REVIEW
A Comprehensive Review on Nutrient Profile and Pharmacological Benefits of Musa paradisiaca
Sciences of Phytochemistry|Vol. 3, Issue 2, pp. 123-143 (2024)
Received
Jun 1, 2024Revised
Nov 5, 2024Accepted
Nov 13, 2024Published
Nov 27, 2024
Abstract
Keywords:
Introduction
Banana (family Musaceae) is one of the world’s most widely cultivated fruit crops and has been a staple for humanity since 600 BC. Known for its accessibility, year-round availability, and exceptional nutritional value, the banana is often referred to as the "plant of virtues" (Kalpataru) due to its extensive applications and economic importance. This complex hybrid, derived from Musa acuminata and Musa balbisiana, is native to Asia but widely distributed across tropical regions worldwide, particularly in South and Southeast Asia and the western tropical Pacific Ocean (1-3). Bananas offer unique nutritional and medicinal properties, with all parts, including flesh and peel, usable in products like chips, powder, biscuits, and juice (4). Bananas are an affordable, nutrient-dense food source, rich in carbohydrates, dietary fiber, essential minerals, and vitamins that support human health (5). Furthermore, bananas contain numerous bioactive phytochemicals, including plant sterols, carotenoids, biogenic amines, and phenolic compounds, all playing significant roles in health by combating inflammation, cancer, and diabetes and serving as antioxidants (6). The banana peel extracts have demonstrated wound-healing properties by enhancing mucosal cell proliferation and DNA synthesis (7).
Despite these notable benefits, a comprehensive review of bananas’ dietary and phytochemical profiles is essential to better understand their wide-reaching pharmacological potential. Given the diversity in banana cultivars, this review addresses the challenges in standardizing banana-derived products and examines the phytochemicals’ specific roles in preventing chronic diseases and enhancing well-being. Additionally, it seeks to evaluate gaps in current knowledge, emphasizing the need for more research into optimal processing and formulation methods to retain and maximize the pharmacological efficacy of banana-based treatments. This review highlights the enormous potential of bananas and considers future research avenues to fully utilize their therapeutic power for human health at a time when the demand for easily accessible and natural nutraceuticals is rising.
Methodology
A comprehensive review of 191 research papers published between 1981 and July 2023 was conducted to examine the health benefits of Musa paradisiaca (banana). The literature search was carried out using databases like PubMed, Scopus, and Google Scholar, focusing on studies exploring bananas’ nutritional, medicinal, and therapeutic properties. Inclusion criteria encompassed original research, clinical trials, in vitro and in vivo studies, and reviews on bananas' bioactive compounds and health effects, explicitly addressing conditions such as cancer, diabetes, inflammation, and hypertension. Exclusion criteria included non-peer-reviewed articles, conference abstracts, papers unrelated to M. paradisiaca, and studies unavailable in English or lacking data on banana-specific effects. Data extraction focused on study types, health outcomes, and phytochemicals, followed by a synthesis of trends in efficacy and a qualitative assessment of study methodologies. Limitations related to study designs and phytochemical standardization were also addressed, suggesting future research directions.
Nutrient Profile of Banana
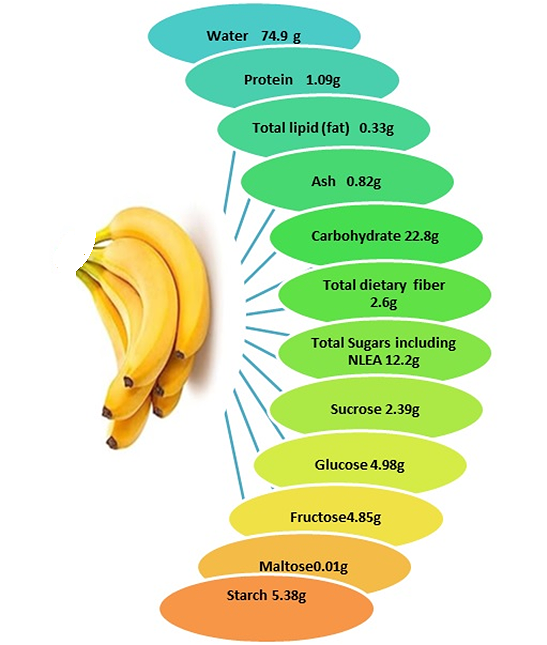
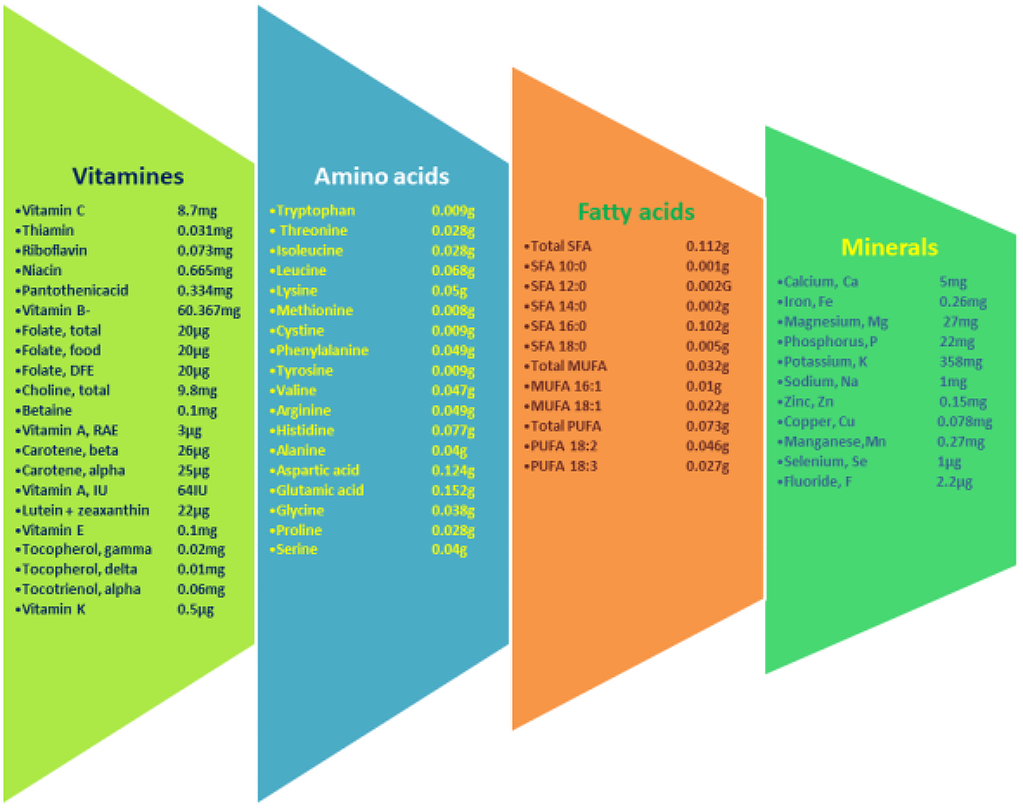
In addition to routine contents like carbohydrates, dietary fiber, minerals, and vitamins, bananas also contain several health-promoting bioactive phytochemicals, including antioxidants, carotenoids, and phenolic compounds (5). Phytochemical analysis of M. paradisiaca showed that along with the usual carbohydrates and reducing sugars, different parts of the banana also contain alkaloids, glycosides, phenols, tannins, steroids, terpenoids, saponins, flavonoids, and several secondary metabolites (8). The ripe banana peel of M. paradisiaca contains various minerals like Na, Mn, Ca, Zn, Cu, N, K, and Fe with concentrations in ppm of 84.53, 18.82, 2.41, 1.01, 1.89, 1.15, 3.96, and 27.83 respectively (9). Ripe peel also contains proteins, carbohydrates, fat, fiber, and ash at concentrations (in percentage) of 7.18, 42.95, 6.22, 14.31, and 22.30, respectively (9). An average-sized banana fruit contains approximately 6 g of fiber and 450-467 mg of potassium, and its pulp is high in cellulose, sugars, fiber, and starch and is a good source of vitamins like vitamins A and C (10). The pulp of the banana fruit, Musa paradisiaca var. sapientum, is also reported to contain several bioactive nitrogen-containing compounds like serotonin, tryptophan, norepinephrine, indole compounds, stearyl acyl glycosides, sitosterol, acyl stearyl glycosides, myoinositol β-D-glucoside, cyclomusalenol, cyclomusalenone, 24-methylene cycloartenol, stigmast-7-methylenecycloartanol, stigmast-7-en-3-ol, lanosterol, and β-amyrin (11, 12). In M. cavendish, antioxidant gallocatechin was reported and was more abundant in the banana peel than in the pulp (13). Flavanoid intake protects against coronary heart diseases (14). Furthermore, the flower has yielded hemi terpenoid glucoside (1, 1-dimethylallyl alcohol β-glucoside), syringin, (6S, 9R)-roseoside, and benzyl alcohol glucoside (15). In addition to the pulp, the banana peel is also a rich source of phytochemicals. Studies on the nutrient content of six varieties of banana peel reported 40-50% dietary fiber with a high protein content of 8-11%, along with essential amino acids like phenylalanine, leucine, valine, and threonine. Among the minerals, potassium was present in significant amounts (16).
Banana peel is valued for its bioactive components, including phenolic compounds such as flavonols, hydroxycinnamic acids, flavan-3-ols, and catecholamines, and is also a potential source of pectins and dietary fibers (17, 18). The pulp and the banana peel are also a rich source of bioactive compounds like carotenoids, flavonoids, phenolics, vitamins like C and E, and amines (19, 2). As the fruit ripens, the starch present in the banana is converted into glucose, sucrose, and fructose (20). Unripe bananas have a lot of digestible starch, while ripe bananas have more resistant starch (21). Chitinase, a transient Vegetative Storage Protein, is common in unripe bananas (22). Also, as the banana ripens, a large increase in fructose, glucose, and total sugar is noted (23). Comparison of phytochemicals and mineral compositions of ripened and unripe banana flours revealed that the total amount of soluble sugars present in unripened bananas ranged between 1.70 to 2.15 mg/100 g, while those in ripened bananas ranged from 37.5 to 43.8 mg/100 g (24). Its inflorescence is edible and is used as food and in medicine (24, 25). Banana is a good and easy source of macronutrients such as carbohydrates, protein, unsaturated fatty acids, vitamins, and various minerals. Soluble solids (17.9%), vitamin A (12.4 mg/100 g), and vitamin C (12.7 mg/100 g) were also observed in bananas by researchers. The nutrient and phytochemical composition of banana fruit (as per 100 g) is given in Figures 1 and 2 (26). Bananas contain various bioactive compounds rich in antioxidants, including carotenoids, flavonoids, phenolics, amines, and vitamins C and E, which offer numerous health advantages (2). The peel is rich in flavonoids, flavan-3-ols, hydroxycinnamic acids, and catecholamines (18). Additionally, Musa cavendishii is notable for its antioxidant dopamine, present in both the peel (80-560 mg/100 g) and pulp (2.5 to 10 mg) (27).
The significant presence of phenolics, biogenic amines, flavonoids, carotenoids, sterols, and various antimicrobial agents makes bananas suitable for enhancing health (29). They contain phenolic acids like ferulic, gallic, vanillic, salicylic, sinapic, p-hydroxybenzoic, syringic, gentisic, and p-coumaric acids (30). Ferulic acid, in particular, has antimicrobial, anticarcinogenic, anti-inflammatory, and vasodilatory properties (31).
An assessment using LC-MS-ESI revealed the presence of flavonoids such as epicatechin and 3-O-rhamnosyl-glucoside. Potassium was the most abundant mineral at 14, 746.73 mg/Kg, while zinc concentration was 3.55 mg/Kg in unripe banana flour (32). Key bioactive compounds with antioxidant properties in banana fruit include gallic acid, tannins, catechin, epicatechin, and anthocyanins. Plantain pulp is rich in hydroxycinnamic derivatives like ferulic acid-hexoside, while banana peel contains high levels of rutin (5). The flavonoids identified from bananas, such as quercetin, myricetin, kaempferol, and cyanidine, are free radical scavengers (33). Carotenoids in bananas include lutein, lycopene, and zeaxanthin (34), with orange and yellow-fleshed varieties having higher trans-carotene content (35). Biogenic amines like dopamine, norepinephrine, and serotonin are prevalent in banana peel and pulp, with serotonin levels ranging from 8 to 50 µg/g and varying dopamine levels in different banana pulps (36). Dopamine in bananas improves LDL resistance to oxidation and reduces plasma oxidative stress, leading to decreased oxidative modification of low-density lipoprotein (37).
Health Benefits of Banana
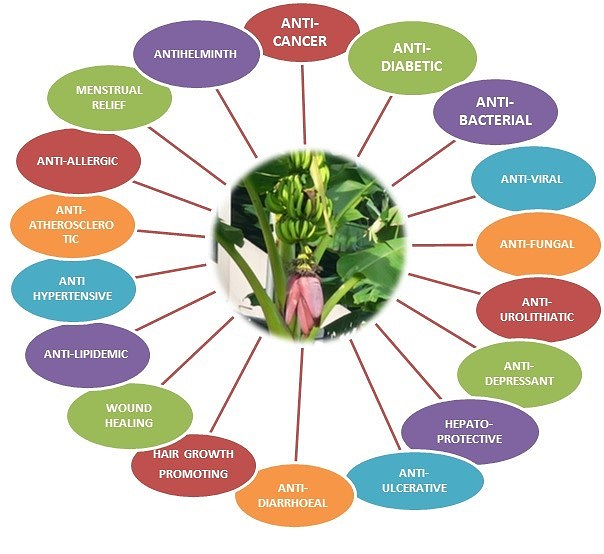
Researchers have uncovered numerous health benefits of bananas (Figure 3). Sitosterol in bananas may alleviate benign prostatic hyperplasia symptoms and lower cholesterol levels (38). Banana ethanol fiber extract reduces cholesterol in obese male mice (39), while gallocatechin gallate aids cholesterol reduction and offers hepatoprotective effects (40, 41). Serum α-carotene levels inversely correlate with CVD and cancer mortality risk (42). Plant sterols like campesterol and stigmasterol in bananas lower cholesterol absorption by replacing LDL in the intestine (43). Additionally, cinnamic acid serves as an aspartame precursor (44), and cycloartenol supports plant steroid biosynthesis (45). Catechins enhance plasma antioxidants, reduce lipid peroxides, and improve LDL oxidation resistance (46).
The serotonin content in bananas has been linked to feelings of well-being and happiness. Although bananas contain serotonin, they do not cross the blood-brain barrier (47). Banana lectin (BanLec-1) isolated from M. paradisiaca showed binding specificity for oligomannosidic. It has been shown to stimulate T-cell proliferation (48). Catecholamines are neurotransmitters and are precursors of benzophenanthridine alkaloids (49). The tannic acid from bananas has been utilized in burn treatment (50).
Anticancer Properties
An anticancer polyphenol, protocatechuic aldehyde (PCA), was identified in green Cavendish bananas (51). PCA increases activating transcription factor 3 (ATF3) expression and shows anticancer properties in leukemia, colorectal, and breast cancer cells (52). A higher incidence of esophageal cancer has been reported in males consuming fewer or no bananas (53). Daily consumption of 2 g of banana in Swiss albino mice with Ehrlich ascites carcinoma cells resulted in growth suppression, allowing 30% of the animals to survive longer (54). A diet with 10% green banana flour in colon cancer mice reduced the number of aberrant crypt foci (a colorectal cancer biomarker) reported in colorectal mucosa (55). A high intake of certain vegetables and fruits reduces the risk of breast cancer (56). Research indicated that 2-pentanone, a compound found in bananas, can inhibit the production of prostaglandin and COX-2 protein expression in colon cancer cells (57). The banana peel methanol fraction inhibited testosterone-induced cell growth in the androgen-responsive LNCaP prostate carcinoma cell line, which may aid in controlling benign prostate hyperplasia (58). Anthocyanin from the bract of M. acuminata regulated the human breast cancer cell line (MCF-7) by inducing apoptosis (59). Protocatechualdehyde showed antiproliferative activity on human colorectal carcinoma cells in a dose-dependent manner by reducing the expression of enzyme histone deacetylase 2 mediated by cyclin- D1suppression (60). Ethanol extract from banana flower demonstrated anticancer properties on HeLa cells, activating apoptosis and increasing caspase-9 enzyme activity by blocking the cell cycle at the G0/G1 phase (61). The hexane fraction of banana pulp and peel has shown in vitro anticancer activity against HCT-116 cells (62) and high toxicity against MCF-7 and HCT-116 tumor cell lines (63). Extracts from banana pseudostems and rhizomes have demonstrated excellent cytotoxicity against HepG2 liver cancer cells (64). Ferulic acid from banana peels activated DNA fragmentation in HeLa cervical cancer cells (65). Three phytochemicals from pseudo stem of a banana can arrest mitotic cell division via proteins, cyclin-dependent kinase 2 and tubulin (66). Banana flower extract (aqueous) exhibited anticancer activity against benign prostatic hyperplasia by inducing cell cycle arrest at the G1 phase (67). Ethyl acetate sub-fraction collected from soft piths of banana (M. paradisiaca) is found to exhibit excellent antiproliferative and cytotoxic activity on human tongue squamous cell carcinoma (HSC-4) (68). Banana peel methanol extract collected from the Nendran variety showed excellent antitumor activity against the MCF-7 breast cancer cell line. Treated cell lines showed condensed nuclei and apoptosis, which showed their efficiency against breast cancer (69). Studies showed that M. cavendish green peel extract has antiproliferative properties on HepG2, A-375, MCF-7, and Caco-2 cancer cells. The extract showed an antiproliferative effect in all cell lines at a concentration of 100 µg/mL. Also, HepG2 induced changes in cell morphology and necrosis related to cell death. The authors suggested that MHE can be considered for developing new drugs with anticancer properties (70). Hyperpigmentation of the cell is considered due to over expression of pigment melanin. Sucrier banana peel (Methanolic) extract inhibits melanogenesis via the p38 signaling pathway in B16F10 mouse melanoma cells (71). Mannose-specific lectin from bananas exhibited antiproliferative activity and induced cell cycle arrest in different cancer cell lines (72). M. acuminata flower methanol extract showed cytotoxic and antiproliferative properties on HeLa cells (73). The ethyl acetate fraction from M. paradisiaca leaves displayed potential anticancer activity against HeLa and A375 cervical cancer cell lines (74).
Antidiabetic Properties
Bananas have a low glycemic index, effectively reducing immediate blood glucose levels. Studies show that methanolic extract from green M. paradisiaca fruits exhibits significant hypoglycemic properties in both healthy and diabetic-induced mice (75). A pectin-type polysaccharide from banana peel demonstrated antidiabetic effects by activating insulin production (76). Alcoholic extracts from M. paradisiaca flowers have also shown excellent antihyperglycemic properties (77). Banana flower has several bioactive compounds and shows antidiabetic properties (78). Consumption of M. paradisiaca inflorescence extracts normalized blood glucose and lipid peroxidation levels in diabetic rats (79).
Malaysian banana flower extracts have been identified as a potent source of antioxidants and postprandial regulators to combat hypoglycemia (80). Insulin tolerance tests indicated that extracts from M. cavendish and M. acuminata had the highest hypoglycemic activity compared to M. sapientum and M. paradisica (81). Methanol and hydroalcoholic extracts from the pseudostem effectively inhibited α-amylase and α-glucosidase enzymes, demonstrating antidiabetic activity (82). Ethanol extracts from M. balbisiana flowers exhibited significant antidiabetic, antihyperlipidemic, and antioxidant effects in diabetic rats. The serum insulin level increases, and glucose absorption from the intestine is reduced considerably (83). M. paradisiaca inflorescence is also an excellent source of soluble dietary fiber, which can enhance glucose and cholesterol adsorption and increase glucose uptake in myoblasts (84). Streptozotocin-induced diabetic rats, when given with aqueous extracts of M. paradisiaca inflorescence showed that the inflorescence could be used as an alternative therapy for treating type 2 diabetes mellitus with promising hypoglycemic effect (85). M. Type 2 diabetic rats (induced by STZ) showed reduced food intake, also the fasting insulin and GLP-1 levels are increased, after the ingestion of banana peel dietary fibres to such rats. Thus, banana peel soluble dietary fiber can be used to treat type 2 diabetes mellitus (86).
Chloroform extracts from banana flowers have shown antidiabetic properties (1). Consumption of resistant starch from green bananas can reduce fasting glucose and body weight, making it a good option for prediabetics (87). An extensive review recommends standardizing dosage and considering the banana variety and ripening level for different age groups (88).
Antibacterial Properties
Different parts of bananas also show potent antibacterial properties. The threshold of inhibitory concentration (ic 50) against Staphylococcus and Pseudomonas with M. paradisiaca bark/peel aqueous extract were 143.5 and 183.1 µg/mL, respectively. The banana peel extract was more active than the leaf extract against both bacteria. Also, the extract was more active against Staphylococcus bacteria than the Pseudomonas species (89). In the laboratory study, ethyl alcohol extracts of banana flowers of M. sapientum have been shown to inhibit the growth of several pathogenic bacteria like B. subtilis, B. cereus, and E. coli. Of the different bioactive compounds isolated (which include β-sitosterol, 12-hydroxy stearic acid, palmitic acid, and d-malic acid), β-sitosterol and malic acid were the active compounds responsible for antibacterial activity. The study may aid in wound healing and infection prevention (90). The antimicrobial property of M. paradisiaca and Cocos nucifera crude extracts on bacteria was studied using the agar disc diffusion method. Both plant extracts hindered the growth of test organisms. The methanol extract of M. paradisiaca fruit peels exhibited pronounced antibacterial activity against bacteria such as Staphylococcus aureus, Bacillus subtilis, E. coli, and Pseudomonas aeruginosa (91). The flower extracts (ethyl alcohol and ethyl alcohol: water extracts) of M. paradisiaca showed antibacterial and antifungal activity against E. coli, B. subtilis, B. cereus, Klebsiella pneumoniae, P. aeruginosa, P. mirabilis, S. aureus, S. pneumoniae, S. typhimurium, C. albidus and C. albicans with minimum inhibitory concentrations that ranged from 5.62-25.81 and 7.60-31.50 µg/mL, respectively (77). Studies also showed that the ethanol extract of unripe banana peel inhibited the microorganism like Pseudomonas aeruginosa, S. aureus, Proteus mirabilis, Bacillus subtilis, Aspergillus niger at a concentration of 100 mg/mL with the inhibition zones of 8.2, 7.8, 8.3, 5.4, 4.6 respectively (9). Ethyl acetate extract of M. paradisiaca demonstrated effective antibacterial properties against multidrug-resistant strains of nosocomial pathogens P. aeruginosa, E. coli and Citrobacter sp (92).
The antibacterial activity of the banana extract was highest against M. catarrhalis and S. aureus (inhibition zone 30 mm), followed by S. pyogenes, E. aerogenes, and K. pneumoniae (93). Alcoholic extract of banana peel has antimicrobial properties against pathogens causing aggressive periodontitis P. gingivalis and A. actinomycetemcomitans (94). Antibacterial potentials of leaf ethanol and aqueous extract of M. paradisiaca against bacteria of clinical importance like S. aureus, B. subtilis, P. aeruginosa, V. cholerae, and S. dysenteriae showed that aqueous fraction has a minimum inhibitory concentration that ranged from 3.125 to 25 mg/mL. The aqueous fraction outperformed the ethanol extract regarding antibacterial activity (95). Methanol extract is reported to have more inhibition than ethanol extract in controlling E. coli, P. aeruginosa, S. aureus, and Salmonella typhi (96). Also, the methanol fraction of M. paradisiaca exhibited greater wound-healing activity in albino rats (97). Streptococcus mutans is reported as the most susceptible bacteria with banana peel ethanol extract, and the inhibition zone ranged from 8 mm at 3.125 mg/mL to 25 mm at 100 mg/mL. It was followed by P. aeruginosa and E. coli. Also, the sensibility of the multidrug-resistant bacteria to the ethanolic peel extract is of great significance in future work (98). The peel extract in methanol had strong antibacterial action on S. aureus and E. coli, and the activity was suggested to be associated with the total flavonoid and the phenolic compounds. The leaf stalk extract of M. acuminatacollected in acetone showed antimicrobial properties against the disease-causing bacteria, P. aeruginosa and E. coli, and therefore, it can be recommended for infections caused by these microorganisms (99). M. acuminata and M. paradisiaca ethanolic leaf extracts act as antibacterial agents for methicillin-resistant strains of S. aureus (100).
Antiviral Properties
Banana lectin was discovered in 1990 in M. paradisiaca (48); however, no significant further work was reported for a long time. The crystal structure of methyl αD-monoxide with banana lectin (from M. paradisiaca) has exposed two primary binding sites for banana lectin (101). Lectins are homodimeric natural proteins capable of binding reversibly with carbohydrates on the cell surface. So, lectin can bind with the surface of pathogens and can damage their structure, preventing the host infection. Studies showed that lectin can bind with mannose and mannose-containing oligosaccharides and is an effective T-cell mitogen. BanLec or banana lectin is extracted from the fruit of M. acuminata. The lectin has high affinity and can bind to mannose structures, especially those located on the glycosylated envelope of viruses like HIV-1, by binding to glycosylated envelope protein gp120 and preventing its further entry into the cell and showing potent anti-HIV properties (103). Substituting a single amino acid, histidine, with threonine in banana lectin (H84T) can reduce mitogenicity and maintain its antiviral properties. It can bind to mannose N-glycans and showed antiviral properties on several high-mannose expressing viruses, like HIV-1, HIV-2, hepatitis C, and influenza A and B. BanLec H84T can inhibit both virus-like particles as well as entry and replication of the Ebola virus mini-genome in cells (103, 104). Ethanol/acetone extract of banana leaf isolated from many cultivars showed excellent anti-Chikungunya virus properties (EC50 around 10 µg/mL) (105). 25 µg/mL BanLec has exhibited an increased rate of inhibition on Bovine Viral Diarrhea 1 (BVDV-1) (of 99.98%) and on Bovine α Herpes Virus (BoHV-1) (of 99.68%) without showing any cell viability (106).
Antifungal Properties
Peel and stalk extract of banana (M. paradisiaca ) exhibited antifungal actions, and the activities were studied using the percentage inhibition test. A stalk extract of 1.0 mg/mL showed 100% inhibition of growth against fungi like Aspergillus oryzae, Aspergillus niger, and Rhizopus stolonifer. Peel extract inhibition was 100% on A. niger, 76.67% on A. oryzae, and 56.67% on R. stolonifer at the concentration of 1 mg/mL (107). Banana leaves also exhibited significant antifungal activity. In another M. acuminata leaf extract experiment, the largest inhibition zone diameters were shown against Candida albicans (108). Similar antifungal activities of banana leaves were observed against Penicillium oxalicum (with inhibition of 40%) and Alternaria alternate (109). Kadali banana dried peel powder and ash extract showed antifungal properties against Aspergillus niger (110).
The antimicrobial property of extracts collected from M. paradisiaca and Cocos nucifera on fungi Candid sp (Candida tropicalis and Candida albicans) and Aspergillus niger was studied using the agar disc diffusion method. Both plant extracts hindered the growth of test organisms. Also, M. paradisiaca extract suppressed Candida albicans more effectively than Cocos nucifera crude extract with a wider zone of inhibition (91). When compared to nystatin, the extract of M. acuminata leaves in methanol demonstrated superior antifungal actions on Staphylococcus epidermidis at a dose of 60 mg/mL and on Trichophyton mentagrophytes at a concentration of 40 mg/mL. The capacity points out that the methanol extract collected from the leaves of M. acuminata served as a powerful antifungal agent (111).
Antiurolithiasis Properties
The impact of M. paradisiaca stem juice on the formation of crystalline substances (stones) and the anti-lithic properties was studied on urolithiasis rats. Oxalate synthesizing enzymes, Glycollic acid oxidase, and Lactate dehydrogenase (LDH) were significantly active in such rats. The extract treatment lowered the enzyme activity, such as glycollic acid oxidase and urinary alkaline phosphatase, reducing oxalate synthesis. There was a reduction in other enzymes like LDH, inorganic pyrophosphatase, and -glucuronidase. There was a lowering in the level of oxalate in the urine. Thus, the M. paradisiaca extract lowers the crystalline component formation (112). In a study, M. paradisiaca and M. sapientum pseudostem core were used to treat urinary stones for two weeks. Results indicated the effectiveness of plant material in treating urolithiasis, mostly that of calcium oxalate stones. Out of the 71 patients treated for 4 weeks, 20 had a complete cure, 43 patients had passed the different-sized stones and 4 patients had reduced the number of calculi (113).
In a review, 103 plants have been reported to show potent litholytic properties, of which M. bulbisiana roots and M. paradisiaca ripe juice (kernel or the pulp) have been listed to have litholytic properties (114). The role of ethanol extract of the banana corm Musa (cultivar monthan) on urolithiasis rats (induced by ethylene glycol and ammonium chloride) was studied concerning kidney stone crystal formation and inhibition. The stone was analyzed spectrophotometrically, and the result showed its efficiency in diuresis and promoting crystallization inhibition (115). In vitro studies in calculi-induced albino rats using Musa formulations (AAB) for treating renal calculi are well studied. The administration of liquid Musa formulations reduced the size of kidney stones significantly, as it contains organic constituents such as β-sitosterol, saponins, quercetin, tannins, and several inorganic contents such as magnesium, potassium, and nitrate. Also, it is suggested that Musa AAB liquid formulations may help overcome the disadvantage of several surgical procedures that may follow due to stone recurrence (116). The efficacy of Musa (banana) pseudostem ethanol extract used in regulating ethylene glycol-induced urolithiasis in rats may be by inhibiting various biochemical pathways associated with renal calcium oxalate metabolism (formation), as well as its antioxidant property and the ability to inhibit biochemical markers of renal impairment (117). Both aqueous and methanol extracts from the fruit peel of M. sapientum (as well as other plants like Malus pumila, and Punica granatum) showed anti-urolithiatic activity in experiments. The result is significant and can be used for treating lithiasis (118). The anti-urolithiasis property of extract in methanol collected from pseudostem of M. acuminate was studied using a spectrophotometer in vitro nucleation and aggregation experiment and by subsequent microscopic examination, showing its efficacy as an alternative medication for kidney stones (119).
Antidepressant Properties
Depression is a mental disorder that affects the mood and thoughts of more than 10% of the population (120) and shows a severe complication of suicidal behavior. Banana peel contains several phytochemicals that can reduce depression. In an experiment to study the antidepressant activity of bananas, fruit paste was administered at concentrations of 5%, 10%, and 20% once daily to Swiss mice for 15 days, and subsequently, the antidepressant property was measured by the methods of Tail Suspension Test and Forced Swim Test. The experiment indicated that the fruit paste could remarkably lower the time for immobility in both tests. M. paradisiaca paste inhibited the malondialdehyde and the Monoamine oxidase enzyme (121). Studies carried out in Swiss albino mice showed that intake of extracts orally from green or yellow banana peel (at 200 and 400 mg per kg, respectively) can be recommended for treating depression (122). In a study with pulp and the banana peel extract (at a dose of 600 mg per kg and 400 mg per kg, respectively) of M. sapientum on male albino mice for 14 days through oral administration, and further assessment of its performance through forced swimming test, light and dark activity and maze activity tests, it showed antianxiety, antidepressant, and memory enhancement properties, possibly through phyto-antioxidants (123). Though banana leaf extracts (M. sapientum) showed antidepressant activity that may be mediated by α1-adrenergic and D2 dopaminergic receptors, no significant anxiolytic effects were reported (124). The essential amino acid tryptophan for synthesizing serotonin is very supportive in relaxing people, improving their mood, and making them happy. Thus it can be used to treat depression (1, 125). Bananas can be recommended for managing depression based on the 2: 1 ratio of ώ 6 to ώ 3 fatty acids (126).
Hepatoprotective Properties
The role of M. paradisiaca in supplementing hepatotoxic rats and the biochemical and histological effects were studied in detail, including transaminase enzymes. Histological examination of liver tissue sections revealed necrosis in hepatotoxic rats; however, varying degrees of regeneration were noted in rats given M. paradisiaca supplements (127). The alcoholic extract of M. paradisiaca stem at dosages of 500 mg/kg, per organism. To a lesser extent, the aqueous extract at a concentration of 500 mg/kg, per organism has a substantial recovery on the liver of carbon tetra chloride and paracetamol caused hepatotoxic damage in rats. The hepatic damage was reversed in these test organisms. It reduced increased levels of various enzymes like serum glutamic oxaloacetic transaminase and serum glutamic pyruvic transaminase (128). Banana contains ferulic acid (30). It acts as a strong hepatoprotective phytochemical and shows no side effects (129, 31). The root extract (aqueous) of M. paradisiaca contains bioactive compounds that can protect the liver and kidney from arsenic-caused damage in albino rats (130).
Methanolic extract of M. sapientum treated for 7 days in albino rats exhibited a considerable lowering of ulcer index, and its ulcer protective effect is similar to omeprazole and ranitidine even though more work is essential to determine the true nature of the therapeutic of the plant (131).
Antiulcerative Properties
Leucocyanidin, an active antiulcer compound from the banana, M. paradisiaca protects aspirin-induced ulcers. This natural flavonoid protects against aspirin-induced damage to the gastric mucosa (132). The antiulcer activity of banana peel water extract on male albino rats is due to the flavonoids (leucocyanidin), tannins, and saponins. Plantain peel extract of 200 mg/Kg body weight showed the lowest mean ulcer index and concluded that the peel extract could be used in ethnomedicine (133). Several banana varieties from northeast Thailand are reported to have a gastrointestinal protective effect against peptic ulcers, with Palo and Hom varieties showing a pronounced effect (134). The ethanolic extract of plantain elevated eicosanoid accumulation in the stomach and colonic mucosa in a concentration-dependent manner, and the banana might have acted possibly by making arachidonate available (135). The ulcer-curing actions of unripe bananas, M. sapientum, were investigated by many (136, 137) and reported a flavonoid leucocyanidin that showed antiulcerogenic properties. Administration of M. sapientum aqueous extract in the prescribed dosage helps to alleviate ulcers (137). It is almost a fact that banana juice can help heal wounds and burns and relieve the pain of ulcers and other gastrointestinal disorders, including acid secretion. The flavonoid leucocyandine increases the mucosal membrane thickness of the stomach (1). A pylorus ligation technique revealed that the methanol extract from M. paradisiaca shows cytoprotective action on peptic ulcers caused by indomethacin (138).
Chloroform and ethanol extracts from the leaves of M. sapientum lowered the ulcer and ulcer index in ulcer-induced rats (139). The banana tepal and peel extracts worked against ulcers by strengthening the stomach mucosa and reducing gastric secretion acidity in ulcer-induced albino mice (140). When treated with M. sapientum methanol extract, ulcer-induced rats showed regeneration of epithelial tissues of the stomach from the third day. By the twelfth day, the regeneration became almost complete. The healing of the ulcer is attributed to the basic fibroblast growth factors (141). The banana flower has several bioactive compounds and shows antiulcer properties (78).
Antidiarrhoeal Properties
Green bananas were used by the public to treat a number of digestive issues, like childhood diarrhoea. Providing a solution with 50 g/L of plantain flour and 3.5 g/L of sodium chloride to children suffering from acute diarrhoeal diseases indicated good rehydration and antidiarrhoeal activity. However, some children have noted decreases in blood sodium and potassium levels (142). Also, in the same year, a comparative study of standard medical care with banana flakes was found to be more effective in treating diarrhoea in enterally fed patients. Banana's antidiarrheal activity might be due to its pectin and other soluble fibers that serve as active antidiarrhoeal substances (143). Children having persistent diarrhoea for 14 days showed that including green bananas in the diet can increase the permeability of the intestine. The increased permeability was proved with lactulose-mannitol drink given to children and by subsequent recovery (after 5 hours) of increased urinary mannitol and reduced lactulose content. Recovery of stool weight was reduced by 50%, and there was a drastic acceleration in the clinical recovery. Thus, the green banana and the pectin's antidiarrhoeal effects are mediated by the increased rate of the permeability of the small intestine, along with the known colonotrophic properties (144). Green banana's antidiarrheal action is believed to be due to its high starch content resistant to amylase, which gets fermented to yield several short-chain fatty acids (SCF) in the colon that stimulate the absorption of salt and water from the colon. The SCF thus produced is an adaptive process as these fatty acids get absorbed in the epithelial cells of the colon, which subsequently helps in the absorption and conservation of fluid and electrolytes. Thus, adding resistant starch that produces SCF in the colon to oral rehydration solution (ORS) might improve the efficiency of the ORS given to children under 5 years suffering from acute diarrhea (145).
The antidiarrheal property of M. paradisiaca sap is considered to be due to the presence of phytochemicals like alkaloids, flavonoids, phenolics, and saponins of the sap that is responsible for the increased absorption of fluid and electrolytes via de novo synthesis of Na-K-ATPase pump (146). Unripe banana (M. paradisiaca) ethanol extract has antidiarrheal activity (147). Studies showed castor oil-induced diarrhea of rats’ stool consistency can be improved with unripe banana peel extract (BPE) containing pectin and other active substances (148).
Wound-Healing Activity
Banana extracts have wonderful wound-healing capacity with its antioxidant effect as well as due to the various wound healing biochemical parameters (like enzyme activity of superoxide dismutase, reduced glutathione, and lipid peroxidation as well as that related to connective tissue development (like hexuronic acid, and quantity of hexosamine and hydroxyproline), it showed that plantain banana extracts have significant healing capacity (149). It was reported that the M. paradisiaca stem exhibits a hemostatic effect. A 10% juice of plantain stem extract was formulated to prepare an ointment and tried for the healing properties of rats. It confirmed a prominent (p< 0.05) enhancement in the healing process. When the wound area closure (in %) was compared between plantain stem juice and silver sulphadoxine, the plantain-based ointment showed 98.9±0.7% closure area, and for the silver sulphadoxine was 100±0.00%. The results conclude the efficiency of plantain-based ointment in healing wounds (150). The dermal application of banana peel extract augments healing through vascular endothelial growth factor (VEGF), which is released from degranulating platelets in large amounts, and new capillary development occurs in the area. An increased collagen concentration occurred in the wound area due to the synthesis and deposition of new collagen (151), aiding in wound healing. There is an increase in vascular and fibroblast proliferation (152). The flavonoids, glycosides, and phenols of banana peel extracts have antibacterial and anti-inflammatory properties (140).
The histopathological and clinical examinations were used to measure wound healing by assessing the wound contraction rate and epithelialization of the wound tissue in rabbits treated with banana peel extract (153). M. paradisiaca peel extract exhibited wound-healing properties in male Wistar rats. The peel's methanol and hexane extract showed better results. This ability is due to the antioxidant property of phytochemicals like alkaloids, phenols, tannins, and saponins in banana peel (154). Methanolic extract from M. paradisiaca Linn. stem exhibited greater wound healing ability in albino rats (97). Unripe peel extract has pharmacological properties and decreased WBC counts (155). Kepok banana (M. paradisiaca L) peel extract has saponins (that show hemostatic properties), tannins (that show vasoconstrictive properties), and flavonoids (also affect blood capillaries) that can stop bleeding, and these bioactive phytochemicals of the peel can be used for healing wounds (156).
Antihyperlipidemic Activity
Banana also exhibits antilipidemic activity. The excretion of bile acid and neutral sterols is found to be higher in fiber-fed (from unripe banana) rats. The neutral detergent fiber from M. paradisiaca can lower cholesterol absorption in rabbits (157). Administration of flavonoids orally isolated from unripe banana (M. paradisiaca) fruits at a concentration of 1 mg per 100 grams body weight/day has a prominent hypolipidemic action in male rats. The quantity of different fats (like cholesterol, phospholipid, and triacylglycerol) significantly decreases in the brain, kidney, and liver (158). The structural similarity of cholesterol with banana phytosterol is also well-accepted. Reports showed that phytosterol replaces the cholesterol in the gut during absorption, thereby reducing the blood cholesterol level (159). Soluble and insoluble components of dietary fiber of banana pulp showed cholesterol lowering property in male rats (160)
The effects of flavonoid, saponin, and tannin present in banana peel of Kepok variety extract in obese male mice, Mus musculus L. (at a concentration of 200 mg per kg body weight) in lowering the level of total cholesterol was established (39). In rats supplemented with M. paradisiaca (at a dose of 10, 20, and 30%) for 21 days, total serum cholesterol was lowered, thus showing an antihyperlipidemic effect of banana (162). Consuming fresh and dried banana peels may modify the risk of acute liver failure patients. A significant increase in HDL-c was observed in all acute liver failure albino rats administrated with dried banana peels (161). Studies show that ripe banana extract is high in aliphatic alcohols, fatty acids (both ω-3 and ω-6), and α-tocopherol, supporting a healthy life (163). All these works emphasize that bananas can easily step into biologically active functional foods, and its inflorescence can be economically consumed as food and medicine (24). In diabetic rats, Kepok banana peel extract shows an antihyperlipidemic effect (164).
Antihypertensive Activity
Fruit preparations reduced heart rate and mean arterial blood pressure in normal albino rats and in rats previously treated with deoxycorticosterone acetate (DOCA), providing evidence for blood pressure-lowering action. Thus, bananas can lower DOCA-induced increased arterial pressure and hypertension in rats (165). The studies with plantain extract on rats' isolated aorta and portal veins showed that the extract has a direct effect by relaxing the noradrenaline and KCl-contracted aortic rings (166). Banana is reported to decrease both systolic and diastolic blood pressure changes induced by cold stress. The activity of plasma angiotensin-converting enzymes (ACE) is reduced considerably. The poovan variety of banana has a high inhibitory effect on ACE (167). Results of a study with 20 randomly selected women of age group 19-22, daily intake of one banana (M. acuminata) for 7 days showed a lowering of blood pressure in the cold stress test (168). The most important phytochemicals in bananas are inhibitors of angiotensin-converting enzyme (ACE). Ripened banana varieties like nendran, poovan, robusta rasthali, safed velchin, and bontha effectively inhibited the enzyme ACE, with nendran as a strong inhibitor. However, unripe bananas showed only a weak response (169). Notably, M. paradisiaca fruit consumption reduced the increase in systolic, diastolic, and arterial blood pressure induced by cold stress. There is a significant drop in plasma ACE activity, implying that the observed blood pressure-lowering effects were due to suppression of this enzyme activity (169).
The potassium in bananas effectively regulates blood pressure by reducing the effect of sodium. Therefore, consuming bananas can help to reduce the feeling of thirst along with muscle weakness caused by potassium deficiency. Incorporating banana juice in the food may reduce blood pressure and protect elderly persons from degenerative disease (170). M. paradisiaca has an antihypertensive effect in albino rats and was found to lower arterial blood pressure and prevent the onset of increased blood pressure caused by DOCA injection in rats (171, 172). Banana’s low salt content helps regulate blood pressure and prevent strokes (125). A study among hypertensive individuals reported that the phytochemicals in bananas efficiently reduced both systolic and diastolic blood pressures. However, more clinical studies in humans are recommended to provide efficacy evidence (173). Many studies also suggested that the intake of bananas significantly lowers blood pressure in elderly persons (174-176).
Antiatherosclerotic Activity
M. paradisiaca inhibits in vitro cholesterol crystallization and nucleation. The plaque contains cholesterol monohydrate, and M. paradisiaca showed anti-atherosclerotic effects in diet-induced atherosclerosis (177). Treatment with M. paradisiaca peel extract (at a concentration of 100 mg/Kg) to atherosclerosis-induced Wistar albino male rats ameliorates several biochemical changes caused by the atherogenic diet, indicating the role of peel extract against induced atherosclerosis and thyroid dysfunction (178). An extensive study conducted in Indonesia proposed that Ambon banana peel extract inhibits the atherosclerosis process and can be considered a therapeutic material for the prevention of atherosclerosis (179).
Antiallergic Activity
Methanol extract of M. paradisiaca stem has anti-inflammatory properties on chemically induced acute edema, like dextran-caused paw edema (180). The powder of the pseudostem of M. paradisiaca displayed an antiallergic potential in rats when treated daily by oral administration (181). The bronchospasm induced in guinea pigs with histamine or acetylcholine is reduced considerably with treatment using hydroalcoholic extract of M. paradisiaca flower (p< 0.001) (182). The study on mice and human macrophages with banana inflorescence extract showed that the inflorescence extract exhibits antiallergic properties and reduced expression of CD86 and HLA-DR receptors on human M1 macrophages, inhibiting eosinophil migration (183).
Anthelmintic Activity
Compared to the usual medication piperazine citrate, the anti-helminthic activity of the corm of banana ethanol extract is dose-dependent and more effective. The anthelmintic qualities of M. paradisiaca cv. Puttabale’s ethanol extracts on Pheretima posthuma are dose-dependent, with a concentration of 100 mg/mL showing a paralyzing time of 42.33 minutes and a death time of 54 minutes, compared to 39.67 minutes for paralysis and 59 minutes for death with piperazine citrate (184). The anthelmintic property of banana peel methanol extract (40 mg/mL) showed the quickest duration for the paralysis and subsequent death of worms compared to the standard drug, albendazole, suggesting the use of banana peels in helminth infestations (185). Sheep fed with dried ground banana leaves may experience a decrease in the survival of Trichostrongylus colubriformis eggs, which is potentially valuable for integrated parasite control strategies (186). A study on the anthelmintic activity of Kepok banana peel extract revealed the highest mortality rate against Ascaridia galli after 10 hours of treatment with a 75% concentration of ethanol extract (187). Aqueous and ethanolic extracts of M. balbisiana Colla leaves had 100% egg-hatching inhibition rates of Haemonchus contortus, while peel and roots showed 93.7% and 62% inhibition, respectively (188).
Menstrual Pain Relief Activity
Banana flowers contain several bioactive compounds like tannins, myoinositol phosphate, vitamin C, and α-tocopherol and are used to treat issues associated with menstrual bleeding and to facilitate lactation (78). Consuming cooked banana blossoms has been used since ancient times to reduce painful bleeding and alleviate muscle cramps by regulating progesterone hormone levels. Reports suggest banana blossoms may benefit women with polycystic ovarian syndrome (189). The antihemorrhagic action of banana blossoms has also been reported, and the flowers can be used as an infusion to alleviate discomfort during menstruation (1).
Hair Growth-Promoting Property
The ability of M. paradisiaca unripe fruit extract to stimulate hair growth was assessed in rats, where hair length and follicles were analyzed over 30 days. The study proved that the extract from unripe M. paradisiaca fruit could potentially promote hair development (190). Bananas are rich in natural oils, carbohydrates, vitamins, and potassium, contributing to hair softness, preventing breakage and split ends, and maintaining its elasticity and health. Using bananas on hair can enhance shine, promote growth, and help prevent dandruff (1). In a study with banana flower extract, there was an increase in hair root diameter, reduced hair loss, and decreased scalp redness, indicating its potential to stimulate hair growth and inhibit genes associated with hair loss while promoting hair-growth-related genes (191).
Conclusions
M. paradisiaca Linn. has survived ages and has a global distribution throughout tropical regions. The fruit satiates hunger and contributes immensely towards nutrition. The presence of carbohydrates, proteins, flavonoids, sterol glycoside, vitamins, minerals, and catecholamines gives it medicinal and dietetic properties. Several parts of the plant have been exploited for treating cancer, diabetes, depression, diarrhea, urolithiasis, and ulcers. It can also be used as an antibacterial, antiviral, antihyperlipidemia, and hepatoprotective agent. Its use for managing hypertension, atherosclerosis, hair problems and hair growth, wound healing, and many other activities has also been well studied. The available primary information paves the foundation for further phyto-analytical studies and clinical and toxicity evaluations. The future of M. paradisiaca lies in its ability to bridge food security, medicine, and sustainability. With the right scientific validation, it could become a cornerstone of global health solutions, offering affordable, natural alternatives to synthetic medicines and contributing to both preventative healthcare and treatments. Trends point towards its greater integration into the wellness, food, and pharmaceutical industries, supported by advances in phytoanalytical research and sustainable practices.
Declarations
Acknowledgment
We would like to express our sincere gratitude to Dr. Mini P.V., Head of the Department of Zoology at Government College Kasaragod, and Dr. Jayakrishnan T.V. for their invaluable support and guidance throughout the course of this study. Their expertise and encouragement have been instrumental in the completion of this comprehensive review. We are deeply appreciative of their contributions and dedication to advancing our understanding of the nutritional and pharmacological benefits of bananas.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
Not applicable.
Ethics Statement
Not applicable.
Funding Information
Not applicable.
References
- Kumar KS, Bhowmik D, Duraivel S, Umadevi M.Traditional and medicinal uses of banana. Journal of Pharmacognosy and Phytochemistry.2012; 1(3), 51-63.
- Singh B, Singh JP, Kaur A, Singh N. Bioactive compounds in banana and their associated health benefits - A review. Food Chem. 2016 1;206:1-11. doi: 10.1016/j.foodchem.2016.03.033. Epub 2016 Mar 11. PMID: 27041291.
- Nayar NM. The Bananas: Botany, Origin, Dispersal. In: Horticultural Reviews Vol. 36, Janick, J. (Ed). Wiley-Blackwell, Hoboken, New Jersey, US. 2010.pp 117-164.
- Ranjha, Modassar & Irfan, Shafeeqa & Nadeem, Muhammad & Mahmood, Shahid. A Comprehensive Review on Nutritional Value, Medicinal Uses, and Processing of Banana. Food Reviews International.2022; 38. 199-225. 10.1080/87559129.2020.1725890.
- Sidhu JS, Zafar TA. Bioactive Compounds in Banana Fruits and Their Health Benefits. Food Qual. Saf. 2018; 2 (4), 183–188. DOI: 10.1093/fqsafe/fyy019.
- Payal Kumari,Supriya S Gur,Ravindra K Tiwari.Banana and its by-products: A comprehensive review on its nutritional composition and pharmacological benefits .eFood 2023September 4:issue 5 /e110.https://doi.org/10.1002/edf2.110
- Afzal MF,Waseem Khalid, Sidra Akram, Muhammad Armghan Khalid, Muhammad Zubair, Safura Kauser, Khalid Abdelsamea Mohamedahmed, Afifa Aziz & Shahida Anusha Siddiqui. Bioactive profile and functional food applications of banana in food sectors and health: a review, International Journal of Food Properties,2022; 25:1, 2286-2300, DOI: 10.1080/10942912.2022.2130940
- Al-Snafi AE,Talab TA,Jafari-Sales A .Nutritional and therapeutic values of Musa paradisiaca-a review..Nativa,Sinop 2023 vol.11No.3 :396-40
- Ighodaro OM. Evaluation study on Nigerian species of Musa paradisiaca peels: Phytochemical screening, proximate analysis, mineral composition and antimicrobial activities. Researcher. 2012;4:17–20.
- Pareek S . Nutritional and Biochemical Composition of Banana (Musa Spp.) Cultivars. Nutri compost fruit cultivars.2016; 49–81.
- Ghoshal S. Steryl glycosides and acyl steryl glycosides from Musa paradisiaca. Phytochemstry.1985; 24(8):1807-1810. DOI: 10.1016/S0031-9422(00)82556-X
- Ragasa, C. Y., Martinez, A., Chua, J. E. Y., & Rideout, J. A. (2007). A triterpene from Musa errans. Philippine Journal of Science, 136(2), 167-171.
- Someya S, Yoshiki Y, Okubo K.Antioxidant compounds from bananas (Musa cavendish). Food Chemistry.2002; 79:351, 00186-354, 00183. DOI: 10.1016/S0308 (02)00186-3
- Hertog MG,Kromhout D,Aravanis C,Blackburn H,Buzina R,Fidanza F,Giampaoli S,Jansen A,Menotti A,Nedeljkovic S,et al .Flavonoid intake and long term risk of coronary heart disease and cancerin te seven countries study.Arch.Intern.Med.1995.Feb.27;155(4):381-386
- Martin TS, Ohtani K, Kasai R, Yamasaki K. A hemiterpenoid glucoside from Musa paradisiaca. Natural Medicine.2000; 54(4):190-192. ISSN: 13403443. ID: 110008732052
- Emaga TH, Andrianaivo RH, Wathelet B, Tchango JT, Paquot M. Effects of the stage of maturation and varieties on the chemical composition of banana and plantain peels. Food Chemistry.2007; 103:590-600. DOI: 10.1016/j.foodchem.2006.09.006
- Emaga TH, T., Robert, C., Ronkart, S.N., Wathelet, B., and Paquot, M., 2008. Dietary fibre components and pectin chemical features of peels during ripening in banana and plantain varieties. Bioresource Technology, 99(10), pp.4346-4354.
- Zaini, H.M., Roslan, J., Saallah, S., Munsu, E., Sulaiman, N.S., and Pindi, W., 2022. Banana peels as a bioactive ingredient and its potential application in the food industry. Journal of Functional Foods, 92, 105054.https://doi.org/10.1016/j.jff.2022.105054.
- Pereira, Aline Rodrigues and Marcelo Maraschin. “Banana (Musa spp) from peel to pulp: ethnopharmacology, source of bioactive compounds and its relevance for human health.” Journal of ethnopharmacology 160 (2015): 149-63 .
- Desai, B. B.; Deshpande, P. B. Chemical Transformation in Three Varieties of Banana (Musa Paradisiaca Linn.) Fruits Stored at 20 Degrees C. Mysore J Agri Sci. 1975.
- Dhull SB, Malik T, Kaur R, Kumar P, Kaushal N, Singh A. Banana Starch: Properties Illustration and Food Applications-A Review. Starch-Stärke. 2021;73(1–2), 2000085. DOI: 10.1002/star.202000085.
- Toledo TT, Nogueira S B, Cordenunsi BR, Gozzo FC, Pilau EJ, Lajolo F, Do Nascimento JROM, Do Nascimento JRO. Proteomic Analysis of Banana Fruit Reveals Proteins that are Differentially Accumulated during Ripening. Postharvest Bio. Technol.2012; 70, 51–58. DOI: 10.1016/j.postharvbio.2012.04.005.
- Phillips KM, McGinty RC, Couture G, Pehrsson PR, McKillop K, Fukagawa NK. Dietary fiber, starch, and sugars in bananas at different stages of ripeness in the retail market. PLoS One. 2021 Jul 8;16(7):e0253366. doi: 10.1371/journal.pone.0253366. PMID: 34237070; PMCID: PMC8266066.
- Kookal SK, Thimmaiah A. Nutritional Composition of Staple Food Bananas of Three Cultivars in India. American Journal of Plant Sciences,2018; 9, 2480-2493. https://doi.org/10.4236/ajps. 2018.912179
- Lau BF,Kong KW,Leong KH,Sun J,He X,Wang Z,Mustafa MR,Ling TC,IsmailA Banana inflorescence :its bioprospects asan ingredient for functional foods .Tren ds in food science and technology .2020.97:14-18
- USDA (United States Department of Agriculture). 2012. Nutrient Database. http://www.nal.usda.gov/fnic/foodcomp/Data/SR17/wtrank/sr17a306.pdf [Accessed: October 2023]
- Kanazawa K, Sakakibara H. 2000. High content of dopamine, a strong antioxidant, in Cavendish banana. J Agric Food Chem, 48: 844-848.
- Kandasamy S, Ramu S, Aradhya SM. 2016. In vitro functional properties of crude extracts and isolated compounds from banana pseudostem and rhizome. J Sci Food Agric, 96: 1347-1355.
- Ghag SB, Ganapathi TR.. Banana and Plantains: Improvement, Nutrition, and Health. In: Mérillon, JM., Ramawat, K. (eds) Bioactive Molecules in Food. Reference Series in Phytochemistry. Springer, Cham.2019; https://doi.org/10.1007/978-3-319-78030-6_73
- Russell WR, Labat A, Scobbie L, Duncan GJ, Duthie GG. Phenolic acid content of fruits commonly consumed and locally produced in Scotland. Food Chemistry, 2009; 115: 100–104.
- Kumar N, Pruthi V. Potential applications of ferulic acid from natural sources. Biotechnology Reports (Amsterdam, Netherlands).2014; 4: 86–93.
- Anyasi TA, Jideani AIO, Mchau GRA. Phenolics and essential mineral profile of organic acid pretreated unripe banana flour. Food Research International (Ottawa, Ont.).2018; 104: 100–109.
- Kevers C, Falkowski M, Tabart J, Defraigne JO, Dommes J, Pincemail J.Evolution of antioxidant capacity during storage of selected fruits and vegetables. J. Agric. Food Chem. 2007;55:8596–8603.doi:10.1021/jf071736j.
- Davey MW, Saeys W, Hof E, Ramon H,Swennen RL, Keulemans J. NApplication of Visible and near-infrared Reflectance Spectroscopy (Vis/NIRS) to Determine Carotenoid Contents in Banana (Musa Spp.) Fruit Pulp. J. Agric. Food Chem. 2009; 57(5), 1742–1751. DOI: 10.1021/jf803137d.
- Dhandapani R, Singh VP, Arora A, Bhattacharya RC, Rajendran A. Differential Accumulation of β-Carotene and Tissue Specific Expression of Phytoene Synthase (Mapsy) Gene in Banana (Musa Sp) Cultivars. Food Technol. 54(13), 4416–4426. DOI: 10.1007/s13197-017-2918-8.
- Mengstu A, Bachheti A, Abate L, Bachheti RK, Husen A. Health-Promoting Benefits, Value-Added Products, and Other Uses of Banana. In Non-Timber Forest Products; Springer, Cham. 2021; pp 339–364.
- Yin X, Quan J, Kanazawa T. Banana Prevents Plasma Oxidative Stress in Healthy Individuals. Plant Foods for Human Nutrition. 2008; 63: 71–76.
- Wilt T, Ishani A, MacDonald R, Stark G, Mulrow C, Lau J. Betasitosterols for Benign Prostatic Hyperplasia. Cochrane Database Systematic Reviews. 1999; 3.
- Berawi KN, Bimandama MA. The Effect of Giving Extract Ethanol of Kepok Banana Peel (Musa acuminata) Toward Total Cholesterol Level on Male Mice (Mus musculus L.) Strain Deutschland-Denken-Yoken (DDY) Obese. Biomedical and Pharmacology Journal. 2018; 11(2): 769–774. DOI: 10.13005/bpj/1431.
- Ikeda I, Kobayashi M, Hamada T, Tsuda K, Goto H, Imaizumi K, Nozawa A, Sugimoto A, Kakuda T. Heat-Epimerized Tea Catechins Rich in Gallocatechin Gallate and Catechin Gallate Are More Effective to Inhibit Cholesterol Absorption than Tea Catechins Rich in Epigallocatechin Gallate and Epicatechin Gallate. J Agric Food Chem. 2003; 51(25): 7303–7307. DOI: 10.1021/jf034728l. PMID: 14640575.
- Rasool MK, Sabina EP, Ramya SR, Preety P, Patel S, Mandal N, Mishra PP, Samuel J. Hepatoprotective and Antioxidant Effects of Gallic Acid in Paracetamol-Induced Liver Damage in Mice. J Pharm Pharmacol. 2010; 62(5): 638–643. DOI: 10.1211/jpp.62.05.0012. PMID: 20609067.
- Li C, Ford ES, Zhao G, Balluz LS, Giles WH, Liu S. Serum α-Carotene Concentrations and Risk of Death Among US Adults: The Third National Health and Nutrition Examination Survey Follow-Up Study. Archives of Internal Medicine. 2011; 171: 507–515.
- Choudhary SP, Tran LS. Phytosterols: Perspectives in Human Nutrition and Clinical Therapy. Current Medicinal Chemistry. 2011; 18: 4557–4567.
- Garbe D. Cinnamic Acid. Ullmann’s Encyclopedia of Industrial Chemistry. Wiley-VCH Verlag GmbH & Co, KGaA. 2000.
- Schaller H. The Role of Sterols in Plant Growth and Development. Progress in Lipid Research. 2003; 42, 163–175.
- Williamson G, Manach C. Bioavailability and Bioefficacy of Poly-Phenols in Humans. II. Review of 93 Intervention Studies. The American Journal of Clinical Nutrition. 2005; 81: 243S–255S.
- Young SN. How to Increase Serotonin in the Human Brain Without Drugs. Journal of Psychiatry & Neuroscience: JPN. 2007; 32: 394–399.
- Koshte VL, Dijk WV, Stelt MEVD, Aalberse RC. Isolation and Characterization of BanLec-I, a Mannoside-Binding Lectin from Musa paradisiaca (Banana). Biochem. J. 1990; 272: 721–726.
- Kuklin AI, Conger BV. Catecholamines in Plants. Journal of the Plant Growth Regulation. 1995; 14: 91–97.
- Siang ST. Use of Combined Traditional Chinese and Western Medicine in the Management of Burns. Panminerva Medica. 1983; 25: 197–202.
- Mulvena D, Webb EC, Zerner B. 3, 4-Dihydroxybenzaldehyde a Fungistatic Substance From Green Cavendish Bananas. Phytochemistry.1969; 8:393–5. doi: 10.1016/S0031-9422(00)85436-9
- Lee JR, Lee MH, Eo HJ, Park GH, Song HM, Kim MK, Lee JW, Jeong JB. The contribution of activation transcription factor 3 to apoptosis of human colorectal cancer cells by protocatechualdehyde, a naturally occurring phenolic compound. Archives of Biochemistry and Biophysics. 2014; 564(15):203–210.
- De Jong UW, Breslow N, Goh Hong JE, Sridharan M, Shanmugaratnam K. Aetiological Factors in Oesophageal Cancer in Singapore Chinese. Int J Cancer. 1974; 13:291–303. doi: 10.1002/ijc.2910130304
- Guha M, Basuray S, Sinha AK. Preventive Effect of Ripe Banana in the Diet on Ehrlich’s Ascitic Carcinoma Cell Induced Malignant Ascites in Mice. Nutr Res. 2003; 23:1081–8. doi: 10.1016/S0271-5317(03)00090-3
- Navarro SD, Mauro MO, Pesarini JR, Ogo FM, Oliveira RJ. Resistant Starch: A Functional Food That Prevents DNA Damage and Chemical Carcinogenesis. Genet Mol Res. 2015; 14:1679–91. doi: 10.4238/2015.March.6.14
- Malin AS, Qi D, Shu XO, Gao YT, Friedmann JM, Jin F. The Intake of Fruits, Vegetables and Selected Micronutrients in Relation to the Risk of Breast Cancer. Int J Cancer. 2003; 105:413–8. doi: 10.1002/ijc.11088
- Pettersson J, Karlsson PC, Göransson U, Rafter JJ, Bohlin L. The flavouring phytochemical 2-pentanone reduces prostaglandin production and COX-2 expression in colon cancer cells. Biol Pharm Bull. 2008; 31(3):534-7. doi: 10.1248/bpb.31.534. PMID: 18310926.
- Akamine K, Koyama T, Yazawa K. Banana Peel Extract Suppressed Prostate Gland Enlargement in Testosterone-Treated Mice. Biosci Biotechnol Biochem. 2009; 73:1911–4. doi: 10.1271/bbb.80770
- Jenshi Roobha J, Saravanakumar M, KM Aravindan and Suganya Devi P. In Vitro Evaluation of Anticancer Property of Anthocyanin Extract From Musa Acuminate Bract. Res Pharm. 2011; 1:17–21.
- Jeong JB, Lee SH. Protocatechualdehyde Possesses Anti-Cancer Activity Through Downregulating Cyclin D1 and HDAC2 in Human Colorectal Cancer Cells. Biochem Biophys Res Commun. 2013; 430:381–6. doi: 10.1016/j.bbrc.2012.11.018
- Nadumane VK, Timsina B. Anti-Cancer Potential of Banana Flower Extract: An In Vitro Study. Bangladesh J Pharmacol. 2014; 9:628–35. doi: 10.3329/bjp.v9i4.20610
- Dahham SS, Mohamad TA, Tabana YM, Majid AMSA. Antioxidant Activities and Anticancer Screening of Extracts From Banana Fruit (Musa Sapientum). Acad J Cancer Res. 2015; 8:28–34. doi: 10.5829/idosi.ajcr.2015.8.2.95162
- Saad SD, Mohamad TA, Tabana YM, Amin MSA. Antioxidant activities and anticancer screening of extracts from banana fruit (Musa sapientum). Academic Journal of Cancer Research. 2015; 8(2):28–34. DOI: 10.5829/idosi.ajcr.2015.8.2.95162
- Kandasamy S, Ramu S, Aradhya SM. In Vitro Functional Properties of Crude Extracts and Isolated Compounds From Banana Pseudostem and Rhizome. J Sci Food Agric. 2016; 96:1347–55. doi: 10.1002/jsfa.7229
- Srinivasan S, Valarmathi S, Illanchezian S, Dhanalakshmi KG. Anticancer Activity and DNA Fragmentation of Ferulic Acid From Banana Peel Using HeLa Cell Line. Int J Res Dev Technol. 2017; 7:631–5.
- Rajesh C, Sahoo S, Balaji SK, Prakash R, Selvapalam N, Palinichelvam K. Three compounds from banana pseudostem inhibit mitotic cell division by interacting with tubulin and cyclin-dependent kinase 2 proteins: in vivo in vitro and in silico approach. J Plant Biochem Biotechnol. 2024; 33:56–67. https://doi.org/10.1007/s13562-023-00861-1
- Liu LC, Lin YH, Lin YC, Ho CT, Hung CM, Way TD. Banana Flower Extract Suppresses Benign Prostatic Hyperplasia by Regulating the Inflammatory Response and Inducing G1 Cell-Cycle Arrest. In Vivo. 2018; 32:1373–9. doi: 10.21873/invivo.11389
- Ghafar SAA, Fikri IHH, Eshak Z. Antioxidant Activity of Musa Paradisiaca (Banana) Soft Pith and its Cytotoxicity Against Oral Squamous Carcinoma Cell Lines. Malaysian J Sci Health Technol. 2019; 3:1–11.
- Kumar PS, Durgadevi S, Saravanan A, Uma S. Antioxidant Potential and Antitumor Activities of Nendran Banana Peels in Breast Cancer Cell Line. Indian J Pharm Sci. 2019; 81:464–73. doi: 10.36468/pharmaceutical-sciences.531
- Barroso WA, Abreu IC, Ribeiro LS, da Rocha CQ, de Souza HP, de Lima TM. Chemical Composition and Cytotoxic Screening of Musa Cavendish Green Peels Extract: Antiproliferative Activity by Activation of Different Cellular Death Types. Toxicol In Vitro. 2019; 59:179–86. doi: 10.1016/j.tiv.2019.04.020
- Phacharapiyangkul N, Thirapanmethee K, Sa-Ngiamsuntorn K, Panich U, Lee CH, Chomnawang MT. Effect of Sucrier Banana Peel Extracts on Inhibition of Melanogenesis Through the ERK Signaling Pathway. Int J Med Sci. 2019;16:602–6. doi: 10.7150/ijms.32137.
- Srinivas BK, Shivamadhu MC, Jayarama S. Musa Acuminata Lectin Exerts Anti-Cancer Effects on HeLa and EAC Cells Via Activation of Caspase and Inhibitions of Akt, Erk, and Jnk Pathway Expression and Suppresses the Neoangiogenesis In in-Vivo Models. Int J Biol Macromol. 2021;166:1173–87. doi: 10.1016/j.ijbiomac.2020.10.272.
- Das A, Bindhu J, Deepesh P, Priya GS, Soundariya S. In Vitro Anticancer Study of Bioactive Compound Isolated From Musa Extract (Musa Acuminata). Indian J Public Health Res Dev. 2020;11:340–6. doi: 10.37506/v11/i1/2020/ijphrd/193841.
- Deep A, Upadhyay ON, Nandal R, Kumar S, Sharma AK, Wadhwa D. Anticancer Potential of Musa paradisiaca as Cervical Carcinoma and Malignant Melanoma. Res Square. 2021;1–16. doi: 10.21203/rs.2.20790/v1.
- Ojewole JA, Adewunmi CO. Hypoglycemic effect of methanolic extract of Musa paradisiaca (Musaceae) green fruits in normal and diabetic mice. Methods Findings in Experimental and Clinical Pharmacology. 2003;25(6):453.
- Chauhan A, Sharma PK, Srivastava P, Kumar N, Duehe R. Plants having potential antidiabetic activity: a review. Der Pharm Lett. 2010;2(3):369–87.
- Jawla S, Kumar Y, Khan MSY. Antimicrobial and antihyperglycemic activities of Musa paradisiaca flowers. Asian Pacific Journal of Tropical Biomedicine. 2012;2(2):s914-s918.
- Soni D, Saxena G. Complete nutrient profile of Banana flower: A review. Jour. Pl. Sci. Res. 2021;37(2):433–37.
- Nisha P, Mini S. Flavonoid-rich ethyl acetate fraction of Musa paradisiaca inflorescence down-regulates streptozotocin-induced oxidative stress, hyperglycemia, and mRNA levels of selected inflammatory genes in rats. Journal of Functional Foods. 2013;5(4):1838–47. doi: 10.1016/j.jff.2013.09.003.
- Marikkar JMN, Tan SJ, Salleh A, Azrina A, Shukri MAM. Evaluation of banana (Musa sp.) flowers of selected varieties for their antioxidative and anti-hyperglycemic potentials. Int Food Res J. 2016;23:1988–95.
- Navghare V, Dhawale SC. In vitro antioxidant, hypoglycemic and oral glucose tolerance test of banana peels. Alexandria Journal of Medicine. 2017;53(3):237–43. doi: 10.1016/j.ajme.2016.05.003.
- Bisht AS, Juyal D, Monika, Verma S, Joshi S. Study on in vitro antidiabetic potential of stem part of Musa paradisiaca Linn. International Journal of Comprehensive and Advanced Pharmacology. 2017;2(1):1–5.
- Borah M, Das S. Antidiabetic, antihyperlipidemic, and antioxidant activities of Musa balbisiana Colla in Type 1 diabetic rats. Indian J Pharmacol. 2017;49(1):71–76. doi: 10.4103/0253-7613.201030.
- Arun KB, Thomas S, Reshmitha TR, Akhil GC, Nisha P. Dietary fiber and phenolic-rich extracts from Musa paradisiaca inflorescence ameliorate type 2 diabetes and associated cardiovascular risks. J Funct Foods. 2017;31:198–207.
- Vilhena RO, Figueiredo ID, Baviera AM, Silva DB, Marson BM, Oliveira JA, Peccinini RG, Borges IK, Pontarolo R. Antidiabetic activity of Musa paradisiaca extracts in streptozotocin-induced diabetic rats and chemical characterization by HPLC-DAD-MS. Journal of Ethnopharmacology. 2020;254:112666. doi: 10.1016/j.jep.2020.112666.
- Wang M, Yang F, Yan X, Chao X, Zhang W, Yuan C, Zeng Q. Anti-diabetic effect of banana peel dietary fibers on type 2 diabetic mice induced by streptozotocin and high-sugar and high-fat diet. J Food Biochem. 2022;46(10):e14275. doi: 10.1111/jfbc.14275.
- Costa EAS, Franca CN, Fonseca FAH, Kato J, Bianco HT, Freitas T, Fonseca HA, Neto AMF, Izar MCO. Beneficial effects of green banana biomass consumption in patients with prediabetes and type 2 diabetes: A randomized controlled trial. The British Journal of Nutrition. 2019;121(12):1–38.
- Falcomer AL, Riquette RFR, de Lima BR, Ginani VC, Zandonadi RP. Health benefits of green banana consumption: A systematic review. Nutrients. 2019;11(6):1222.
- Alisi CS, Nwanyanwu CE, Akujobi CO, Ibegbulem CO. Inhibition of dehydrogenase activity in pathogenic bacteria isolates by aqueous extracts of Musa paradisiaca (var. sapientum). African Journal of Biotechnology. 2008;7(12):1821–25.
- Jahan M, Warsi M, Khatoon F. Concentration influence on antimicrobial activity of banana blossom extract-incorporated chitosan-polyethylene glycol (CS-PEG) blended film. Journal of Chemical and Pharmaceutical Research. 2010;2(5):373–78.
- Karadi RV, Shah A, Parekh P, Azmi P. Antimicrobial activities of Musa paradisiaca and Cocos nucifera. International Journal of Research in Pharmaceutical and Biomedical Sciences. 2011; 2(1), 264-267.
- Karuppiah P, Mustaffa M. Antibacterial and antioxidant activities of Musa sp. leaf extracts against multidrug-resistant clinical pathogens causing nosocomial infection. Asian Pac J Trop Biomed. 2013; 3(9), 737-742. https://doi:10.1016/S2221-1691(13)60148-3
- Chabuck ZAG, Al-Charrakh AH, Hindi NKK, Hindi SKK. Antimicrobial effect of aqueous banana peel extract, Iraq. Res Gate Pharm Sci. 2013; 1:73–75.
- Kapadia SP, Pudakalkatti PS, Shivanaikar S. Detection of antimicrobial activity of banana peel (Musa paradisiaca L.) on Porphyromonas gingivalis and Aggregatibacter actinomycetemcomitans: An in vitro study. Contemp Clin Dent. 2015; 6(4), 496-499. doi: 10.4103/0976-237X.169864. PMID: 26681854; PMCID: PMC4678547.
- Asuquo EG, Udobi CE. Antibacterial and toxicity studies of the ethanol extract of Musa paradisiaca leaf. Cogent Biology. 2016; 2(1), 1219248.
- Okorondu SI, Mepba HD, Okorondu MMO, Aririatu LE. Antibacterial properties of Musa paradisiaca peel extract. Curr Trends Microbiol. 2010; 6, 21–26.
- Amutha K, Selvakumari U. Wound healing activity of methanolic stem extract of Musa paradisiaca Linn. (Banana) in wistar albino rats. International Wound Journal. 2016; 13(5), 763-767.
- Evbuomwan L, Onodje GO, Jacob I, Patric CE. Evaluating the antibacterial activity of Musa acuminata (banana) fruit peels against multidrug-resistant bacterial isolates. International Journal of Novel Research in Life Sciences. 2018; 5(3), 26–31.
- Ismail TN, Awang RA, Mohamad S, Shahidan WNS. Chemical compounds and antimicrobial activity of acetone Musa acuminate AA/AAA leaf stalk extracts on selective gram-negative bacteria. Malaysian Journal of Analytical Sciences. 2018; 22(6), 957–964. https://doi.org/10.17576/mjas-2018-2206-05
- Lalita Ambigai Sivasamugham, Virusha Nimalan, Geetha Subramaniam. Antibacterial effects of Musa sp. ethanolic leaf extracts against methicillin-resistant and susceptible Staphylococcus aureus. South African Journal of Chemical Engineering. 2021; 35, 107-110. https://doi.org/10.1016/j.sajce.2020.09.007
- Singh DD, Saikrishnan K, Kumar P, Surolia A, Sekar K, Vijayan M. Unusual sugar specificity of banana lectin from Musa paradisiaca and its probable evolutionary origin. Crystallographic and modelling studies. Glycobiology. 2005; 15(10), 1025-1032.
- Swanson MD, Winter HC, Goldstein IJ, Markovitz DM. A lectin isolated from banana is a potent inhibitor of HIV replication. J Biol Chem. 2010; 285(12), 8646-8655.
- Swanson MD, Boudreaux DM, Salmon L, Chugh J, Winter HC, Meagher JL, André S, Murphy PV, Oscarson S, Roy R. Engineering a therapeutic lectin by uncoupling mitogenicity from antiviral activity. Cell. 2015; 163, 746–758. doi: 10.1016/j.cell.2015.09.056
- Covés-Datson EM, Dyall J, DeWald LE, King SR, Dube D, Legendre M, Nelson E, Drews KC, Gross R, Gerhardt DM, Torzewski L, Postnikova E, Liang JY, Ban B, Shetty J, Hensley LE, Jahrling PB, Olinger GG Jr, White JM, Markovitz DM. Inhibition of Ebola Virus by a molecularly engineered banana lectin. PLoS Negl Trop Dis. 2019; 13(7): e0007595. doi: 10.1371/journal.pntd.0007595. PMID: 31356611; PMCID: PMC6687191.
- Panda SK, Castro AHF, Jouneghani RS, Leyssen P, Neyts J, Swennen R, Luyten W. Antiviral and cytotoxic activity of different plant parts of banana (Musa spp.). Viruses. 2020; 12(5), 549. doi: 10.3390/v12050549. PMID: 32429324; PMCID: PMC7291111.
- Laura Junqueira de Camargo, Tony Picoli, Geferson Fischer, Ana Claudia Oliveira de Freitas, Rodrigo Bozembecker de Almeida, Luciano da Silva Pinto. Antiviral activity of native banana lectin against bovine viral diarrhea virus and bovine alpha herpes virus type 1. International Journal of Biological Macromolecules. 2020; 157, 569–576. https://doi.org/10.1016/j.ijbiomac.2020.04.125
- Okorondu SI, Akujobi C, Nwachukwu I. Antifungal properties of Musa paradisiaca (Plantain) peel and stalk extracts. International Journal of Biological and Chemical Sciences. 2012; 6, 10.4314/ijbcs.v6i4.12.
- Bankar AM, Dole MN. Formulation and evaluation of herbal antimicrobial gel containing Musa acuminata leaves extract. Journal of Pharmacy and Phytochemistry. 2016; 5(1), 01–03.
- Bisht R, Chanyal S, Agrawal P. Antimicrobial and phytochemical analysis of leaf extract of medicinal fruit plants. Asian Journal of Pharmacy and Clinical Research. 2016; 9(4), 131–136.
- Prakash B, Sumangala CH, Melappa G, Gavimath C. Evaluation of antifungal activity of banana peel against scalp fungi. Mater Today Proc. 2017; 4, 11977–11983.
- Harith SS, Yasim NH, Harun A, Omar WS, Musa MS. Phytochemical screening, antifungal and antibacterial activities of Musa acuminata plant. Malaysian Journal of Analytical Sciences.2018; 22(3), 452–457. https://doi.org/10.17576/mjas-2018-2203-11
- Devi VK, Baskar R, Varalakshmi P. Biochemical effects in normal and stone forming rats treated with the ripe kernel juice of plantain (Musa paradisiaca). Anc Sci Life. 1993; Jan;12(3-4):451-61. PMID: 22556626; PMCID: PMC3336560.
- Gopakumara Pillai R. The core of the pseudo stem of Musa in the treatment of urinary stones. Ancient Science of Life., 1995; 15(1): 2 -6.
- Joy JM, Prathyusha S, Mohanalakshmi S, Praveen Kumar AVS, Kumar CKA. Potent herbal wealth with litholytic activity: A Review. International Journal of Innovative Drug Discovery., 2012; 2(2): 66-75.
- Kalpana S. Inhibition of calcium oxalate crystallization in vitro by extract of banana cultivar monthan. International Journal of Pharmacy and Pharmaceutical Sciences.2013; 5. 649-653.
- Prasobh GR, Revikumar KG. A study on the use of Musa species in the management of renal calculi. Asian Journal of Pharmaceutical and Clinical Research. 2014; 7(3): 123-125.
- Panigrahi PN, Dey S, Sahoo M, Dan A. Antiurolithiatic and antioxidant efficacy of Musa paradisiaca pseudostem on ethylene glycol-induced nephrolithiasis in rat. Indian J Pharmacol. 2017; Jan-Feb;49(1):77-83. doi: 10.4103/0253-7613.201026. PMID: 28458427; PMCID: PMC5351243.
- Karunasree Varicola, Amreen S. A., Dintyala K., & Ventrapati G. Fruit peel extracts with potential for dissolving simulated renal stones in in vitro conditions. International Journal of Pharmacy and Pharmaceutical Sciences, 2018; 10(11), 74-77.
- Abu Zarin M, Joo Shun T, Murugan P, Rosma A. Investigation of potential anti-urolithiatic activity from different types of Musa pseudo-stem extracts in inhibition of calcium oxalate crystallization. BMC Complementary Medicine and Therapies. 2020; 20. 317. 10.1186/s12906-020-03113-0.
- Gopalakrishna H N, Rajeshwari S, Pemminati S, Ashok S K, Alwar M C. A preliminary study on antidepressant activity of NR-ANX-C polyhedral product) in mice. J. Pharmacy Res. 2010; 3:550-553.
- Milind Parle, Suman Malik. Anti-Depressant Potential of Banana Fruit. International Journal of Medical Sciences. 2010; (October, 2009 to March, 2010) Vol. 2 Issue 2: 148-151.
- Tee T P, Halijah Hassan. Antidepressant-Like Activity of Banana Peel Extract in Mice. Current Research in Medicine. 2011; 2: 59-64.
- Samad N, Muneer A, Ullah N, Zaman A, Ayaz MM, Ahmad I. Banana fruit pulp and peel involved in antianxiety and antidepressant effects while invigorating memory performance in male mice: Possible role of potential antioxidants. Pak J Pharm Sci. 2017; 30(3(Suppl.)):989-995. PMID: 28655697.
- Salako OA, Akindele AJ, Balogun AO, Adeyemi OO. Investigation of Antidepressant, Anxiolytic, and Sedative Activities of the Aqueous Leaf Extract of Musa sapientum Linn. (Banana; Musaceae). Drug Res (Stuttg). 2019; 69(3):136-143. doi: 10.1055/a-0651-7978. Epub 2018 Aug 3. PMID: 30075482.
- Jyothirmayi N, Rao NM. Banana Medicinal Uses. J. Med. Sci. Tech. 4(2):152-160.
- Tavakkoli-Kakhki M, Motavasselian M, Mosaddegh M, Esfahani MM, Kamalinejad M, Nematy M. Omega-3 and Omega-6 content of medicinal foods for depressed patients: Implications from the Iranian traditional medicine. Avicenna Journal of Phytomedicine. 2014; 4:225-230.
- Iweala EEJ, Obichi IC, Omotosho OE. Biochemical and Histological Responses of Hepatotoxic Rats Fed Musa paradisiaca L. Supplemented Diet. International Journal of Pharmacology. 2011; 4:471-477.
- Nirmala M, Girija K, Lakshman K, Divya T. Hepatoprotective activity of Musa paradisiaca on experimental animal models. Asian Pacific Journal of Tropical Biomedicine. 2012; 2(1):11-15.
- Rajagopalan R, Aruna K, Penumathsa S, Menon VP. Hepatoprotective role of ferulic acid: A dose-dependent study. Journal of Medicinal Food. 2004; 7(4):456-461.
- Oyewole OI, Akinbamijo TO, Omoboriowo OJ, Ukana NU. Hepatorenal Protective Activities of Plantain Root (Musa paradisiaca) on Arsenic-induced Oxidative Damage in Rats. Advances in Biochemistry. 2015; 3(2):30-34.
- Manish Kumar Prasad, Sukanta Sen, Nadeem Arshad, Matiur Rahman, Choudhary CB. Comparative study of methanolic extract of banana, ranitidine, and omeprazole on total acid secretion and gastric ulcer healing effect in albino rats. International Journal of Contemporary Medical Research. 2020; 7(2):B1-B6. DOI: http://dx.doi.org/10.21276/ijcmr.2020.7.2.9
- Lewis DA, Fields WN, Shaw GP. A natural flavonoid present in unripe plantain banana pulp (Musa sapientum L. var. paradisiaca) protects the gastric mucosa from aspirin-induced erosions. Journal of Ethnopharmacology. 1999; 65:283–288.
- Enemchukwu BN, Onyedinma EI, Ubaoji KI, Ngele KK. Antiulcer effect of aqueous extract of unripe plantain peels on male Wistar (albino) rats. Biotechnology an Indian Journal. 2014; 9(12):511-515.
- Pannangpetch P, Vuttivirojana A, Kularbkaew C, Tesana S, Kongyingyoes B, Kukongviriyapan V. The antiulcerative effect of Thai Musa species in rats. Phytotherapy Research. 15:407–410.
- Goel RK, Tavares IA, Bennett A. Stimulation of gastric and colonic mucosal eicosanoid synthesis by plantain banana. J Pharm Pharmacol. 1989; Nov;41(11):747-750. doi: 10.1111/j.2042-7158.1989.tb06358.x. PMID: 2576041.
- Goel RK, Sairam K. Antiulcer drugs from indigenous sources with emphasis on Musa sapientum, Tamrabhasma, Asparagus racemosus, and Zingiber officinale. Indian Journal of Pharmacology. 34(2):100-110.
- Prabha P, Karpagam T, Varalakshmi B, Packiavathy AS. Indigenous anti-ulcer activity of Musa sapientum on peptic ulcer. Pharmacognosy Res. 2011; Oct;3(4):232-238. doi: 10.4103/0974-8490.89742. PMID: 22224045; PMCID: PMC3249781.
- Herbert BE, Joyce OO, Chukwudum KA, Onuebunwa CL. Cytoprotective Effects of Musa Paradisiaca and in combination with catecholamines on indomethacin-induced peptic ulceration in rats. American Journal of Pharm Tech Research. 2013; 3(4):702-711.
- Ganwar AK, Ghosh AK. To estimate the antiulcer activity of leaves of Musa sapientum Linn. by ethanol-induced method in rats. International Journal of Pharmacognosy and Phytochemical Research. 2014; 6(1):53-55. Available from: www.ijppr.com
- Rao MUS, Ahmad BA, Mohd KS. Antiulcer activity of Musa paradisiaca (Banana) tepal and skin extracts in ulcer-induced albino mice. Malaysian Journal of Analytical Sciences. 2016; 20(5):1203-1216.
- Onasanwo SA, Emikpe BO, Ajah AA, Elufioye TO. Anti-ulcer and ulcer-healing potentials of Musa sapientum peel extract in laboratory rodents. Pharmacognosy Res. 2013; Jul;5(3):173-178. doi: 10.4103/0974-8490.112423. PMID: 23900937; PMCID: PMC3719258.
- Arias MM, Alcaráz GM, Bernal C, González G. Oral rehydration with a plantain flour-based solution in children dehydrated by acute diarrhea: a clinical trial. Acta Paediatrica. 1997; 86(10):1047-1051.
- Emery EA, Ahmad S, Koethe JD, Skipper A, Perlmutter S, Paskin DL. Banana flakes control diarrhea in enterally fed patients. Nutrition in Clinical Practice. 1997; 12(2):72-75.
- Rabbani GH, Teka T, Saha SK, Zaman B, Majid N, Khatun M, Wahed MA, Fuchs GJ. Green banana and pectin improve small intestinal permeability and reduce fluid loss in Bangladeshi children with persistent diarrhea. Dig Dis Sci. 2004; 49(3):475-484. doi: 10.1023/b:ddas.0000020507.25910.cf. PMID: 15139502.
- Binder HJ. Role of colonic short-chain fatty acid transport in diarrhea. Annual Review of Physiology. 2010; 72:297–313.
- Yakub MT, Nurudeen QO, Salimon SS, Yakubu MO, Jimoh RO, Nafiu MO, Akanji MA, Oladiji AT, Williams FE. Antidiarrhoeal activity of Musa paradisiaca sap in Wistar rats. Hindawi. 2015; Article ID 683726. https://doi.org/10.1155/2015/683726.
- Ningsih Arista Wahyu, Maftuchah Rochmanti, Achmad Basori. Effectiveness of Antidiarrheal Unripe Wooden Banana (Musa paradisiaca L.) in male Balb-C mice induced with Escherichia coli. Fol Med Indones. 2020; Vol. 56 No. 3 September:208-215.
- Martin Adhitya Subagio, Cecilia Noviyanti Salim, Qorina Putri Srisantoso, Yora Nindita, Astika Widy Utomo, Nani Maharani. Utilization of banana (Musa paradisiaca L.) peel as pectin source as antidiarrheal on castor oil-induced diarrheal Wistar rats model. Journal of the Medical Sciences (Berkala Ilmu Kedokteran). 2020; Volume 52, Number 2; http://dx.doi.org/1019106/ JMedSci005202202002
- Agarwal PK, Singh A, Gaurav K, Goel S, Khanna HD, Goel RK. Evaluation of wound healing activity of extracts of plantain Banana (Musa sapientum Var. Paradisiaca) in rats. Indian J Exp Bio. 2009; 47:32–40.
- Weremfo A, Pappoe ANM, Adinortey MB. Wound Healing Potential of Musa paradisiaca L. (Musaceae) Stem Juice Extract Formulated into an Ointment. Research J. Pharmacology and Pharmacodynamics. 2011; 3(6):294-296.
- Rosida, Sukardiman, Khotib J. The increasing of VEGF expression and re-epithelialization on dermal wound healing process after treatment of banana peel extract (Musa acuminata Colla). International Journal of Pharmacy and Pharmaceutical Sciences, 2014; 6(11):427-430.
- Von Atzingen DA, Mendonça AR, Mesquita Filho M, Alvarenga VA, Assis VA, Penazzo AE, Muzetti JH, Rezende TS. Repair of surgical wounds in rats using a 10% unripe Musa sapientum peel gel. Acta Cir Bras. 2015; Sep;30(9):586-92. doi: 10.1590/S0102-865020150090000001. PMID: 26465101.
- Tamri P, Aliasghar Hemmati, Ali Amirahmadi, Jaber Zafari, Babak Mohammadian, Mohammadshafi Dehghani, N allahyari. Evaluation of wound healing activity of hydroalcoholic extract of banana (Musa acuminata) fruits peel in rabbit. Archives. 2016; 3:203-8.
- Padilla-Camberos E, Flores-Fernández JM, Canales-Aguirre AA, Barragán-Álvarez CP, Gutiérrez-Mercado Y, Lugo-Cervantes E. Wound healing and antioxidant capacity of Musa paradisiaca Linn. peel extracts. J Pharm Pharmacogn Res. 2016; 4(5):165-173.
- Akpanyung E, Archibong I, Umoh I, Bassey U. Effect of methanol extract of the unripe peels of Musa paradisiaca on some haematological and biochemical indices in male albino wistar rats. International Journal of Agriculture and Environmental Research. 2018; 4(6):1250-1257.
- Achmad H, Putri AP. Contents of banana peel extract as hemostasis in wound healing. 2021. Annals of the Romanian Society for Cell Biology, 4800-4810. https://annalsofrscb.ro/index.php/journal/article/view/625
- Usha V, Vijayammal PL, Kurup PA. Effect of dietary fiber from banana (Musa paradisiaca) on cholesterol metabolism. Indian Journal of Experimental Biology. 1984; 22(10):550-554.
- Vijayakumar S, Presannakumar G, Vijayalakshmi NR. Investigations on the effect of flavonoids from banana, Musa Paradisiaca L., on lipid metabolism in rats. Journal of Dietary Supplements. 2009; 6(2):111-123.
- Marangoni F, Poli A. Phytosterols and cardiovascular health. Pharmacol Res. 2010; 61(3):193-199. doi: 10.1016/j.phrs.2010.01.001. Epub 2010 Jan 11. PMID: 20067836.
- Horigome T, Sakaguchi E, Kishimoto C. Hypocholesterolaemic effect of banana pulp in the rat fed on cholesterol-containing diet. Br J Nutr. 1992; 68(1):231-244.
- Mosa ZM, Khalil AF. The effect of banana peels supplemented diet on acute liver failure rats. Ann Agricult Sci. 2015; 60:373–379.
- Uhegbu FO, Imo C, Onwuegbuchulam CH. Hypoglycemic, hypolipidemic, and antioxidant activities of Musa Paradisiaca supplemented diet on Alloxan-induced diabetic albino rats. Asian Journal of Biochemistry. 2016; 11(3):162-167.
- Vilela, Carla, Santos, Sonia, Villaverde, Juan, Oliveira, Lúcia, Nunes, Alberto, Cordeiro, Nereida, Freire, Carmen, Silvestre, Armando. Lipophilic phytochemicals from banana fruits of several Musa species. Food Chemistry. 2014; 162:247-252. doi: 10.1016/j.foodchem.2014.04.050
- Indriawati R, Atiyah FU. IOP Conf. Ser.: Earth Environ. Sci. 2022; 985:012040. DOI: 10.1088/1755-1315/985/1/012040
- Perfumi M, Massi M, de Caro G. Effects of banana feeding on deoxycorticosterone-induced hypertension and salt consumption in rats. International Journal of Pharmacognosy. 1994; 32(2):115-125. https://doi.org/10.3109/13880209409082982
- Orie NN. Direct vascular effects of plantain extract in rats. Experimental Physiology. 1997; 82(3):501-506.
- Sarkar C, Bairy KL, Rao NM, Udupa EG. Effect of banana on cold stress test and peak expiratory flow rate in healthy volunteers. Indian J Med Res. 1999; 110:27-29. PMID: 10709336.
- Suherman J, Megawati R. Effect of Pisang ambon (Musa acuminata Colla) on adult female blood pressure during a cold stress test. Jurnal Medika Planta. 2010; 1(2):21-25.
- Rao N, Mallikarjuna KVS, Prasad RG, Pai KSR. Angiotensin-converting enzyme inhibitors from ripened and unripened bananas. Current Science. 1999; 76(1):86-88.
- Fitri Y, Suryana S, Ahmad A, Hendra A, Fitrianingsih E, Arnisam, Yunianto AE. Effectiveness of banana juice (Musa acuminata Linn.) on blood pressure, blood sugar levels, and low-density lipoprotein in the elderly. Food Research. 2022; 6(3):239-244. DOI: https://doi.org/10.26656/fr.2017.6(3).213
- Osim EE, Ibu JO. The Effect of Plantains (Musa paradisiaca) on DOCA-Induced Hypertension in Rats. Pharm. Biol. 1991; 29(1): 9-13.
- Osim EE, Orie NN, Bose S, Etra KM. The effect of plantain and banana extracts on blood pressure and heart rate in albino rats. Nigerian J. Physiol. Sci. 1990; 6: 114-119.
- Dayanand G, Sharma A, Ahmed M, Jyothi PP, Rani M. Effect of banana on blood pressure of hypertensive individuals: a cross-sectional study from Pokhara, Nepal. Medical Science. 2015; 3(2):233-7.
- Pramono A, Noriko N, Komara SB. Musa paradisiaca on the elderly female hypertensive patient. IOP Conference Series: Materials Science and Engineering. 2017; 193, 012058. https://doi.org/10.1088/1757-899X/193/1/012058
- Chrisanto E.Y. The Effectiveness of Ambon Banana Against Lowering Blood Pressure in Patients with Hypertension in the Work Area of Krui Health Center, Pesisir Barat Regency. Jurnal Kesehatan Holistik (The Journal of Holistic Healthcare). 2017; 11(3), 167–174.
- Yulianti, I. and Prameswari, E.V.T.W. (2019). The Effect of Giving Ambon Banana to Blood Pressure in Elderly Patients with Hypertension. Journal of Nurse and Midwifery, 6(1), 070–076. https://doi.org/10.26699/jnk.v6i1.ART.p070–076
- Saraswathi NT, Gnanam FD. Effect of medicinal plants on the crystallization of cholesterol. Journal of Crystal Growth. 1997; 179(3-4), 611-617.
- Parmar HS, Kar A. Protective role of Citrus sinensis, Musa paradisiaca, and Punica granatum peels against diet-induced atherosclerosis and thyroid dysfunctions in rats. Nutrition Research. 2007; 27(11), 710-718.
- Arlinda SP, Kartika PR, Eria NP, Dyah PA, Wafiyah H. The effectiveness of Ambon banana peel extract (Musa sapientum) as atherosclerosis prevention through inhibition of NF-κβ and increased e-NOS expression in atherogenic rat model. International Journal of Medical Research and Health Sciences. 2017; 6(9):114-120. Available from: www.ijmrhs.com
- Biswas C, Basak D, Chakaroverty R, Banerjee A, Dey S, Mazumder UK. Effect of methanol extract of Musa paradisiaca (LINN) stem juice on chemically induced acute inflammation. International J. of Pharmacy and Pharmaceutical Sciences. 2012; 4:148-150.
- García Mesa MT, Duménigo González A, Acosta de la Luz LL. Antiallergic potential of a pseudo-stem powder of Musa paradisiaca L. (banana). Int J Phytocos Nat Ingred. 2019;6:5. doi:10.15171/ijpni.2019.05.
- Patro G, Panda M, Das P, Bhaiji A, Panda HB. Pharmacological evaluation of Musa paradisiaca (Linn.) on bronchial asthma. Egypt. Pharm. J. 2016; 15, pp. 25-30. doi:10.4103/1687-4315.184030.
- Francisco Allysson Assis Ferreira Gadelha, Raquel Fragoso Pereira Cavalcanti, Giciane Carvalho Vieira, Larcia Karla Diega Paiva Ferreira, Gabriela Ribeiro de Sousa, José Maria Barbosa Filho, Mario A. Barbosa, Susana Gomes dos Santos, Marcia Regina Piuvezam. Immunomodulatory properties of Musa paradisiaca L. inflorescence in Combined Allergic Rhinitis and Asthma Syndrome (CARAS) model towards NF-κB pathway inhibition. Journal of Functional Foods. 2021; Volume 83, 104540. https://doi.org/10.1016/j.jff.2021.104540.
- Venkatesh V, Krishna K, Girish Kumar K, Pradeepa SR, Santosh Kumar K, Vijay. Anthelmintic Activity of Musa paradisiaca (L.) cv. Puttabale. International Journal of Pharmaceutical Sciences and Drug Research. 2013; 67-69.
- Danamoni Sai Krishna Priya, Vaddesandhya, Buragayala Gayathri, Guntupalli Gopikrishna, Varicolakaruna Sree. Peels of Selected Fruits with Antioxidant and Anthelmintic potential. Int. J. Curr. Microbiol. App. Sci. 2016; 5(2): 649-660. doi: http://dx.doi.org/10.20546/ijcmas.2016.502.073.
- Gregory L, Yoshihara E, Silva LKF, Marques EC, Ribeiro BLM, de Souza Meira EB, Rossi RS, do Amarante AFT, Hasegawa MY. Anthelmintic Effects of Dried Ground Banana Plant Leaves (Musa Spp.) Fed to Sheep Artificially Infected With Haemonchus Contortus and Trichostrongylus Colubriformis. Afr J Tradit Complement Altern Med. 2016; 14(1):138-144. doi:10.21010/ajtcam.v14i1.15. PMID: 28480391; PMCID: PMC5411864.
- Annastasya Yuliani Rahayu, Retno Sri Wahjuni, Dewa Ketut Meles, Retno Wulansari. Anthelmintic Efficacy of Ethanol Extract of Raw Kepok Banana Peel (Musa paradisiaca L.) Against Ascaridia galli Worms In Vitro. Journal of Parasite Science. 2020; Vol. 4 No. 2 September 2020. eISSN: 2656-5331, pISSN: 2599-0993.
- Rivera-Torres Diana Laura, Hernandez Villegas MM, Bolio López GI, Almenares López D, Córdova Sánchez S, De la Cruz Burelo P. In vitro Anthelmintic activity of Musa balbisiana Colla (Square banana) against Haemonchus contortus eggs. Agro Productividad. 2021; 14(4). https://doi.org/10.32854/agrop.v14i4.1877.
- Jadhav SS, Kachare S, Hole K. A review on banana blossom as an effective agent for polycystic-ovarian syndrome (PCOD). International Journal of Noval Research and Development. 2023; 8(12):1-11. ISSN:4256-4184.
- Savali AS, Bhinge SD, Chitapurkar HR. Evaluation of hair growth promoting activity of Musa paradisiaca unripe fruit extract. Journal of Natural Pharmaceuticals. 2011; 2(3), 120-124.
- Liang C-H, Yung-Hsiang Lin, Yung-Kai Lin, Chi-Fu Chiang.Hair growth-promotion effects and antioxidant activity of the banana flower extract HappyAngel®: double-blind, placebo-controlled trial,Food Science and Human Wellness.2023;Volume 12, Issue 5, Pages 1917-1923,ISSN 22134530,https://doi.org/10.1016/ j.fshw.2023.02.043.
