RESEARCH ARTICLE
Phytoconstituents, Fourier-Transform Infrared Characterization, and Antioxidant Potential of Ethyl Acetate Extract of Corchorus olitorius (Malvaceae)
Academic Editor: Adeleye Ademola Olutayo
Sciences of Phytochemistry|Vol. 3, Issue 1, pp. 1-10 (2024)
Received
Dec 26, 2023Revised
Jan 28, 2024Accepted
Feb 3, 2024Published
Feb 10, 2024
Abstract
Introduction
Oxidative stress is a state of imbalance where reactive oxygen species (ROS) are generated in excess amounts in the body compared to the available endogenous antioxidants. Diseases such as neurodegenerative diseases, diabetes mellitus, cancer, cardiovascular, and other several conditions are associated with oxidative stress (1-4), particularly during the progression stage of the diseases. Thus, creating a disturbance in the balance between the ROS generation and the number of endogenous antioxidants present. Oxidative stress has been coined as an important contributor to carcinogenesis (5). In cancer cells, ROS are generated at a higher rate to allow for the proliferation of the cancer cells. However, these cells generate high amounts of antioxidants to counter the ROS, thereby preventing the initiation of apoptosis or senescence (6). In neurodegenerative diseases, ROS species don't trigger these diseases. However, they are crucial in aggravating its progression through damaging mitochondrial integration. This leads to mitochondrial dysfunction (7), such conditions include Alzheimer's disease, Parkinson’s disease, and Huntington's disease.
Cardiovascular diseases are also associated with oxidative stress leading to the depletion of the available nitric oxide, vasoconstriction, and hypertension. The continued generation of excess ROS leads to disturbance in myocardial calcium handling, arrhythmia, increased cardiac remodeling, and formation of atherosclerotic plaque (8). In diabetes, an increased metabolic rate generates ROS leading to decreased insulin sensitivity by β-cell and mitochondrial impairment, decreased activity of GLUT-4, inflammatory responses, and disturbance in the insulin signaling pathway (9). Oxidative stress is associated with the progression of many diseases, thus, a vital target for the management of such diseases. Many antioxidant supplements are applied as a remedy for oxidative stress. However, the use of medicinal plants has been in practice for a very long time. Many plants are associated with bioactive compounds and phytochemicals with antioxidant potentials against ROS. These phytoconstituents exert their pharmacological activities against oxidative stress-linked diseases via different mechanisms (10-12). Previous studies reported the in vitro antioxidant activities of different plants demonstrating their radical scavenging activities of the plants (12-17). Thus, exhibiting their therapeutic potential against oxidative stress.
Corchorus olitorius is utilized in traditional medicinal practice in the treatment of malaria, fever, typhoid, and sexual weakness (18). Moreover, the plant is applied in the treatment of dysentery, pain management, and degenerative disorders and is regarded as a potential source of herbal therapeutics to promote health in rural communities in Africa (19). C. olitorius also serves as a source of food, phytoconstituents, and pharmaceutics with good potential for the treatment of diseases and as functional food (20). C. olitorius was also reported to exert antibacterial activities, supporting its traditional use against bacterial infections (21). The plant exerts antidiabetic activities with good potential in preventing the postprandial rise in glucose and safety profile (22). An anticancer study revealed that C. olitorius exerts its anticancer effects by inducing apoptosis and is effective against colon cancer. Thus, implying its potential in phytotherapy for the management of cancer as opposed to chemotherapy (23). In alcohol liver damage, C. olitorius ameliorates liver damage, insinuating its application for the management of alcohol-induced liver damage (24). Thus, in the present study, the phytoconstituents and antioxidant potential of C. olitorius were investigated to ascertain its potential in the management of oxidative stress-linked diseases.
Experimental Section
Materials
C. olitorius leaves were obtained from the Yola market of Yola South Local Government, Adamawa State, Nigeria. The plant was identified by a Forest Technologist from the Forestry Technology Department of Adamawa State Polytechnic, Yola where a voucher specimen (ASP/FT/128) was deposited.
The chemicals and reagents used were of analytical grade; Ethyl acetate (LOBA CHEMIE, Loba Chemie PVT. Ltd, India), Potassium ferrocyanide (Amichem Research Lab LLP, India), Trichloroacetic acid (Shree Vissnu Scientific Company, India), Ferric Chloride (Alpha Chemika, India), L-Ascorbic acid (Oxford Lab Fine Chem LLP, India), Sodium phosphate (Research Products International, USA), Ammonium molybdate (Scientific Syndicate, India), Linolenic acid (Shandong Zhi Shang Chemical Co. , Ltd. , China), Ethanol (Guangdong Guanghua Sci-Tech Co. , Ltd. , China), Ammonium thiocyanate (Naisha Enterprises, India), Thiobarbituric acid (Isolab Laborgeräte GmbH, Germany), Ferrous chloride (LOBA CHEMIE, Loba Chemie PVT. Ltd, India).
Methods
Sample Extraction
The leaves were dried under shade and ground to powder using a blender. The sample was extracted by the maceration of 200 g of the powder in 1 L of 80% (v/v) ethyl acetate with daily gentle agitation for 7 days at room temperature, followed by filtration and concentration to dryness with a rotary evaporator (Buchi Rotavapor R-200) at 150 rpm under reduced pressure at 40 oC (25).
Phytochemical Analysis
Phytochemicals present in ethyl acetate leaf extract of C. olitorius (ELEC) were identified using the method reported previously to detect alkaloids, saponins, steroids, glycosides, terpenoids, and flavonoids (25).
The quantification of phytochemicals was carried out by methods previously reported as follows:
Alkaloids Content
Alkaloids were quantified by the gravimetric method (21). Briefly, 0.5 g of the extract was dispensed into a conical flask containing 10 mL of 10 % ammonium hydroxide to convert alkaloidal salts into the free base; the mixture was stirred and left for 4 h before filtering. The filtrate was evaporated to one-quarter of its original volume on a water bath and concentrated ammonium hydroxide solution was added dropwise to the mixture to precipitate the alkaloids. The precipitate was filtered using a weighed filter paper and washed with 10% ammonium hydroxide solution. The precipitate was dried with the filter paper in an oven at 60°C for 30 minutes and reweighed. The percentage alkaloid content was calculated according to Equation 1.
Saponins
Quantification of saponins was done by the method previously described (26). Briefly, 0.5 g extract was introduced into a conical flask and 10 ml of 20% aqueous ethanol was added. The sample was heated over a water bath for 1 h with continuous stirring at about 55°C. The concentrate was transferred into a 250 ml separator funnel and 5 mL of diethyl ether was added and shaken vigorously. The aqueous layer was recovered and the ether layer was discarded. About 10 ml of n-butanol was then added followed by the addition of 2 ml of 5% aqueous NaCl. The remaining solution was heated over a water bath. After evaporation, the sample was dried in the oven to a constant weight. The percentage saponin content was calculated according to Equation 1.
Flavonoids
Quantification of flavonoids was done by the method described previously (27). Briefly, 0.5g of the extract was mixed with 10 mL of 80% aqueous methanol. The solution was filtered through the Whatman filter paper. The filtrate was transferred to a pre-weighed crucible evaporated into dryness over a water bath and weighed. The percentage flavonoid content was determined according to Equation 1.
Fourier-transform Infrared Spectroscopy (FTIR) Characterization
FTIR was carried out to determine the functional groups present in ELEC using the Perkin-Elmer FTIR (series 100). Exactly, 0.5 g of the sample was placed directly on the light path to collect the spectra of the reflectance. The readings were recorded with 4000 to 400 cm-1 wavelengths at 4 cm-1 resolution from 4 scans at 0.2 cm-1 s-1. Furthermore, the functional groups were determined from the interpretation of the spectrum using the IR spectrum Table (28).
Antioxidant Potential
Total Antioxidant Capacity (TAC)
The TAC was determined by the method described previously (24). Briefly, 0.5 ml of sample (300 µg/ml) was mixed with 2 ml of phospho-molybdate reagent (0.6 M H2SO4, 0.028 M sodium phosphate, and 1% ammonium molybdate) and capped. The mixture was incubated at 95 ℃ for 10 minutes, followed by reading the absorbance at 695 nm against a blank after cooling to room temperature. The blank was made up of the reaction mixtures without the sample. Additionally, different concentrations (20, 40, 60, 80, and 100 µg/ml) of AA were used to obtain the standard calibration curve of concentration versus absorbance. The TAC was expressed as AAE in µg/ml. All values were determined in triplicates.
Total Reducing Power (TRP)
The TRP was determined as previously described (29). Briefly, 0.25 ml of the sample (200 µ/ml) was mixed with 0.625 ml of 1% (w/v) potassium ferrocyanide and 0.625 ml of 0.2 M phosphate buffer (pH 6.6). The mixture was incubated at 50oC for 20 minutes and 0.625 ml of 10% (w/v) trichloroacetic acid (TCA) was added, followed by 10 minutes of centrifugation at 3000 rpm and collection of 1.8 ml of the upper layer. Exactly, 1.8 ml of distilled water and 0.36 ml of 0.1% FeCl3 were added and the absorbance was read at 700 nm against a blank solution of distilled water and the reagents without samples. Additionally, different concentrations (20, 40, 60, 80, and 100 µg/ml) of ascorbic acid (AA) treated similarly to the sample were used to obtain the standard calibration curve and determine the ascorbic acid equivalent (AAE) of the samples in µg/ml. The TRP was expressed as AAE in µg/ml. All values were determined in triplicates.
Ferric Thiocyanate Method (FTC)
The procedure previously described (30) was used to determine the lipid peroxidation inhibitory potential of the extracts. Briefly, 4.1 and 4 ml of 2.52% (v/v) linolenic acid and 1 mg/ml of sample, respectively dissolved in absolute ethanol were mixed with 0.05 M phosphate buffer (pH 7) and 3.9 ml of distilled water. The mixture was capped and incubated for 10 minutes in a dark oven at 40 ℃. Exactly 0.1 ml of the solution was mixed with 9.7 ml of 75% (v/v) ethanol, 0.1 ml of 30% (w/v) ammonium thiocyanate, and 0.1 ml 0.02 M ferrous chloride in 3.5% (v/v) HCl. The absorbance was initially read 532 nm after adding ferrous chloride. Furthermore, AA was used as standard while the reaction mixture without the sample was used as control. The absorbance was read every 24 hours until the absorbance of the control reached maximum. The percentage inhibition was determined according to Equation 2. All values were determined in triplicates.
Where At = Absorbance of sample while Ac = Absorbance of control.
Thiobarbituric Acid Method (TBA)
The protocol of Kikuzaki and Nakatani (30) was adopted to determine the inhibitory effects of the samples on lipid peroxidation. Exactly 1 ml of the sample and AA solutions from the FTC method were separately mixed with 2 ml of 20% (w/v) TCA and 2 ml of 0.5% (w/v) thiobarbituric acid solution. This was followed by capping, 10 minutes of incubation at 90 ℃ in a water bath, and 20 minutes of centrifugation at 3000 rpm after cooling. The reaction mixture without the sample or AA was used as a control. The absorbance of the supernatant was read at 532 nm on the last day of the FTC method described earlier. The malonaldehyde (MDA) concentration was determined by Equation 3 using the extinction coefficient 156 mM-1 cm-1 as previously described (31). All values were determined in triplicates.
Where OD = Absorbance of the sample while EC = Extinction coefficient
Statistical analysis
The result obtained was expressed as mean ± standard error of the mean of triplicate determinations (± SEM) and evaluated by independent-samples T-test at p < 0.05 level of significance, using Statistical Package for the Social Sciences (SPSS) version 22 Software.
Result and Discussion
Phytoconstituents of ELEC
The phytochemicals identified in ELEC are presented in Table 1. Alkaloids, saponins, and flavonoids were present while steroids glycoside and terpenoids were absent. Phytochemicals play vital therapeutic roles in the management of different ailments including oxidative stress (32-35).
| Phytochemical | Inference |
|---|---|
| Alkaloids | Present |
| Saponins | Present |
| Steroids | Absent |
| Glycosides | Absent |
| Terpenoids | Absent |
| Flavonoids | Present |
Alkaloids were reported to influence oxidative stress by inhibiting NADPH-oxidase activity and activating nuclear Nrf2 pathway (36). Neurodegenerative diseases that are linked with oxidative stress were reported to be alleviated by alkaloids, acting as an agonist of muscarinic and adenosine receptors, and also inhibition of anti-amyloid, acetylcholinesterase, butyrylcholinesterase, and α-synuclein aggregation (37). The alkaloid boldine was reported to exert protective effects on the endothelium by preventing the depletion of nitric oxide caused by ROS, thereby, inhibiting the oxidative stress pathway, vital in diabetes and hypertension (38).
Saponins were reported to possess strong antioxidant properties and are implicated in the restoration of mitochondrial function and calcium balance in cells, in addition to inhibiting inflammatory factor synthesis and apoptosis in heart cells (39, 40). Saponins were also reported to exert antioxidant properties by regulating oxidative stress and preventing neuroinflammation and apoptosis (41). In other studies, saponins were reported to exert antioxidants on hypertension by blocking ROS generation and regulating rheology in red blood cells (42, 43). Saponins also elevate the concentrations of endogenous antioxidants, catalase, and glutathione in serum (44).
Flavonoids exert antioxidative effects by acting on alpha gamma-aminobutyric acid (α-GABA) in epilepsy, associated with elevated production of ROS during seizures, thus showing potential as a therapeutic in epilepsy (45). The generation of ROS is an important factor in the progression of neurodegenerative diseases. Flavonoids act as antioxidants and signaling molecules in slowing down the progression of the disease and improving cognitive performance (46). Quercetin which is another flavonoid exerts neuroprotective properties by preventing ROS damage to DNA through mild intercalation (46). Flavonoids were reported to exert an anti-inflammatory effect credited to their antioxidant properties acting by inhibiting pathways that lead to the generation of ROS (47).
The phytochemicals quantified in the ELEC are shown in Table 2. Flavonoids had the highest concentration (19.17% ± 0.73), followed by saponins with a concentration of 11.83% ±0.73 while alkaloids were quantified in the lowest concentration (8.50% ± 2.65).
| Phytochemical | Concentration (%) |
|---|---|
| Alkaloids | 8.50 ±2.65 |
| Saponins | 11.83 ±0.73 |
| Flavonoids | 19.17 ±0.73 |
Values are in triplicate determinations ± SEM.
In a study reported previously, alkaloids, saponins, and flavonoids were detected in leaves of C. olitorius which correlates with the result of the present study (48). The present study also agrees with previous studies for the detection of flavonoids, alkaloids, and saponins in the leaf of C. olitorius (49, 50). In another study on Jute whole plant, only flavonoids were detected with the absence of alkaloids and saponins in the ethyl acetate extract (51). The present study doesn’t agree with this study as both saponins and alkaloids were present in ELEC. The present study partially agrees with this study as glycosides were not detected, which might be due to the difference in solvents used for extraction (52). In a similar study, alkaloids, saponins, and flavonoids were detected in ethanol extract of C. olitorius in concentrations of 2.40 ±0.10, 3.32 ±0.02, and 6.30% ±0.10, respectively (53), lower than the values reported in our study (Table 2). The difference in the concentrations might be attributed to the difference in the polarity of solvents used in extraction (52).
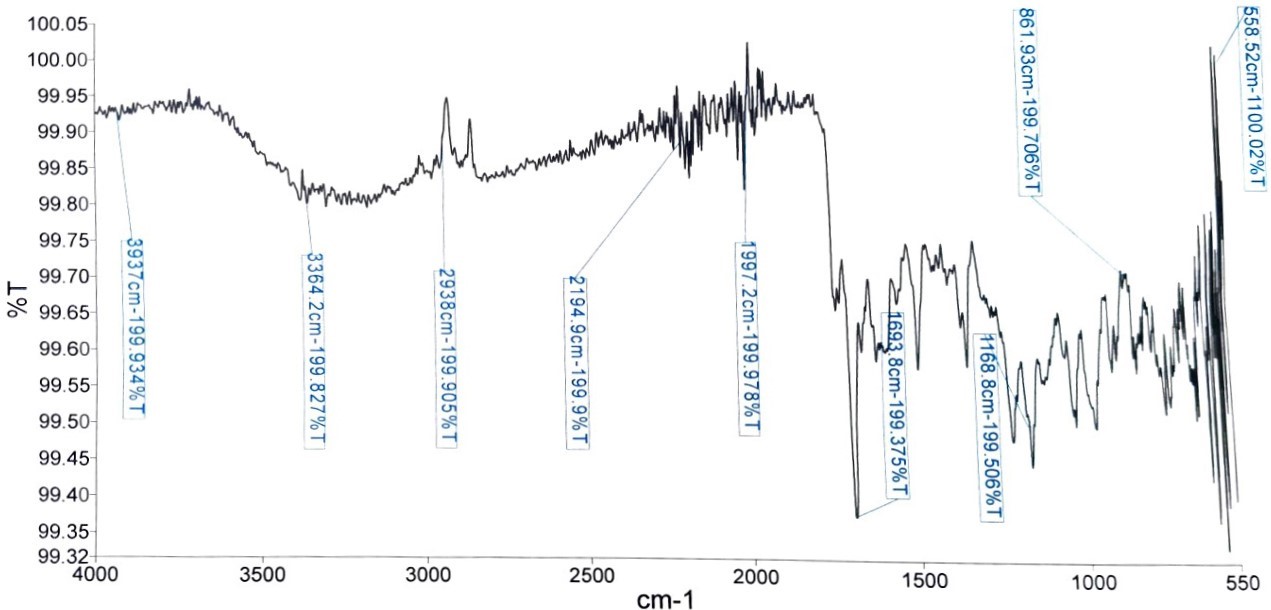

Fourier-Transform Infrared (FTIR) Characterization
Figure 1 shows the FTIR spectrum of the ELEC depicting the various peaks. A total of 9 peaks were detected, including six at the group frequency region. The first absorption peak was at 3937.00 cm-1 corresponding to the O-H stretching frequency of free alcohol. The second (3364.2 cm-1) and third (2938.00 cm-1) peak at the frequency regions corresponds to the O-H stretching and N-H stretching frequencies of carboxylic acid and amine salt, respectively. The 2194.9 and 1997.2 cm-1 peaks correspond to the C≡C stretching and C=C=C stretching frequencies of monosubstituted alkyne and allene respectively while 1693.8 cm-1 corresponds to the C=N stretching of imine/oxime. In the fingerprint region, the peak at 1168.8 (C-O stretching), 861.93 cm-1 (C-H bending), and 558.52 (C-I stretching) fingerprint regions corresponds to the ester, trisubstituted alkane, and halo compound, respectively.
The FITR spectrum of ELEC presented in Figure 1 shows the presence of different functional groups. Hydroxyl groups were reported to contribute significant antioxidant activities for compounds, including flavonoids and polyphenols, attributed to the electron-donating effects of the OH groups (54). Additionally, the presence of the OH group might also be attributed to the flavonoids detected. The carboxylic acid functional groups detected in the present study were reported to contribute to the antioxidant properties of molecules via transition metal ions chelation (55). Furthermore, the amine groups reported in our study were reported to enhance the antioxidant activities of genistein and its derivatives (56). The presence of oxime was previously reported to enhance the antioxidant activity of naringin compared to pure naringin (57). Additionally, the anti-inflammatory, antioxidant, and anticancer activities of oximes were previously reported (58). Thus, the presence of these functional groups in ELEC might contribute to the antioxidant activities of the plant reported in our study.
Antioxidant Potential of ELEC
Figure 2 presents the TAC and TRP of ELEC, depicting the AA standard calibration curves and the AAE TAC and TRP. The ELEC exhibited a significantly (p < 0.05) higher TAC (49.26 ±2.44 AAE µg/ml) compared to its TRP (11.06 ±1.34 AAE µg/ml).
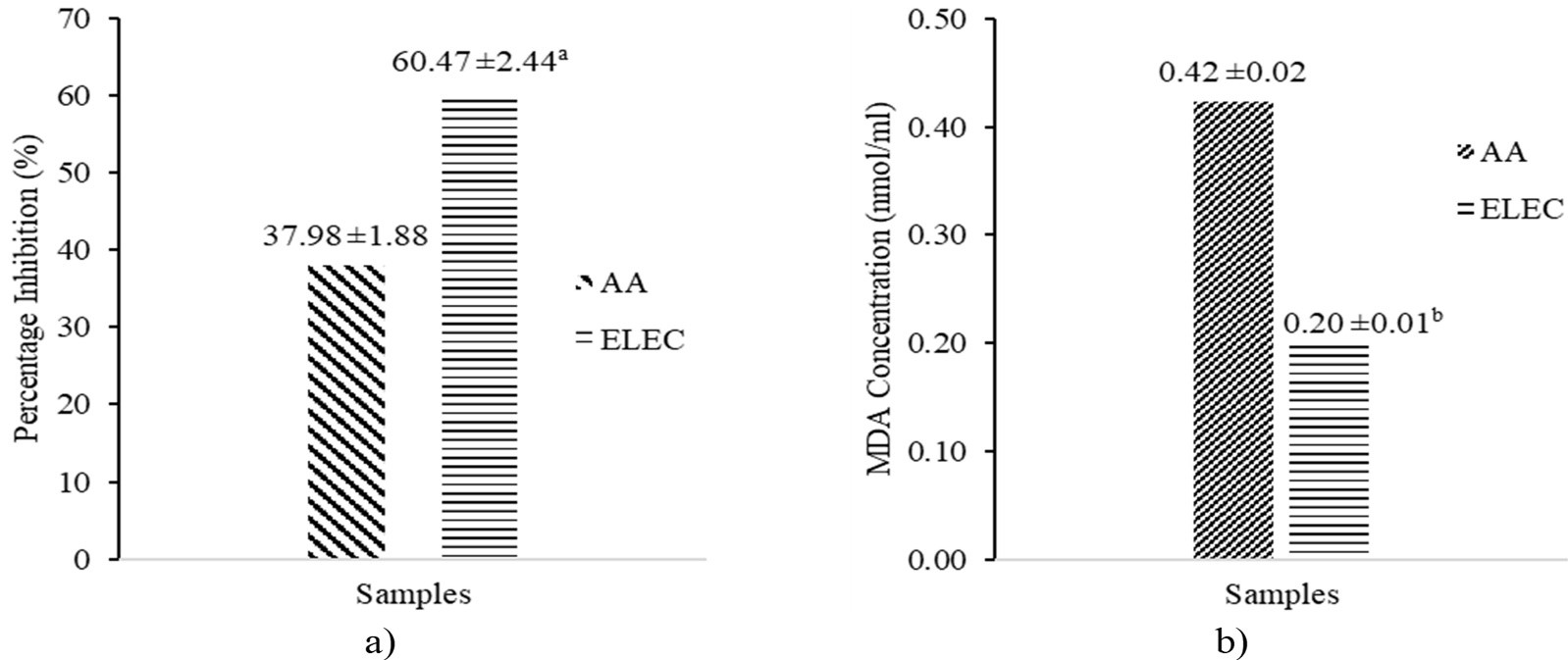
The lipid anti-peroxidation potential of ELEC by the FTC and TBA method is presented in Figure 3 depicting the percentage inhibition and MDA concentration. The ELEC exhibited a significantly (p < 0.05) higher (60.47% ±2.44) percentage inhibition than AA (37.98% ±1.88). Furthermore, the ELEC showed a significantly (p < 0.05) lower (0.20 ±0.01 nmol/ml) MDA concentration than AA (0.42 ±0.02 nmol/ml).
Free radicals are generated in normal metabolic pathways, however, the presence of endogenous antioxidants that absorb the free radicals prevents oxidative stress due to ROS. Problems arise when the free radicals are produced in amounts higher than can be absorbed by the endogenous antioxidant, a characteristic of many ailments (1-4). A previous study identified phytochemicals such as alkaloids, saponins, and flavonoids to be responsible for the antioxidant properties of plants (59). In another study, these phytochemicals were reported to exert antioxidant effects against lipid peroxidation (53). In our study, the functional groups identified were previously attributed to different antioxidant activities. AA used in the present study exhibited higher TAC and TRP than ELEC which be attributed to its higher antioxidant capacity and the polar nature of the test medium (60). The electron-donating ability of a compound is closely related to its reducing power. In our study, the lower TRP of ELEC might be due to the presence of compounds with poor electron-donating abilities (61). Although AA (polar antioxidant) demonstrated superior TAC and TRP, ELEC exhibited better anti-lipid peroxidation as observed in the FTC and TBA methods where the test medium was non-polar. Moreover, the slightly polar properties of ethyl acetate might lead to the extraction of non-polar antioxidants. Thus, lower peroxidation inhibition and higher MDA concentrations compared to ELEC were observed.
Flavonoids were previously reported to be associated with high free radical scavenging exerting high reducing power and anti-lipid peroxidation (62). In another study, Zanthoxylum armatum seeds and barks were reported to exert higher TAC than AA attributed to their flavonoid contents. Furthermore, a correlation was observed between the antioxidant activity and flavonoid content (63). In another study, Saponins exhibited a concentration-dependent reducing power and anti-lipid peroxidation comparable to tannic acid demonstrating its strong antioxidant activity (64). In a similar study, the antioxidant properties of C. olitorius using DPPH and ATBS increased with increased concentration (65). C. olitorius was previously reported to exert antioxidant properties, attributed to the flavonoids and saponins detected in the study (66). Similarly, a study on the methanol extract of C. olitorius using DPPH revealed a concentration-dependent increase in the antioxidant activity of the plant, credited to the phytochemicals reported in the study (49). A similar study reported the antioxidant and antidiabetic effects of the plant and was suggested to be a source of therapeutic for diabetes (67). Ethanol extract C. olitorius was previously reported to possess antioxidant properties which were attributed to hydroxyl groups of phenolic compounds (68). The present study revealed the antioxidant potential of ELEC which might be attributed to the phytochemicals and their functional groups.
Conclusion
The present study revealed the presence of phytochemicals that were previously reported to possess different pharmacological properties including antioxidant effects. The presence of alkaloids (8.50% ± 2.65), saponins (11.83 ± 0.73), and flavonoids (19.17% ± 0.73) in the ELEC might contribute to the TAC (49.26 ± 2.44 AAE µg/ml) and TRP (11.06 ±1.34 AAE µg/ml) exhibited by the extract. Furthermore, these phytochemicals might contribute to the higher anti-lipid peroxidation (60.47% ± 2.44 percentage inhibition and 0.20 ± 0.01 nmol/ml MDA concentration) than AA (37.98% ±1.88 percentage inhibition and 0.42 ±0.02 nmol/ml MDA concentration). Oxidative stress is associated with several diseases, thus, the antioxidant and anti-lipid peroxidation potential demonstrated by C. olitorius makes it a good candidate for the management of oxidative stress in such diseases and might also be a vital source of novel therapeutics against oxidative stress-linked diseases. However, further studies are recommended to identify the specific compounds and their mechanism of action.
Declarations
Acknowledgment
The authors express their appreciation to the Departments of Science Laboratory Technology and Pharmaceutical Technology, Adamawa State Polytechnic, Yola for institutional support in carrying out the research.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Funding Information
Not applicable.
References
- Sies H. Oxidative stress: a concept in redox biology and medicine. Redox biology. 2015;4:180-3.
- Hayes JD, Dinkova-Kostova AT, Tew KD. Oxidative Stress in Cancer. Cancer Cell. 2020;38(2):167-97.
- Kattoor AJ, Pothineni NVK, Palagiri D, Mehta JL. Oxidative Stress in Atherosclerosis. Current Atherosclerosis Reports. 2017;19(11):42.
- Salim S. Oxidative Stress and the Central Nervous System. J Pharmacol Exp Ther. 2017;360(1):201.
- Klaunig JE. Oxidative Stress and Cancer. Curr Pharm Des. 2018;24(40):4771-8.
- Redza-Dutordoir M, Averill-Bates DA. Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica et Biophysica Acta (BBA)-Molecular Cell Research. 2016;1863(12):2977-92.
- Dias V, Junn E, Mouradian MM. The Role of Oxidative Stress in Parkinson's Disease. J Parkinsons Dis. 2013;3:461-91.
- Senoner T, Dichtl W. Oxidative Stress in Cardiovascular Diseases: Still a Therapeutic Target? Nutrients [Internet]. 2019; 11(9).
- Yaribeygi H, Sathyapalan T, Atkin SL, Sahebkar A. Molecular Mechanisms Linking Oxidative Stress and Diabetes Mellitus. Oxid Med Cell Longev. 2020;2020:8609213.
- Hassan W, Noreen H, Rehman S, Gul S, Amjad Kamal M, Paul Kamdem J, et al. Oxidative Stress and Antioxidant Potential of One Hundred Medicinal Plants. Curr Top Med Chem. 2017;17(12):1336-70.
- Al-Snafi AE. Medicinal plants possessed antioxidant and free radical scavenging effects (part 3)-A review. IOSR Journal of Pharmacy. 2017;7(4):48-62.
- Gholamian-Dehkordi N, Luther T, Asadi-Samani M, Mahmoudian-Sani MR. An overview on natural antioxidants for oxidative stress reduction in cancers; a systematic review. Immunopathol Persa. 2017;3(2):e12.
- Chigurupati S, Aladhadh HS, Alhowail A, Selvarajan KK, Bhatia S. Phytochemical composition, antioxidant and antidiabetic potential of methanolic extract from Corchorus olitorius Linn. grown in Saudi Arabia. Medicinal Plants-International Journal of Phytomedicines and Related Industries. 2020;12(1):71-6.
- Dahiru MM, Ahmadi H, Faruk MU, Aminu H, Hamman AGC. Phytochemical Analysis and Antioxidant Potential of Ethylacetate Extract of Tamarindus Indica (Tamarind) Leaves by Frap Assay. Journal of Fundamental and Applied Pharmaceutical Science. 2023;3(2):45-53.
- Daniel AI, Gara TY, Ibrahim YO, Muhammad FM, Salisu FE, Tsado R, et al. In vivo antidiabetic and antioxidant activities of chloroform fraction of Nelsonia canescens Leaf in Alloxan-induced Diabetic Rats. Pharmacological Research-Modern Chinese Medicine. 2022;3:100106.
- Dubey K, Dubey R, Gupta RA, Gupta AK. Anti-diabetic and antioxidant potential of saponin extract of leaves of Ziziphus mauritiana. Journal of Drug Delivery and Therapeutics. 2019;9(2-A):75-7.
- Dahiru MM, Nadro MS. Phytochemical Composition and Antioxidant Potential of Hyphaene thebaica Fruit. Borneo Journal of Pharmacy. 2022;5(4):325-33.
- Adebo HO, Ahoton LE, Quenum FJ, Adoukonou-Sagbadja H, Bello DO, Chrysostome CA. Ethnobotanical knowledge of jute (Corchorus olitorius L.) in Benin. Journal of Medicinal Plants. 2018;26(1):1-11.
- Kumari N, Choudhary SB, Sharma HK, Singh BK, Kumar AA. Health-promoting properties of Corchorus leaves: A review. Journal of Herbal Medicine. 2019;15:100240.
- Biswas A, Dey S, Huang S, Deng Y, Birhanie ZM, Zhang J, et al. A Comprehensive Review of C. capsularis and C. olitorius: A Source of Nutrition, Essential Phytoconstituents and Pharmacological Activities. Antioxidants [Internet]. 2022; 11(7).
- Namwase H, Najjuka F, Bbosa G. Anti-bacterial activity of Corchorus olitorius L. and Acmella caulirhiza Del. On Streptococcus mutans, a cariogenic bacterium. Afr Health Sci. 2021;21(4):1685-91.
- Nakaziba R, Lubega A, Ogwal-Okeng J, Alele PE. Phytochemical Analysis, Acute Toxicity, as well as Antihyperglycemic and Antidiabetic Activities of Corchorus olitorius L. Leaf Extracts. The Scientific World Journal. 2022;2022:1376817.
- Soykut G, Becer E, Calis I, Yucecan S, Vatansever S. Apoptotic effects of Corchorus olitorius L. leaf extracts in colon adenocarcinoma cell lines. Prog Nutr. 2018;20(4):689-98.
- Do MH, Lee HHL, Kim Y, Lee H-B, Lee E, Park JH, et al. Corchorus olitorius L. ameliorates alcoholic liver disease by regulating gut-liver axis. Journal of Functional Foods. 2021;85:104648.
- Evans WC. Trease and Evans' pharmacognosy: Elsevier Health Sciences; 2009.
- Obadoni B, Ochuko P. Phytochemical studies and comparative efficacy of the crude extracts of some haemostatic plants in Edo and Delta States of Nigeria. Global Journal of pure and applied sciences. 2002;8(2):203-8.
- Harborne A. Phytochemical methods a guide to modern techniques of plant analysis: springer science & business media; 1998.
- Merck KGaA. IR Spectrum Table & Chart: Merck KGaA; 2023 [cited 2024. Available from: https://www.sigmaaldrich.com/NG/en/technical-documents/technical-article/analytical-chemistry/photometry-and-reflectometry/ir-spectrum-table.
- Oyaizu M. Studies on products of browning reaction antioxidative activities of products of browning reaction prepared from glucosamine. The Japanese journal of nutrition and dietetics. 1986;44(6):307-15.
- Kikuzaki H, Nakatani N. Antioxidant effects of some ginger constituents. J Food Sci. 1993;58(6):1407-10.
- Kwon TW, Watts B. Determination of Malonaldehyde by Ultraviolet Spectrophotometry. J Food Sci. 2006;28:627-30.
- Ranjan A, Ramachandran S, Gupta N, Kaushik I, Wright S, Srivastava S, et al. Role of Phytochemicals in Cancer Prevention. International Journal of Molecular Sciences [Internet]. 2019; 20(20).
- Velmurugan BK, Rathinasamy B, Lohanathan BP, Thiyagarajan V, Weng C-F. Neuroprotective Role of Phytochemicals. Molecules [Internet]. 2018; 23(10).
- Forni C, Facchiano F, Bartoli M, Pieretti S, Facchiano A, D’Arcangelo D, et al. Beneficial Role of Phytochemicals on Oxidative Stress and Age-Related Diseases. BioMed Research International. 2019;2019:8748253.
- Teoh SL, Das S. Phytochemicals and their effective role in the treatment of diabetes mellitus: a short review. Phytochemistry Reviews. 2018;17(5):1111-28.
- Macáková K, Afonso R, Saso L, Mladěnka P. The influence of alkaloids on oxidative stress and related signaling pathways. Free Radic Biol Med. 2019;134:429-44.
- Hussain G, Rasul A, Anwar H, Aziz N, Razzaq A, Wei W, et al. Role of plant derived alkaloids and their mechanism in neurodegenerative disorders. Int J Biol Sci. 2018;14(3):341.
- Lau YS, Ling WC, Murugan D, Mustafa MR. Boldine ameliorates vascular oxidative stress and endothelial dysfunction: Therapeutic implication for hypertension and diabetes. J Cardiovasc Pharmacol. 2015;65(6):522.
- Zhang S-W, Liu Y, Wang F, Qiang J, Liu P, Zhang J, et al. Ilexsaponin A attenuates ischemia-reperfusion-induced myocardial injury through anti-apoptotic pathway. PLoS One. 2017;12(2):e0170984.
- Yin B, Hou X-w, Lu M-l. Astragaloside IV attenuates myocardial ischemia/reperfusion injury in rats via inhibition of calcium-sensing receptor-mediated apoptotic signaling pathways. Acta Pharmacol Sin. 2019;40(5):599-607.
- Ikram M, Jo MH, Choe K, Khan A, Ahmad S, Saeed K, et al. Cycloastragenol, a Triterpenoid Saponin, Regulates Oxidative Stress, Neurotrophic Dysfunctions, Neuroinflammation and Apoptotic Cell Death in Neurodegenerative Conditions. Cells [Internet]. 2021; 10(10).
- Zhang M, Guan Y, Xu J, Qin J, Li C, Ma X, et al. Evaluating the protective mechanism of panax notoginseng saponins against oxidative stress damage by quantifying the biomechanical properties of single cell. Anal Chim Acta. 2019;1048:186-93.
- Pan C, Huo Y, An X, Singh G, Chen M, Yang Z, et al. Panax notoginseng and its components decreased hypertension via stimulation of endothelial-dependent vessel dilatation. Vascul Pharmacol. 2012;56(3-4):150-8.
- Wang Y, Yang Z, Wei L, Ma X, Zhenping W. Research progress of saponins in Panax notoginseng and their pharmacological effects. Chinese herbal medicine. 2015;46(9):1381-92.
- Diniz TC, Silva JC, Lima-Saraiva SRGd, Ribeiro FPRdA, Pacheco AGM, de Freitas RM, et al. The Role of Flavonoids on Oxidative Stress in Epilepsy. Oxidative Medicine and Cellular Longevity. 2015;2015:171756.
- Simunkova M, Alwasel SH, Alhazza IM, Jomova K, Kollar V, Rusko M, et al. Management of oxidative stress and other pathologies in Alzheimer’s disease. Arch Toxicol. 2019;93(9):2491-513.
- Hussain T, Tan B, Yin Y, Blachier F, Tossou MCB, Rahu N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid Med Cell Longev. 2016;2016:7432797.
- Mohammed RMO. Phytochemical investigation of antimicrobial and antioxidant activity leaves extracts of Corchorus olitorius. Open Access Library Journal. 2016;3(1):1-5.
- Sadat A, Hore M, Chakraborty K, Roy S. Phytochemical analysis and antioxidant activity of methanolic extract of leaves of Corchorus olitorius. Int J Curr Pharm Res. 2017;9(5):59-63.
- Hasan HT, Kadhim EJ. Phytochemical investigation of Corchorus olitorius L. leaves cultivated in Iraq and it’s in vitro antiviral activity. Iraqi Journal of Pharmaceutical Sciences (IJPS). 2018:115-22.
- Patil DK, Jain AP. Extraction, qualitative and quantitative determination of secondary metabolites of Corchorus olitorius. Journal of Drug Delivery and Therapeutics. 2019;9(1-s):252-5.
- Mohammed Junaid Hussain D, Sathish Kumar K, Darul Raiyaan GI, Mohamed Khalith SB, Sundarapandian S, Kantha Deivi A. Effect of Solvents on Phytochemical Composition and Antioxidant Activity of Cardiospermum halicacabum (L.) Extracts. Pharmacognosy Journal. 2020;12(6).
- Ekpo G, Eteng OE, Bassey S, Leah A, Abam K, Ufort S, et al. Determination of Proximate Composition and phytochemical constituents of Corchorus oliterious (Ewedu) harvested in Ugep. Cross River State. Journal of Pharmaceutical Research International. 2019;27(3):1-12.
- Moalin M, van Strijdonck GPF, Beckers M, Hagemen GJ, Borm PJ, Bast A, et al. A planar conformation and the hydroxyl groups in the B and C rings play a pivotal role in the antioxidant capacity of quercetin and quercetin derivatives. Molecules. 2011;16(11):9636-50.
- Godlewska-Żyłkiewicz B, Świsłocka R, Kalinowska M, Golonko A, Świderski G, Arciszewska Ż, et al. Biologically Active Compounds of Plants: Structure-Related Antioxidant, Microbiological and Cytotoxic Activity of Selected Carboxylic Acids. Materials [Internet]. 2020; 13(19).
- Wang L, Yang F, Zhao X, Li Y. Effects of nitro- and amino-group on the antioxidant activity of genistein: A theoretical study. Food Chemistry. 2019;275:339-45.
- Özyürek M, Akpınar D, Bener M, Türkkan B, Güçlü K, Apak R. Novel oxime based flavanone, naringin-oxime: Synthesis, characterization and screening for antioxidant activity. Chem Biol Interact. 2014;212:40-6.
- Surowiak AK, Lochyński S, Strub DJ. Unsubstituted Oximes as Potential Therapeutic Agents. Symmetry [Internet]. 2020; 12(4).
- Raaman N. Antioxidant activites and phytochemical analysis of methanol extract of leaves of Hygrophila auriculata (Schumach) heine. Int J Curr Pharm Res. 2015;7(4):100-5.
- Norma Francenia S-S, Raúl S-C, Claudia V-C, Beatriz H-C. Antioxidant Compounds and Their Antioxidant Mechanism. In: Emad S, editor. Antioxidants. Rijeka: IntechOpen; 2019. p. Ch. 2.
- Mohamed H, Ons M, Yosra ET, Rayda S, Neji G, Moncef N. Chemical composition and antioxidant and radical‐scavenging activities of Periploca laevigata root bark extracts. J Sci Food Agric. 2009;89(5):897-905.
- Adebiyi OE, Olayemi FO, Ning-Hua T, Guang-Zhi Z. In vitro antioxidant activity, total phenolic and flavonoid contents of ethanol extract of stem and leaf of Grewia carpinifolia. Beni-Suef University Journal of Basic and Applied Sciences. 2017;6(1):10-4.
- Dibacto REK, Tchuente BRT, Nguedjo MW, Tientcheu YMT, Nyobe EC, Edoun FLE, et al. Total Polyphenol and Flavonoid Content and Antioxidant Capacity of Some Varieties of Persea americana Peels Consumed in Cameroon. The Scientific World Journal. 2021;2021:8882594.
- Akinpelu BA, Igbeneghu OA, Awotunde AI, Iwalewa EO, Oyedapo OO. Antioxidant and antibacterial activities of saponin fractions of Erythropheleum suaveolens (Guill. and Perri.) stem bark extract. Scientific Research and Essays. 2014;9(18):826-33.
- Oh S, Kim D-Y. Characterization, Antioxidant Activities, and Functional Properties of Mucilage Extracted from Corchorus olitorius L. Polymers [Internet]. 2022; 14(12).
- Biswas A, Dey S, Li D, Liu Y, Zhang J, Huang S, et al. Comparison of Phytochemical Profile, Mineral Content, and In Vitro Antioxidant Activities of Corchorus capsularis and Corchorus olitorius Leaf Extracts from Different Populations. Journal of Food Quality. 2020;2020:2931097.
- Chigurupati S, Aladhadh HS, Alhowail A, Selvarajan KK, Bhatia S. Phytochemical composition, antioxidant and antidiabetic potential of methanolic extract from Corchorus olitorius Linn. grown in Saudi Arabia. Int J Phytomed Related Ind. 2020;12:71-6.
- Hussien NM, Labib SE, El-Massry RA, Hefnawy HTM. Phytochemical Studies and Antioxidant Activity of Leaves Extracts of Corchorus olitorius L.(MOLOKHIA). Zagazig Journal of Agricultural Research. 2017;44(6):2231-9.