RESEARCH ARTICLE
Suspension of Sonchus arvensis L Leaves Ethanolic Extract Affects Blood Creatinine and Urea Levels in Streptozotocin-Induced Wistar Male Rats
Academic Editor: Adeleye Ademola Olutayo
Sciences of Pharmacy|Vol. 3, Issue 1, pp. 18-23 (2024)
Received
Oct 18, 2023Revised
Jan 10, 2024Accepted
Jan 12, 2024Published
Jan 14, 2024
Abstract
Keywords:
Introduction
The diabetic condition has an impact on renal function. Diabetes can adversely affect renal function by causing damage to the kidneys through processes such as glomerular damage, hypertension, inflammation, the formation of advanced glycation end-products, changes in renal blood flow, and impaired tubular function (1, 2). Over time, these factors contribute to the development of diabetic nephropathy, characterized by progressive kidney damage and a decline in function. This condition can change biochemical parameters in the bloodstream, including the concentrations of creatinine and urea (3). Creatinine, a byproduct of creatine metabolism primarily from muscle tissue, serves as a reliable marker for renal function. Urea, the end product of protein metabolism, is excreted through urine, and its accumulation in the blood is indicative of kidney dysfunction. Herbal treatments may offer a holistic approach, potentially aiding in maintaining renal health and managing these crucial biochemical markers in individuals with diabetes. Indeed, herbal remedies, often containing multiple compounds, have the potential to work synergistically to improve renal conditions and regulate metabolites (4).
S. arvensis L. , known as Tempuyung in Indonesia, is widely used as a traditional medicine against nephrolithiasis (5). S. arvensis L. ethanolic extract (SALEE) contains mineral ions such as potassium, magnesium, silica, and sodium. Organic compounds found in SALEE include saponins, flavonoids, taraxasterol, inositol, mannitol, coumarin, polyphenols, α-lactocerol, β-lactocerol, and phenolic acids (6). SALEE is said to have an adaptive immune response effect (5), increase potassium oxalate solubility (7), and reduce pro-inflammatory cytokines (8). It has also been reported to lower creatinine levels and improve glomerular cell impairment through histopathological observations (9). Although SALEE holds promise as a supplement, direct extract consumption might deter patients due to taste or impracticality for daily use. Hence, a more convenient and practical formulation of SALEE is desirable for ease of regular administration.
Suspensions, a pharmaceutical formulation where active ingredients like plant extracts are broken down into small particles and dispersed in a liquid medium, offer several benefits for enhancing the effectiveness of extract usage (10). Firstly, they enhance solubility by breaking down the extract into tiny particles, facilitating better contact with the dispersing medium (11). This improves the overall stability, preventing settling or separation of the extract over time. The even distribution of small particles ensures consistency in dosage across the formulation, aiding in absorption through cell membranes for improved bioavailability. Additionally, suspensions provide dosing flexibility and ease of use for patients, especially when dealing with extracts with strong tastes or odors, allowing for efficient utilization of the active ingredients (12, 13).
Based on the information provided, this research aimed to investigate the potential therapeutic effects of SALEE in suspension form on renal function and biochemical markers associated with kidney health, specifically focusing on creatinine and urea levels, in a diabetic rat model induced by streptozotocin.
Experimental Section
Material
S. arvensis L. leaves were collected from Pasangkayu district, West Sulawesi, Indonesia. Male Wistar rats were obtained from test animal suppliers based at Tadulako University. Materials used were NaCMC (Sigma Aldrich, St. Louis, MO, USA), hydrochloric acid (PT Lamurindo, Jakarta, Indonesia), aqua pro injection (Bernofarm Pharmaceutical Company, Sidoarjo, Indonesia), iron (III) chloride (SinarLab, Jakarta, Indonesia), citrate-buffer solution (Sigma Aldrich, St. Louis, MO, USA), dragendrof LP (Merck, Kenilworth, NJ, USA), and 96% absolute ethanol (Sigma Aldrich, St. Louis, MO, USA).
Sonchus arvensis L leaves extraction
The extraction of S. arvensis L. leaves was carried out utilizing the maceration method. Initially, 1, 050 grams of sifted simplicia powder of S. arvensis L leaves (from no. 40 mesh sifter) was weighed. Subsequently, this powder was subjected to extraction using 96% ethanol solvent in a quantity of 7.5 liters over a period of three days. The extraction process was divided into three maceration vessels, with occasional stirring to prevent saturation. The resulting filtrate was concentrated using a rotary evaporator at a temperature of 60°C. Following this, the concentrated extract underwent further evaporation using a water bath until a thick extract was obtained. The extract was sampled for initial phytoscreening, it includes alkaloid, flavonoid, saponin, fenol, and tannin (14, 15).
S. arvensis L leaves extract suspension
The SALEE was weighed to prepare suspension dosage, each containing 0.4 g, 0.6 g, and 0.8 g. Subsequently, 0.5% NaCMC was added to each extract, and the volume was adjusted with distilled water to reach 25 ml. The suspension was then thoroughly shaken until a homogeneous mixture was achieved.
Animal preparation
All experiments were conducted in adherence to the animal welfare standards outlined by the World Organisation for Animal Health (OIE) and were approved by the Research Ethics Committee, Faculty of Medicine, Tadulako University, under approval number 2576/UN.28.1.30/K/2019. Male Wistar rats weighing between 200–250 g were acclimated in specific local animal cages. These rats were given a 14-day adaptation period. The selection criteria for the rats included being approximately three months old male rats with body weight within the range of 200-250 g, having white fur, and displaying active behavior.
Animals treatment
Streptozotocin powder was weighed and dissolved in pH 4.5 citrate-buffered saline with a final concentration of 10 mg/mL. All rats (in group II-V) were induced intraperitoneally (IP) at a dose of 40 mg/kg BW (Tuldjannah et al. , 2018). After the induction, blood samples were collected and the creatinine and urea levels were measured spectrophotometrically. For the treatment, healthy control group (Group I) and control negative group (Group II) received only 0.5% NaCMC. Group III-V received 200, 300, and 400 mg/kg BW SALEE. All treatments were done daily for 14 days. Blood sample was taken from the tail on day 0, 7, 14, and 21.
Statistical analysis
Statistical analysis was conducted to determine a significance of difference between groups of treatments. ANOVA test was used for multivariate comparison analysis, followed by Post Hoc tests (Tukey). All statistical analyses were performed using R Studio (Version 4.2.1, RStudio Inc, Boston, USA).
Result and Discussion
Extract yield and pythochemicals
The extract derived from 1, 050 grams of dried S. arvensis L. leaves yielded 71 grams, which is a 6.76% yield. A qualitative analysis of this extract confirmed the presence of flavonoids, phenols, alkaloids, tannins, and saponins, aligning with existing literature (16). Refer to Table 1 for a detailed analysis of the SALEE.
| No | Secondary metabolite compounds | Result | |
|---|---|---|---|
| SALEE characteristics after reaction | Present | ||
| 1 | Alkaloid | An orange precipitate formed | + |
| 2 | Flavoniod | Formed brick red color | + |
| 3 | Saponin | Formed froth that persists for at least 1 minute | + |
| 4 | Fenol | Blackish green color formed | + |
| 5 | Tannin | Blackish blue color formed | + |
Creatinine level
Elevated levels of creatinine serve as a parameter for kidney failure. Creatinine is a byproduct of phosphocreatine breakdown, a compound utilized by muscles for energy production. The kidneys function to filter the blood and excrete creatinine through urine. In individuals with diabetes, particularly poorly controlled type 2 diabetes, high blood glucose levels can damage small blood vessels in the kidneys. This damage impairs the kidneys' ability to efficiently filter the blood, causing creatinine and other substances that should be excreted in the urine to persist in the bloodstream. Accumulation of creatinine in the blood indicates a decline in kidney function. The normal serum creatinine levels in rats range from 0.4 to 0.8 mg/dL (17).
In Figure 1, it can be observed that all streptozotocin-induced groups exhibit significantly higher levels of creatinine compared to the normal control group (Group I, non-diabetic and no treatment) on day 0. This suggests that streptozotocin can effectively induce an elevation in creatinine levels. This aligns with the initial expectation that diabetes conditions can impact kidney function, ultimately resulting in the accumulation of creatinine in the blood. On day 0, it is also evident that the negative control group (Group II) and Group III show a significant increase in creatinine levels compared to Group I, whereas the other groups exhibit a non-significant increase in comparison to Group I. This indicates that the immediate effects of the high-dose SALEE (300 and 400 mg/kg BW) can be observed right after administration.
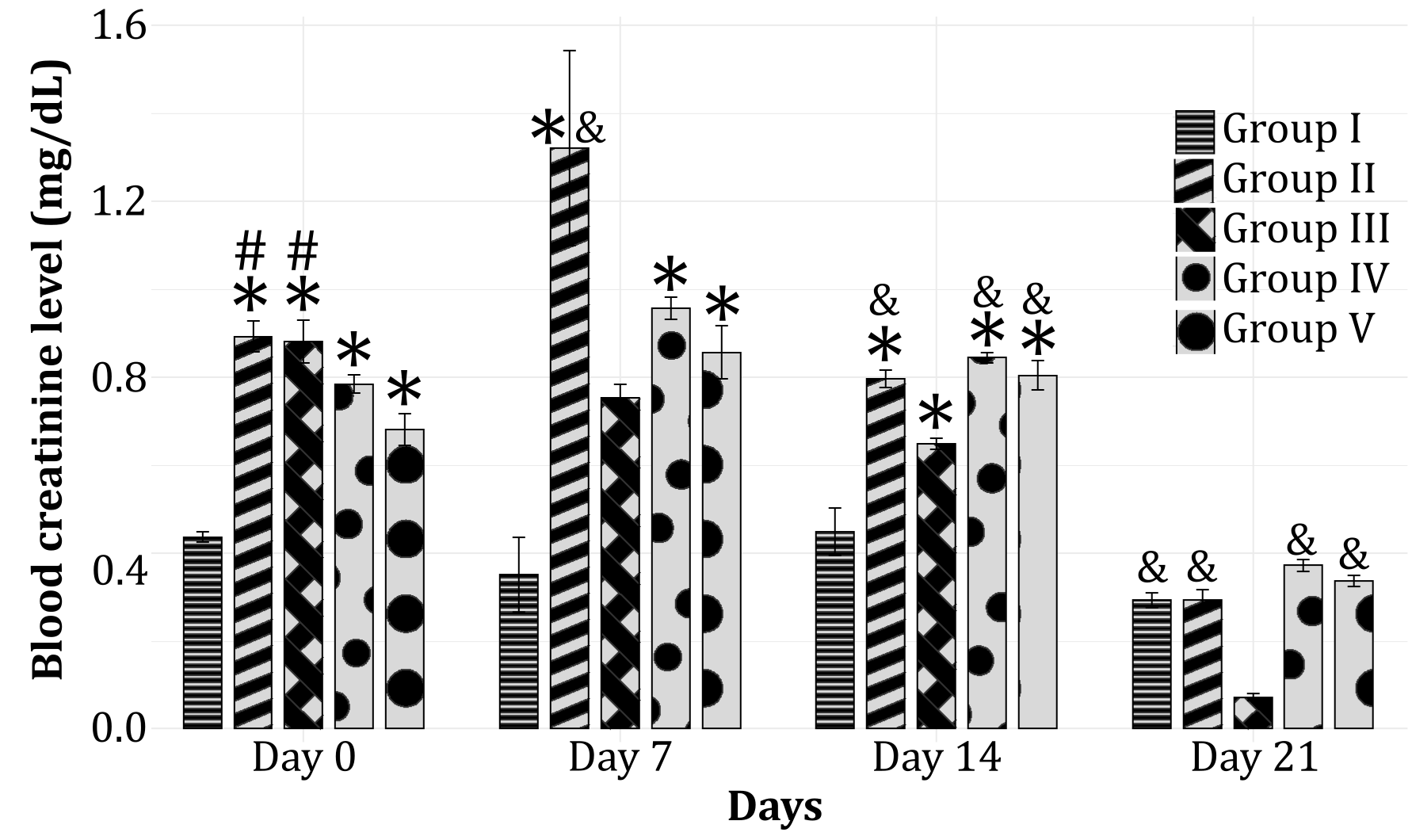
On the 7th day, the creatinine levels in Group II significantly increased, indicating a sustained accumulation in the blood. All diabetes-induced groups still maintained elevated levels on that day, but only Group III (receiving 200 mg/kg BW of SALEE) did not show a statistically significant difference in creatinine levels compared to healthy group. This suggests that a smaller dose of SALEE demonstrates better and consistent long-term effects, even though these effects are not immediately visible after administration as observed on day 0. By the 14th day, a decrease in creatinine levels began to be noticeable across all groups, though it remained significant compared to both Group I and III. A significant decrease in creatinine levels became evident on the 21st day, with Group III displaying the lowest creatinine levels. Based on these observations, it can be concluded that SALEE at a dose of 200 mg/kg BW is more efficient in reducing the accumulation of creatinine in the blood.
Urea level
Urea is the main metabolite resulting from the breakdown of dietary proteins and tissue protein turnover. It is relatively small molecules (around 60 Dalton) and it disperses throughout the body's total water content. Serum urea nitrogen (BUN) can be quantified from blood urea levels and constitutes roughly half (28/60 or 0.446) of the blood urea. Normal BUN levels range from 5-20 mg/dL, and the BUN-to-serum creatinine ratio is approximately 15: 1 (18). Based on this literature, the healthy control group (non-diabetic and untreated) demonstrates consistent results where their blood urea levels range from 15-20 mg/dL, resulting in BUN levels of approximately 6-9 mg/dL and serum creatinine levels of around 0.4 mg/dL. The obtained BUN-to-serum creatinine ratio ranges from 15: 1 to 22.5: 1. However, the observed blood urea levels appear to fluctuate from day to day. This fluctuation aligns with existing literature, which confirms that BUN is more influenced by dietary and physiological conditions than kidney function, leading to fluctuations over time (18).

In Figure 2, it can be observed that in Group IV (receiving 300 mg/kg BW of SALEE), the blood urea levels are the lowest and significantly different compared to the healthy control group (Group I). However, it is not possible to draw any conclusions at this point because the negative control group also experiences a reduction in blood urea. Nonetheless, the pattern of increase in blood urea is more evident in Group V, followed by Group IV on the final day of observation. As discussed earlier, although BUN accumulation can depict kidney function, BUN levels can also be influenced by dietary and physiological conditions (19). The increase in the BUN-to-serum creatinine ratio observed on days 14 and 21 in Group V may be influenced by a higher protein intake (18). This is supported by a study conducted by Yelvia N. et al. (2014), where the body weight of mice significantly increased on day 90, and an increase in appetite was indeed observed (20).
Conclusion
The ethanol extract of S. arvensis L leaves contains various secondary metabolite compounds, such as alkaloids, flavonoids, phenols, tannins, and steroids. SALEE has an impact on creatinine and urea levels in white male rats induced by streptozotocin. A dose of 200 mg/kg BW of the SALEE shows a gradual and consistent decrease in serum creatinine levels up to day 21, although immediate effects after administration were not observed as seen with doses of 300 and 400 mg/kg BW. Interestingly, BUN levels in the test subjects actually increased with higher doses of SALEE (400 mg/kg BW). However, BUN levels also showed fluctuations in the other groups, suggesting that the increase in BUN is influenced more by the test subjects' appetite and higher protein intake. Based on the findings, it can be concluded that suspension of SALEE (200 mg/kg BW) is a potential treatment or supplement for diabetics-induced renal failure.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
The study received approval from the Research Ethics Committee at the Faculty of Medicine, Tadulako University, with the assigned approval number 2576/UN.28.1.30/K/2019.
Funding Information
Not applicable.
References
- Vallon V, Komers R. Pathophysiology of the Diabetic Kidney. In: Comprehensive Physiology. Wiley; 2011. p. 1175–232.
- Thomas MC, Brownlee M, Susztak K, Sharma K, Jandeleit-Dahm KAM, Zoungas S, et al. Diabetic kidney disease. Nat Rev Dis Prim. 2015 Jul 30;1(1):15018.
- Dabla PK. Renal function in diabetic nephropathy. World J Diabetes. 2010;1(2):48.
- Wink M. Modes of Action of Herbal Medicines and Plant Secondary Metabolites. Medicines. 2015 Sep 8;2(3):251–86.
- Maghfiroh K, Rifa’i M, Widyarti S. Adaptive Immune Response Stimulation on Nephrolithiasis Mice Model after Treatment of Tempuyung (Sonchus arvensis L.) Leaf Extract. J Exp Life Sci. 2014 Dec 1;4(2):34–8.
- Wulandari TM, Chandra B, Zulharmita Z, Rivai H. An Overview of the Traditional Uses, Phytochemicals, and Pharmacological Activities of Tempuyung (Sonchus arvensis L.). Int J Pharm Sci Med. 2021 Jun 30;6(6):34–41.
- Hidayati A, Yusrin, Anggraini H. Pengaruh Frekuensi Penggunaan Teh Daun Tempuyung Kering (Soncsus arvensis) Terhadap Daya Larut. J Kesehat. 2009;2(2):30–7.
- Hidayat R, Reagan M, Hayati L. Tempuyung Leaves (Sonchus arvensis) Ameliorates Monosodium Urate Crystal-Induced Gouty Arthritis in Rats through Anti-Inflammatory Effects. Open Access Maced J Med Sci. 2020 May 21;8(A):220–4.
- Imelda I, Achadiyani A, Sekarwana N. Protective Effect of Ethanolic Extract Tempuyung Leaf (Sonchus arvensis L.) in Gentamicin-induced Acute Tubular Necrosis on Wistar Rats. Indones J Pharm. 2018 May 11;29(2):86.
- Bhalodiya M, Chavda J, Mori D, Patel N, Manek R, Dudhat K. Formulation and evaluation of amorphous solid dispersion Boerhaavia diffusa methanolic root extract for improving dissolution properties. J Drug Deliv Sci Technol. 2021 Dec;66:102740.
- Savjani KT, Gajjar AK, Savjani JK. Drug Solubility: Importance and Enhancement Techniques. ISRN Pharm. 2012 Jul 5;2012:1–10.
- Kathpalia H, Phadke C. Novel Oral Suspensions: A Review. Curr Drug Deliv. 2014 May 4;11(3):338–58.
- Batchelor HK, Marriott JF. Formulations for children: problems and solutions. Br J Clin Pharmacol. 2015 Mar 20;79(3):405–18.
- Trease GE, Evans WC. Pharmacognosy. London: Saunders Publishers. 15th Ed 584 p. 2002;
- Abba M, Usman S, Ahmad M, Tahir A, Umar A. Exploring the Antiepileptic Potential of Amaranthus spinosus: An Experimental Study in Albino Mice. Sci Pharm. 2023 Aug 5;2(3):106–14.
- Tandi J, Sutrisna INE, Pratiwi M, Handayani TW. Potential Test of Nephropathy Sonchus arvensis L. Leaves on Male Rats (Rattus norvegicus) Diabetes Mellitus. Pharmacogn J. 2020 Aug 5;12(5):1115–20.
- Thammitiyagodage MG, de Silva NR, Rathnayake C, Karunakaran R, WGSS K, Gunatillka MM, et al. Biochemical and histopathological changes in Wistar rats after consumption of boiled and un-boiled water from high and low disease prevalent areas for chronic kidney disease of unknown etiology (CKDu) in north Central Province (NCP) and its comparison wit. BMC Nephrol. 2020 Dec 31;21(1):38.
- Hosten AO. Clinical Methods: The History, Physical, and Laboratory Examinations. Ann Intern Med. 1990;113(7):563.
- Hammond AC. Use of BUN and MUN as Guides for Protein and Energy. Cienc Tecnol Agropecu. 1998;2(2):44–8.
- Nurianti Y, Hendriani R, Sukandar EY, Anggadiredja K. Acute and subchronic oral toxicity studies of ethyl acetate extract of sonchus arvensis L. leaves. Int J Pharm Pharm Sci. 2014;6(5):343–7.
