Sodium Bicarbonate, lactose, and PVP K30 solution was blended in a mortar until achieving a smooth dough-like consistency, followed by sieving through a mesh with a pore size of 12, and subsequently subjected to drying at 40°C for a duration of 15 minutes, resulting in Mass 1. Concurrently, the mixture of citric acid, tartaric acid, lactose, and PVP K30 solution was similarly combined in a mortar to form a homogenous dough, sieved through mesh 12, and subjected to drying at 40°C for 15 minutes, yielding Mass 2. Subsequently, Mass 1, Mass 2, and L. acidophilus bacteria were combined, sieved through mesh 12, and subjected to a 3-hour drying process, followed by dry sieving through mesh 18. Finally, the granules were evaluated for their characteristics.
Physical Characterization of Lactobacillus acidophilus Effervescent Granules
Examinations for the physical characteristics of effervescent granules containing L. acidophilus bacteria include organoleptic evaluation, flow rate examination, angle of repose analysis, compressibility assessment, loss on drying measurement, dissolving time evaluation, pH testing, and foam height examination
Organoleptics
An ample quantity of granules was collected, followed by a direct observation where parameters such as shape, color, taste, and aroma were taken into consideration (18).
Flow Rate
The flow rate test was carried out by inserting 100 g of granule into the funnel. The bottom cover of the funnel was opened and the fall time of the granules was calculated using a stopwatch (20). The flow rate is said to be good if it has a flow rate of less than 10 g/sec (21).
Angle of Repose
As much as 50 g of granules was put into the funnel or into the Granule Flow Tester tool which was stored on a flat surface. The funnel valve was then opened until all the granules fall down form the cone. The diameter and height of the granule cone formed were measured (22). Requirements for a good angle of repose range from 25o – 40o (23).
Compressibility
As much as 50 g of granules was put into a volumenometer measuring cup, then the initial height of the granules was recorded. The measuring cup was tapped for 250 beats at a constant speed after which the granule weight was obtained. It was compressed and the real specific gravity and incompressed specific gravity were measured (24). Good compressibility value is less than 20% (25).
Loss on Drying
As much as 2 g of dry granules was weighed and put into the moisture balance tool. After 5 minutes, the water content was measured (26). The water content requirements for effervescent granules range from 0.4% - 0.7% (27).
Dissolving Time, Foam Height, and pH
Ten grams of granules were placed into a beaker glass and dissolved with 250 mL of water. The dissolution process was observed using a stopwatch, and the results obtained were recorded. The time taken for the granules to completely dissolve was determined (28). The test was conducted to assess the dispersion time of the granules in water. The prepared granules met the criteria for a good granule, with a dissolution time ranging between 1-2 minutes, as per the effervescent granule dissolving time requirement (20). During the granule dispersion process, foam height was measured to assess the granules' dispersion capability (28). pH of the solution was then measured.
Viability Test
A total of 68.2 grams of MRSA media was dissolved in 1000 milliliters of distilled water and heated until homogeneous. The media was then sterilized in an autoclave at 121 degrees Celsius for 15 minutes to ensure complete sterilization. Subsequently, a total of 15 milliliters of sterile MRSA media was dispensed into a petri dish and allowed to solidify. Once solidified, effervescent granules were sprinkled onto the surface of the MRSA media and evenly spread. The prepared petri dish was then incubated for 24 hours with the lid inverted to prevent condensation. Following the incubation period, the colonies that grew on the media were manually counted using a colony counter, and the total plate count was calculated based on these observations.
Result
Morphological Identification of Lactobacillus acidophilus Isolate
The confirmation test for bacterial growth was conducted using MRSA selective media, with the goal of enhancing bacterial proliferation at 37°C during a 24-hour incubation period. The growth of L. acidophilus isolates was conspicuously manifested through the development of turbidity on the agar's surface, indicating robust growth. The outcomes of macroscopic observations of LAB are depicted in Figure 1, illustrating the distinct characteristics observed. Additionally, the results of microscopic observations with Gram staining can be seen in Figure 1, providing a detailed view of the bacterial morphology and arrangement.




Organoleptic Test
The result of organoleptic assessment, the manufactured effervescent granule formulations underwent evaluations concerning their shape, color, and aroma. Organoleptically, all three formulas yielded a white appearance, presented a slightly sour taste, and exhibited a relatively mild scent. The inclusion of the active ingredient L. acidophilus bacteria in the formula did not noticeably influence the color, resulting in the observed white coloration as depicted in Figure 1.
Flow Rate of Effervescent Granules
The granules flow rate test aims to determine the quality of the effervescent granules because, in the tablet printing process, the flow rate of the powder/granules is greatly affected by the flow rate test (29). The flow rate is said to be good if it has a flow rate of not less than 10 g/sec (21). The results of the flow rate test showed that formula 1 had a better granule flow rate than formulas 2 and 3 (see Figure 2).
Angel of Repose
The angle of repose test is intended to determine the flow properties of granules. The angle of repose can be determined by pouring the granule into the funnel. Then, a repose angle will be formed. The angle of repose requirement for granules ranges from 25o– 40o (23). The results of the angle of repose test show that formula 1 has a better granule angle of repose or equal to 40o so that formula 1 is closer to the requirements than formulas 2 and 3 (see Figure 3).
Compressibility Test
The compressibility test is intended to describe the stable and compact nature of the powder when pressed in the tablet molding process. The better the ability of the granules to flow, the lower the compressibility value. The compressibility test requirement for granules of not less than 20% (21). The compressibility test results show that all formulas meet the requirements. Formula 3 has a smaller compressibility value compared to formulas 1 and 2 (see Figure 4).
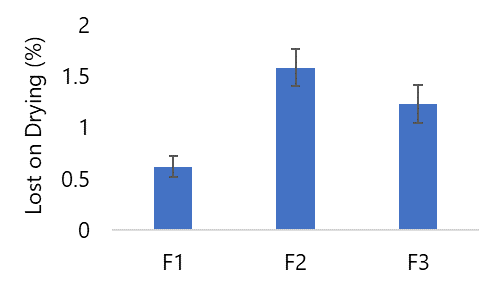
Loss on Drying
When testing the water content, which was carried out by weighing several granules and putting them into the moisture balance, the result of formula I was 0.62%, formula II was 1.54%, and formula III was 1.26% (see Figure 5). From the three results of the water content of formulations II and III, it was said that they did not meet the requirements because the water content requirements for effervescent granules, according to Fausett (2000), ranged from 0.4% - 0.7% (27). This is due to the formulation's concentration of acids and bases, and the room temperature where the preparation is made has high enough humidity.

Dissolving Time
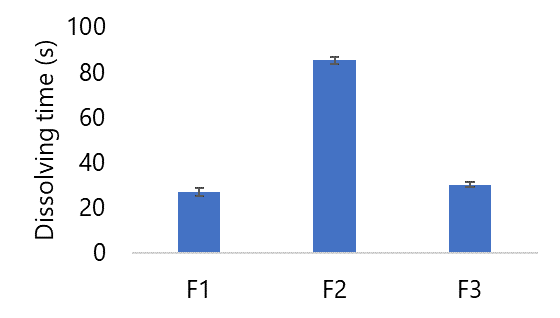
Testing the dissolving time of effervescent granules, the tool used is a stopwatch. The dissolving time test is characterized by the granules completely dissolving in water until an acid and base reaction is formed which can produce carbon dioxide gas (CO2). Between 1-2 min is the effervescent granule dissolving time. effervescent granules dissolve well (dispersed) within = 5 min can be categorized as meeting the dissolution time requirements (20). From the results of the dissolving time test, formula 1 has faster dissolving time test results than formulas 2 and 3 (see Figure 6).
pH Test
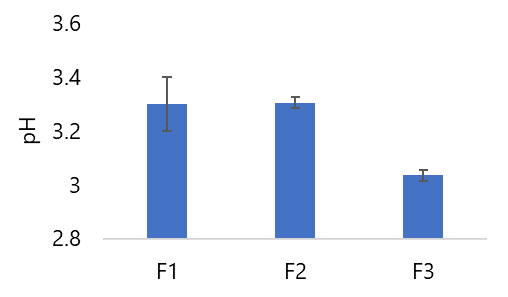
The acidity level was measured using a pH meter in an effervescent solution that had been dissolved as much as 1 g in 9 mL. The test was carried out 2 times for each formulation. The results showed that all formulas met the requirements because the pH test value was < 5 (see Figure 7).
Foam Height
Measured based on the height of the foam produced by a sample of 1 g of sample for each formulation that has been dissolved, it can be seen that the highest foam was produced by formula I (see Figure 8).
In Vitro Viability
Testing the viability of probiotic granules is a test that needs to be considered so that the probiotic bacteria contained in the granules can provide a therapeutic effect.
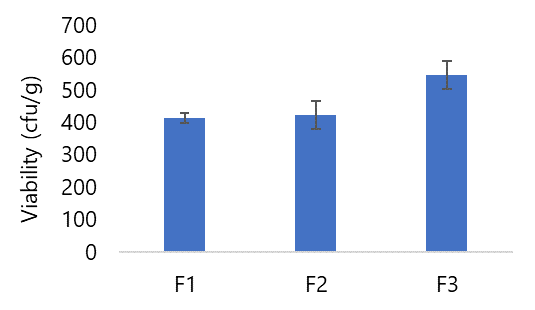
The Lactic Acid Bacteria viability test of probiotic products used the spread method. The spread method is a technique for growing microorganisms in agar media by spreading the preparations on agar media that have solidified (30). World Health Organization (WHO) requirements Lactic Acid Bacteria Viability are 107 cfu/g (14). The viability of the lactic acid bacteria for all formulas can be seen in Figure 9.
Discussion
In addition to the MRSA selective media confirmation test for bacterial growth (depicted in Figure 1), morphological identification was performed through Gram staining of bacteria obtained from pure L. acidophilus isolates. The purpose of Gram staining is to microscopically visualize colony morphology and distinguish between Gram-negative and Gram-positive bacteria. Bacteria that retain a purple hue after alcohol dissolution, following Methylene Blue absorption, are termed Gram-positive Bacteria. Conversely, bacteria that lose their purple color when washed with alcohol subsequently absorb Fuchsin dye, resulting in a pink hue; these are referred to as Gram-negative Bacteria. The response to staining is also influenced by the cell wall structure. Gram-positive bacteria, primarily featuring peptidoglycan in their cell walls, retain the violet color (31).
As indicated by the results of Gram staining, the bacteria exhibit a rod-like shape and belong to the Gram-positive category. This observation aligns with the findings of Putri (2018), who noted that Lactic Acid Bacteria generally assume the form of Gram-positive rods and are generally non-spore-forming members of the LAB group (31). Abedi and Hashemi (2020) also affirmed that Lactic Acid Bacteria are Gram-positive, non-spore-forming microorganisms capable of thriving in oxygen-rich environments and producing lactic acid through carbohydrate fermentation (e. g. , glucose, lactose) (12).
In the lactic acid fermentation process, this process begins with the change of one glucose molecule into 2 pyruvic acid molecules. This process is a stage of glycolysis so that 2 ATP molecules are produced. After the glycolysis process does not proceed to the oxidative decarboxylation stage unless 2 pyruvic acids are converted into 2 molecules of lactic acid.
During the organoleptic assessment, the manufactured effervescent granule formulations underwent evaluations concerning their shape, color, and aroma. Organoleptically, all three formulas yielded a white appearance, presented a slightly sour taste, and exhibited a relatively mild scent. The inclusion of the active ingredient L. acidophilus bacteria in the formula did not noticeably influence the color, resulting in the observed white coloration as depicted in Figure 1.
The outcomes of the effervescent granules' flow rate evaluation are presented in Figure 2, revealing distinct flow rates for Formulas I, II, and III. The table's data indicate the influence of varying concentrations of acidic ingredients on the flow rate of the effervescent granules. Specifically, Formula I exhibited a more favorable flow rate compared to Formulas II and III. As per the available literature, tartaric acid boasts a higher density, implying that granules containing greater tartaric acid concentrations exhibit increased density. Consequently, higher density corresponds to larger molecular weight, thus leading to enhanced ease of flow due to heightened gravitational forces (32).
Zuraidah's research (2018) aligns with these findings, demonstrating that increased tartaric acid concentration corresponds to an improved flow rate (33). Furthermore, in line with Hermanto's findings (2019), variations in granule shape and size also contribute to the attainment of a desirable flow rate (34). Additionally, Syahrina's work (2021) emphasizes the role of the binder in influencing flow rate. Notably, the present study employs PVP K30 as the binder, akin to Syahrina's study (2021). Syahrina's research findings highlight that PVP K30 can diminish cohesion forces, thereby augmenting particle size and leading to favorable flow rates (21).
Figure 3 illustrates the outcomes of the angle of repose measurements, all of which are equal to or exceed 40 degrees. These angle of repose data collectively indicate deficient flow properties across Formulas I, II, and III. Drawing insights from both Figure 2 dan Figure 3, the flow rate and angle of repose results in Formula I are notably smaller when compared to Formulas II and III. This correspondence is consistent with the findings of Zuraidah (2018), who emphasized the impact of citric acid and tartaric acid concentrations on variations in angle of repose across the three formulas (33).
Within Formula I, a smaller angle of repose is observed compared to Formulas II and III, an effect attributed to the substantial molecular weight of tartaric acid and its consequent influence on particle size. The presence of larger particles reduces particle cohesiveness, leading to a swifter flow rate and subsequently yielding a smaller angle of repose (33). Correspondingly, Salmatuzzahro's study (2022) indicated that the angle of repose is subject to the influences of granule shape, size, and moisture content. Variations in granule shape might stem from divergent emphases during the granulation process, potentially resulting in distinct angle of repose measurements (35).
In accordance with the study, the results of the compressibility test indicated that Formula I exhibited a value of 11.45%, Formula II measured 14.03%, and Formula III recorded 7.27%. The data presented in Figure 4 demonstrate that all attained values conform to the prescribed criteria for satisfactory compressibility, as denoted by values falling below 20% (25). Notably, the highest compressibility value is observed in Formula II. This outcome is attributed to the influence of moisture content within the powder; granules possessing elevated water content, when subjected to pressure or vibration, tend to undergo substantial volume reduction, consequently leading to heightened compressibility. Moreover, the configuration and dimensions of granule particles significantly impact compressibility values. Optimal compressibility values are associated with granules that feature uniform shapes and sizes, facilitating a smoother tablet compression process (34).
The assessment outcomes, as presented in Figure 5, demonstrate that Formula I aligns with the specified requirements. This alignment can be attributed to the hygroscopic nature of citric acid, enabling it to readily absorb atmospheric moisture. This observation resonates with the findings of Romantika (2017), who reported that augmenting citric acid content leads to increased water content in effervescent baby java oranges (36). This trend is further supported by Widyaningrum's research (2015), which highlighted that citric acid addition in pandan leaf effervescent granules correlates with heightened water content (37). Moreover, according to Audrey (2022) quoted in Lieberman et al. (1994) emphasized that citric acid exhibits marked hygroscopic characteristics, rendering effervescent granules with higher citric acid content exceptionally prone to water absorption during the manufacturing process, consequently resulting in elevated water content (38).
Figure 6 displays that the dissolving time for Formula I is notably swifter when contrasted with Formula II and Formula III. This divergence underscores the influence of distinct citric acid and tartaric acid concentrations on the solubility testing of effervescent granules. This aligns with the findings of Zuraidah's study (2018), indicating that the pronounced hygroscopic nature of higher-concentration tartaric acid fosters enhanced water absorption and, consequently, quicker reactivity in effervescent granules (33). Correspondingly, Salmatuzzahro's research (2022) expounded on the faster dissolution time of the citric acid formula characterized by the lowest concentration (35). Consistently, the current study's findings parallel these trends. Formula I boasts the lowest citric acid concentration coupled with the highest tartaric acid concentration, leading to the most rapid dissolution time compared to Formula II and Formula III. The rapid carbonation reaction between sodium bicarbonate, tartaric acid, and citric acid is facilitated by the hygroscopic nature of both citric and tartaric acids. Notably, Figure 6 illustrates that all three formulations exhibit dissolution times ≤ 5 min or within the range of 1 to 2 min. Thus, all three formulas satisfy the requirements of the solubility test.
In Figure 7, the pH value of Formula I exceeds that of Formula II and Formula III. This divergence stems from the variations in citric acid and tartaric acid concentrations, particularly the elevated tartaric acid concentration in Formula I, which significantly impacts the pH value. Kusnadhi's study (2003) highlighted that the emergence of carbon dioxide (CO2) during the interaction of two effervescent components in water, leading to the partial formation of carbonic acid, results in a reduction of H+ ions within the solution. This reduction contributes to increased solution alkalinity and consequently elevates the pH value (39). Furthermore, the properties of L. acidophilus bacteria exert an influence on the preparation's pH. Given that the optimal growth pH for L. acidophilus bacteria is ≤ 5, the presence of these bacteria in the effervescent granule formulation maintains the solution's acidity even after granulation. Based on the outcomes for the three formulas, all are classified as compliant with the pH test requisites, as their pH values are ≤ 5.
The mean froth height measurement extracted from L. acidophilus bacteria effervescent granules (Figure 8) ranged from 2 to 3 cm. Formula I exhibited a froth height of 3.1 cm, Formula II recorded 2.55 cm, and Formula III showed 2.25 cm. The optimal froth height outcome aligns with the effervescent market standard, indicating a minimal deviation of approximately 3 cm. As articulated by Bryant (1970), foam comprises numerous min bubbles originating from liquid and arising due to either chemical reactions (acidulant and carbonate) or mechanical manipulation (stirring). As these bubbles swiftly accumulate and expand on the liquid's surface, foam emerges. The substantial froth formation observed in this study can be attributed to the nature of tartaric acid. In comparison to other acids, tartaric acid exhibited the most robust outcomes in terms of carbon dioxide generation, albeit with a longer disintegration period.
In Gantini's investigation (2016), the optimal conditions involving a temperature of 45°C and a drying duration of 2 h yielded a Lactic Acid Bacteria viability of 7.43 cfu/g in instant probiotic granules. In this current study, the temperature applied was 40°C for a duration of 3 h (13). As demonstrated in Figure 9, the average Lactic Acid Bacteria viability for effervescent granules was 412.5 x 101 cfu/g in Formula I, 422.7 x 101 cfu/g in Formula II, and 544.7 x 101 cfu/g in Formula III. These findings signify the continued viability of L. acidophilus bacteria within the effervescent granule preparations. However, all formulated formulas fell short of meeting the WHO criteria for Lactic Acid Bacteria viability in probiotic products. This discrepancy could potentially be attributed to the drying duration, indicating an influence on Lactic Acid Bacteria viability. Gantini's research (2016) underscores that prolonged granule drying periods correlate with reduced bacterial viability. This underscores the critical role of drying time in determining the survival or demise of bacteria (13).
Conclusion
Based on the undertaken research focusing on the formulation and evaluation of L. acidophilus bacteria effervescent granules, the conclusion drawn is that among the three formulations, Formula I emerges as the most favorable (L. acidophilus 3%; Polyvinylpyrrolidone 3%; Citric Acid 0.3 %; Tartaric Acid 49.7%; Sodium Bicarbonate 25% and Lactose Ad). Formula I demonstrates the closest alignment with the physical requisites stipulated for effervescent granule preparations, as corroborated by the outcomes of the comprehensive tests, including the organoleptic assessment. Specifically, the granules are in the appropriate shape, white in color, and devoid of odor. The flow rate test yielded a value of 1.497 g/sec, the angle of repose was measured at 40.75°, the compressibility test resulted in 11.45%, loss on drying test showed 0.62%, and foam height measured 3.075 cm.
All three formulations, i. e. , Formulas I, II, and III, effectively incorporate L. acidophilus bacteria after being converted into effervescent granules, as substantiated by the viability of Lactic Acid Bacteria. Specifically, Formula I exhibited a Lactic Acid Bacteria viability of 412.5 x 101 cfu/g, Formula II registered 422.7 x 101 cfu/g, and Formula III recorded 544.7 x 101 cfu/g, as evidenced by the results of the Lactic Acid Bacteria viability test.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
Not applicable.
Funding Information
Not applicable.
References
- Tanujaya D, Riniwasih L. Formulasi dan Uji Stabilitas Fisik Tablet Effervescent yang Mengandung Bakteri Probiotik Lactobacillus Bulgariscus dengan Metode Granulasi Basah. Indoensia Nat Res Pharm J. 2019;4(2):pp.101-112.
- Halder D, Mandal M, Chatterjee SS, Pal NK, Mandal S. Indigenous probiotic Lactobacillus isolates presenting antibiotic like activity against human pathogenic bacteria. Biomedicines. 2017;5(2):1–11.
- Surendran Nair M, Amalaradjou MA, Venkitanarayanan K. Antivirulence Properties of Probiotics in Combating Microbial Pathogenesis [Internet]. Vol. 98, Advances in Applied Microbiology. Elsevier Ltd; 2017. 1–29 p. Available from: http://dx.doi.org/10.1016/bs.aambs.2016.12.001
- Ali MS, Lee EB, Hsu WH, Suk K, Sayem SAJ, Ullah HMA, et al. Probiotics and Postbiotics as an Alternative to Antibiotics: An Emphasis on Pigs. Pathogens. 2023;12(7).
- Cotter PD, Ross RP, Hill C. Bacteriocins-a viable alternative to antibiotics? Nat Rev Microbiol [Internet]. 2013;11(2):95–105. Available from: http://dx.doi.org/10.1038/nrmicro2937
- Rusli R, Amalia F, Dwyana Z. POTENSI BAKTERI Lactobacillus acidophilus SEBAGAI ANTIDIARE DAN IMUNOMODULATOR. Bioma J Biol Makassar. 2018;3(2):25–30.
- Shabbir I, Al-Asmari F, Saima H, Nadeem MT, Ambreen S, Kasankala LM, et al. The Biochemical, Microbiological, Antioxidant and Sensory Characterization of Fermented Skimmed Milk Drinks Supplemented with Probiotics Lacticaseibacillus casei and Lacticaseibacillus rhamnosus. Microorganisms. 2023;11(10).
- Badgeley A, Anwar H, Modi K, Murphy P, Lakshmikuttyamma A. Effect of probiotics and gut microbiota on anti-cancer drugs: Mechanistic perspectives. Biochim Biophys Acta - Rev Cancer [Internet]. 2021;1875(1):188494. Available from: https://doi.org/10.1016/j.bbcan.2020.188494
- Aini M, Rahayuni S, Mardina V, Quranayati Q, Asiah N. BAKTERI Lactobacillus spp DAN PERANANNYA BAGI KEHIDUPAN. J Jeumpa. 2021;8(2):614–24.
- Leoanggraini U, Muhadi BI. Fermentasi Mikroaerofilik Lactobacillus acidophilus untuk Produksi Probiotik. Ind Res Work Natl Semin. 2011;188–92.
- Hidayati H, Afifi Z, Triandini HR, Permata I. Pembuatan Yogurt Sebagai Minuman Probiotik untuk Menjaga Kesehatan Usus. Pros SEMNAS BIO 2021 Univ Negeri Padang. 2021;1265–70.
- Abedi E, Hashemi SMB. Lactic acid production – producing microorganisms and substrates sources-state of art. Heliyon [Internet]. 2020;6(10):e04974. Available from: https://doi.org/10.1016/j.heliyon.2020.e04974
- Gantini SN, Widayanti A. ANALISA DATA RESPONSE SURFACE METHODOLOGY PADA PENGEMBANGAN GRANUL PROBIOTIK LACTOBACILLUS CASEI. Penelit Pengenbangan Iptek. 2016;109.
- The World Health Organization. Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria. Fao Who [Internet]. 2001;(October):1–34. Available from: Cordoba, Argentina
- Mindawarnis, Hasanah D. Formulasi Sediaan Tablet Ekstrak Daun Nangka (Artocarpus heterophyllus L) dengan variasi Polivinil Pirolidon (PVP ) sebagai Pengikat dan Evaluasi Sifat Fisiknya. Univ Nusant PGRI Kediri [Internet]. 2017;01(1):1–7. Available from: http://www.albayan.ae
- Anam C, Kawiji, Setiawan RD. Study of physical and sensory characteristics and antioxidant activity of beet fruit effervescent granules (beta vulgaris) with different granulation methods and combination of acid sources. J Teknosains Pangan. 2013;2(2):21–8.
- Andarilla W, Sari R, Apridamayanti P. OPTIMASI AKTIVITAS BAKTERIOSIN YANG DIHASILKAN OLEH Lactobacillus casei DARI SOTONG KERING. J Pendidik Inform dan Sains. 2018;7(2):187.
- Tungadi R. Teknologi sediaan solid. Team WADE Publish, editor. Ponorogo; 2018. 136 p.
- Sidoretno WM. Formulasi Dan Evaluasi Granul Effervescent Kombinasi Ekstrak Kering Rimpang Jahe Merah, Temulawak Dan Kayu Manis. JOPS (Journal Pharm Sci. 2022;5(2):21–35.
- Santosa L, Yamlean PVY, Supriati HS. Formulasi Granul Effervescent Sari Buah Jambu Mete (Annacardium Ocidentale L.). Pharmacon. 2017;6(3):56–64.
- Syahrina D, Noval N. Optimasi Kombinasi Asam Sitrat dan Asam Tartrat sebagai Zat Pengasam pada Tablet Effervescent Ekstrak Ubi Jalar Ungu (Ipomoea batatas L). J Surya Med. 2021;7(1):156–72.
- Maulida Yuniar Widya Putri, Wilda Amananti HP. UJI SIFAT FISIK GRANUL EFFERVESCENT EKSTRAK DAUN SALAM ( Syzygium polyanthum ) DENGAN PEMANIS DAUN STEVIA ( Stevia Rebaudiana. Politek tegal. 2020;3(1):1–10.
- Yenrina R, Sayuti K, Anggraini T. Effect of natural colorants on color and antioxidant activity of “kolang kaling” (Sugar palm fruit) jam. Pakistan J Nutr. 2016;15(12):1061–6.
- Pratama R, Roni A, Fajarwati K, Keilmuan K, Farmasi F, Bhakti U, et al. UJI SIFAT FISIK GRANUL INSTAN EKSTRAK PEGAGAN ( Centella asiatica ). 2022;52:299–304.
- Noval N, Kuncahyo I, Pratama AFS, Nabillah S, Hatmayana R. Formulasi Sediaan Tablet Effervescent dari Ekstrak Etanol Tanaman Bundung (Actionoscirpus grossus) sebagai Antioksidan. J Surya Med. 2021;7(1):128–39.
- Sudarsono APP, Nur M, Febrianto Y. Pengaruh Perbedaan Suhu Pengeringan Granul (40°C,50°C,60°C) Terhadap Sifat Fisik Tablet Paracetamol. J Farm Sains Indones. 2021;4(1):44–51.
- Fausett H, Gayser C, Dash AK. Evaluation of quick disintegrating calcium carbonate tablets. AAPS PharmSciTech. 2000;1(3).
- Rani KC, Parfati N, Muarofah D, Sacharia SN. Formulasi Granul Effervescent Herba Meniran (Phyllanthus niruri L.) dengan Variasi Suspending Agent Xanthan Gum, CMC-Na, dan Kombinasi CMC-Na-Mikrokristalin Selulosa RC- 591. J Sains Farm Klin. 2020;7(1):39.
- Putra. D., Antari. N., Putri. N., Arisanti. C. SP. Penggunaan Polivinill Pirolidon (PVP) Sebagai Bahan Pengikat Pada Formulasi Tablet Ekstrak Daun Sirih (Piper betle L.). J Farm Udayana. 2019;8(1):14–21.
- Damayanti NWE, Abadi MF, Bintari NWD. Perbedaan Jumlah Bakteriuri Pada Wanita Lanjut Usia Berdasarkan Kultur Mikrobiologi Menggunakan Teknik Cawan Tuang Dan Cawan Sebar. Meditory J Med Lab. 2020;8(1):1–4.
- Putri AA, Erina, Fakhrurraz. Isolasi Bakteri Asam Laktat Genus Lactobacillus Dari Feses Rusa Sambar ( Cervus unicolor ). Jimvet E-Issn : 2540-9492. 2018;2(1):170.
- Egeten KR, Yamlean PVY, Supriati HS. Formulasi dan Pengujian Sediaan Granul Effervescent Sari Buah Nanas (Ananas comosus L. (Merr.). Pharmacon J Ilm Farm. 2016;5(3):116–21.
- Zuraidah N, Ayu WD, Ardana M. Pengaruh Variasi Konsentrasi Asam Sitrat dan Asam Tartrat terhadap Sifat Fisik Granul Effervescent dari Ekstrak Daun Nangka (Artocarpus heterophyllus L.). Proceeding Mulawarman Pharm Conf. 2018;8(November 2018):48–56.
- HERMANTO. Pengaruh PVP Dan HPMC Sebagai Bahan Pengikat Terhadap Sifat Fisik Tablet Effervescent. 2019;
- Salmatuzzahro A, Nawangsari D, Fitriana AS. Uji Sifat Fisik Tablet Effervescent dari Ekstrak Kulit Buah Pisang Raja ( Musa X Paradisiaca L ) dengan Perbandingan Asam Sitrat dan Natrium Bikarbonat. skripsi. 2022;
- Romantika RC, Wijana S, Perdani CG. Formulasi dan Karakteristik Tablet Effervescent Jeruk Baby Java (Cytrus sinensis L. Osbeck) Kajian Proporsi Asam Sitrat Formulation and Characterization of Baby Java Orange (Cytrus Sinensis L. Osbeck) Effervescent Tablets Study on Cytric Acid Proportion. J Teknol dan Manaj Agroindustri [Internet]. 2017;6(1):15–21. Available from: http://www.industria.ub.ac.id
- Widyaningrum A, Lutfi M, Argo BD. Karakterisasi Serbuk Effervescent dari Daun Pandan (Pandanus amaryllifolius Roxb) dengan Variasi Komposisi Jenis Asam. J Bioproses Komod Trop. 2015;3(2):1–8.
- Perbandingan Konsentrasi Asam Sitrat Dan Asam Malat Terhadap Karakteristik Granul Effervescent Bunga Telang P, Sophia Rachma Putri A, Luh Ari Yusasrini N, Timur Ina P, Studi Teknologi Pangan P, Teknologi Pertanian F, et al. The Effect of Comparative Concentration of Citric Acid and Malic Acid on the Characteristics of Effervescent Granules of Butterfly Pea Flower (Clitoria ternatea L.). Online) Audrey Sophia RP dkk / Itepa. 2022;11(4):2022–788.
- Astuti RD, Wahyu AW. Formulasi dan Uji Kestabilan Fisik Granul Effervescent Infusa Kulit Putih Semangka. J Kesehat. 2016;11(1):162–71.
