REVIEW
Toxicity and Safety Analysis of Polyhexamethylene Guanidine: A Comprehensive Systematic Review
Academic Editor: Adeleye Ademola Olutayo
Sciences of Pharmacy|Vol. 3, Issue 3, pp. 153-166 (2024)
Received
Jul 14, 2024Revised
Aug 17, 2024Accepted
Aug 31, 2024Published
Sep 9, 2024
Abstract
Introduction
The emergence and spread of resistant bacteria, combined with the shortage of new antibiotics, has escalated into a global health crisis. Nevertheless, research and development programs for new antimicrobial agents are considered unattractive investments for pharmaceutical companies. Among the potential solutions to delay the onset of the "post-antibiotic era" is the use of established antibiotics in combination with the local application of antiseptics, which is common practice, for example, in wound treatment. In ophthalmology, particularly in developing countries, it has even been suggested to use antiseptics instead of antibiotics for the treatment of conjunctivitis or keratitis due to the low availability of the latter. However, given the increasing number of pathogens resistant to antiseptics as well, there is an urgent need for antimicrobial approaches that can effectively inactivate pathogens without the risk of developing resistance. Polyhexamethylene guanidine (PHMG) may be used to reduce the spread of antibiotic resistance by inactivating extracellular DNA (eDNA), commonly found in bacterial biofilms, and by limiting the development of the biofilms themselves (1-7).
PHMG belongs to the family of polymeric guanidines and have been used for many years as antiseptics. It is often the subject of numerous studies in medicine, vaccine production, veterinary, agriculture, food industry, and water purification systems (8-16). The main representative of the class of polyguanidines is PHMG hydrochloride (PHMG-H). PHMG phosphate (PHMG-P) has stronger biocidal and flocculating properties. Despite the fact that PHMG-H and PHMG-P are the most common modifications of PHMG, there are also other chemical modifications, for example, PHMG salt with citric acid and others. It is believed that PHMG hydrocitrate, compared with PHMG-H, has increased biocidal activity against bacteria, mold fungi, and yeast, while having lower toxicity (17). There is experience in obtaining PHMG hydrosuccinate. It was shown that PHMG hydrosuccinate has a more pronounced sporicidal effect against Aspergillus niger than PHMG-H. PHMG hydrosuccinate is also superior to PHMG-H in its disintegrating effect on formed biofilms of Pseudomonas aeruginosa. Therefore, this compound may be useful for the treatment of infectious eye diseases (18). A natural question arises: is it possible to use PHMG and its derivatives in human medicinal products, and how safe is it?
Despite the widespread use of PHMG in different fields, its safety has come under comprehensive scrutiny, and such concerns are not unfounded. Evidence of this lies in the health damage caused by PHMG in humidifier disinfectants (HD) available for sale in South Korea from 1994 to 2011. These products led to interstitial lung diseases (ILD) among the population, with average PHMG concentrations in HD brands reaching 3100.9 ppm (19, 20). It is important to note that the average molecular weights of PHMG in products HD range from 422 to 678 g/mol, indicating the oligomeric nature of PHMG, where each isomeric polymer exhibits different biocidal effects depending on the end group (21). Subsequently, most studies have focused on investigating the toxic effects and mechanisms of action of PHMG when inhaled. Another significant incident that drew serious criticism towards PHMG involved a surge in cases of poisoning from alcohol-containing household chemical products, affecting 12, 500 people. The presumed concentration of PHMG in the consumed products ranged from 0.10 to 0.14 ± 0.01%. For these reasons, the European Commission prohibited the placement on the market and use of PHMG as a biocide through the directive of the European Parliament and the Council of the European Union (22-24).
The main objective of this systematic review was to comprehensively assess the toxicity and safety of PHMG not only through inhalation but also through oral ingestion, skin contact, and eye exposure. Evaluative tests included studies conducted on human cells and those involving patients, including those who succumbed to the consequences of exposure to this compound. This information will help establish the guidelines for using PHMG in medicines and medical devices.
Methodology
Inclusion and Exclusion Criteria
Articles were included based on the following criteria: (I) published work with full text; (II) coverage of various salts of PHMG, particularly PHMG-H, and PHMG-P, as they represent the most common chemical modifications used, along with their various applications in the System Organ Class: oral cavity, eyes, skin, liver, kidneys, heart, blood components, reproductive function; (III) assessment of the toxicity of these compounds in vitro, primarily in human cell models; (IV) evaluation of the toxicity of these compounds in animal studies, primarily in rodents, providing data on acute and subchronic toxicity, as well as reproductive toxicity, nephrotoxicity, and cardiac toxicity; (V) assessment of potential adverse events in healthy volunteers and patients, including those who died as a result of exposure to the study compounds; (VI) regardless of the year of publication; and (VII) in vitro and in vivo studies covering the maximum possible number of test models, as well as a wide range of substance concentrations.
The exclusion criteria were the following: articles (I) that do not evaluate the toxicity/safety of PHMG, for example, articles devoted to the study of compositions containing PHMG, or various modifications of PHMG, with the exception of various salts of PHMG, as well as articles studying regimens for the relief of pathological symptoms caused by exposure to PHMG, as in the study "Anti-fibrotic effect of Pycnogenol® in a polyhexamethylene guanidine-treated mouse model"; (II) without access to full text; (III) duplicate articles; (IV) articles that examined similar routes of administration or used the same test models, or that showed similar concentrations of the same component as other studies reviewed; (V) articles investigating the effect of unhealthy habits on PHMG toxicity, such as in PHMG-induced pulmonary fibrosis; (VI) articles that mention polyhexamethine biguanidine (PHMB); (VII) studies where aquatic organisms served as a test model; (VIII) studies considering environmental aspects, particularly the use of PHMG as a means of recycling biological waste; (IX) studies related to multidrug resistance developed, including as a result of exposure to PHMG; (X) studies characterizing the molecular weight characteristics of PHMG; and (XI) studies where PHMG was used as a plant treatment.
Search Strategy
A search was performed on January 20, 2024, in PubMed, ScienceDirect, CENTRAL, and CyberLeninka and updated on August 10, 2024. The search included 7 Mesh (Medical Subject Heading): (polyhexamethylene guanidine), (PHMG), (safety profile), (human), (adverse event), (side effect), and (toxicity) joined by the Boolean operators "OR" and "AND". Following the inclusion and exclusion criteria, using the bibliographic manager Zotero, the titles and abstracts of 250 articles were analyzed. When the information in the abstract was not sufficient, we proceeded to read the full text.

For data analysis, the year of publication, authors, type of study, country, and journal were recorded. For the synthesis of the methodology of the studies, a summary table was made with the following data: availability of open access literature source, the main purpose of the study, subjects used/participated in the study, their type (human cell lines and animals), number (animals, patients, and healthy volunteers).
The main purpose of this study is to provide a comprehensive overview of the toxicity of PHMG. The information presented aims to clarify in which forms PHMG is strictly prohibited for use and to identify potential areas of its application. Based on in vitro studies, the authors sought to determine the subcytotoxic doses of various chemical modifications of PHMG for different System Organ Classes (lungs, skin, eyes, oral cavity, liver), primarily using human cell lines. The scope of PHMG applications has been significantly expanded through the screening of animal study data, including acute and subchronic toxicity, as well as studies on cardiac toxicity, hematotoxicity, nephrotoxicity and reproductive toxicity. This provides a more complete picture of the possible side effects associated with the use of PHMG in humans. At the end of the study, data regarding the effects of PHMG on healthy volunteers and patients, including those who died as a result of exposure to the compound, are also presented.
Quality and Risk of Bias Assessments
The risk of bias in the included studies was analyzed according to the Systematic Review Centre for Laboratory animal Experimentation (SYRCLE) risk of bias tool for animal studies. According to this guide, the answer "Yes" indicates a low risk of bias, while the answer "No" indicates a high risk of bias. The answer "Unclear" indicates an unclear risk of bias. If any of the relevant signaling questions were answered "No," it indicated a high risk of bias for that particular entry (25).
Result and Discussion
Study Selection and Flow Diagram
The search produced a total of 250 articles related to the toxicity and safety profile of PHMG. One hundred twenty-six were found on PubMed, 48 from ScienceDirect, 57 in CENTRAL, and 19 in CyberLeninka. One hundred fifty-two records were irrelevant to the review topic and were subsequently discarded. With the bibliographic manager Zotero, 22 duplicated records were discarded, leaving 76 reports. On reading the title and abstract of the resulting articles, 27 were excluded, as they did not meet the inclusion criteria (see Figure 1). The remaining 49 articles were read in full text. Of these, 11 were involved in vitro studies, 28 involved in vivo studies, and the remaining 10 were involved in patient studies. These studies include 11 articles focused exclusively on in vitro experiments with human cell lines exposed to PHMG and 28 articles involving animal experiments. The remaining 10 articles were conducted on patients and healthy volunteers, including medical records data for cases where subjects died from PHMG exposure.
| Study | Subject | Outcome parameters | Results |
|---|---|---|---|
| Pulmonary toxicity | |||
| Wei et al (2021) (26) | A549 | Determination of cytotoxicity of PHMG (0–32 μg/ml) and study of the expression of IL-81 and IL-6. | PHMG (≤ 2 μg/ml) had little effect on cell proliferation, cytotoxicity was observed at > 4 μg/ml at 6 and 24 h, and IL-8 and IL-6 expression was also increased. |
| Lee et al (2022) (27) | HPAEpiC | Long-term exposure concentration of PHMG-P that is not surface reactive at 24, 48, and 72 h and determination of the carcinogenic effect of PHMG-P. | CV using 2 μg/ml PHMG-P after 72 h was ~90%. Exposure to 1 μg/ml PHMG-P for 10 days caused changes in 2 protein-coding genes and 5 non-coding genes and for 27–35 days - 24 protein-coding and 5 non-coding genes. |
| Choi et al (2022) (28) | BEAS-2BA549 | Effect of PHMG-P (2.5 μg/ml, 1 h) on SG2 formation under stress conditions. | PHMG-P under stress and infection conditions increased the formation of SG, induced the expression of fibrotic genes, and caused cell death due to DNA3 damage. |
| Jin et al (2020) (29) | BEAS-2B | Determination of CV4 under conditions of exposure to PHMG-P (1–16 μg/ml for 4–24 h). | Exposure to > 4 μg/mL PHMG-P reduced CV by > 50% at 24 h and also increased LDH5 release by 3-fold, promoting epithelial barrier breakdown. |
| Jung et al (2014) (30) | A549IMR-90BEAS-2B | Determination of the cytotoxic effect of PHMG and its mediated changes in gene expression in A549 cells. | PHMG (5 μg/ml) reduced the viability of IMR-90 and BEAS-2B cells by more than 90% after 24 h, while the viability of A549 cells exceeded 60%. |
| Song et al (2019) (31) | A549MRC-5THP-1 | Determine the toxic effects of PHMG-P by assessing cell survival rates as a function of dose (0.25–20 μg/mL) and time (24, 48, and 72 h). | Exposure of A549 and MRC-5 to PHMG-P (5 μg/ml) reduced CV by 44.7% and 64.9% at 24 h, by 19% and 25.8% at 48 h, and by 11.2% and 12.7% at 72 h, respectively, while THP-1 was 86.9% at 24 h, 73.8% at 48 h, and 69.4% at 72 h in THP-1 cells. |
| Hepatic toxicity | |||
| Kim et al (2013) (32) | THP-1 | Studying the hepatotoxicity of PHMG-P by assessing the number of SA-β-gal-positive cells6. | By the 13th passage, cells treated with PHMG (0.03%) showed an 8-fold increase in the number of SA-β-gal-positive cells compared to the control, indicating the effect of acute aging of liver tissue. |
| Kim et al (2019) (33) | HepG2 | Study of hepatotoxicity under the influence of PHMG-P for 24, 48, and 72 h based on CV. | The IC507 values after 24, 48, and 72 h of incubation with PHMG-P in HepG2 cells were 7.612, 5.822, and 5.840 μg/mL, respectively. |
| Skin toxicity | |||
| Yang et al (2021) (34) | THP-1 | Skin sensitization potential of PHMG by MIT8 assessment by h-CLAT9. | The MIT for PHMG was 0.87 μg/mL, a strong sensitizer. |
| Oral toxicity | |||
| Vitt et al (2017) (35) | HGF10 | Cytotoxic effect of PHMG-P (0.005% and 0.00009%) by measuring the number of vital cells at a clinically relevant exposure time (1-30 min) and for 24 h. Determination of the immunomodulatory effect of PHMG-P by assessing the levels of PGE211, IL-6, IL-8, and MMP-112. | Exposure to PHMG-P (0.005%) for 1–30 min resulted in loss of CV within 5 min. Exposure to PHMG-P (0.00009%) for 24 h resulted in loss of fibroblast viability. The addition of PHMG-P together with IL-1b significantly reduced PGE2 levels (p< 0.001) as well as the production of IL-6, IL-8, and MMP-1 by fibroblasts (p< 0.05) at all concentrations tested. |
| Ocular toxicity | |||
| Park et al (2019) (36) | RHClE13 | Study the ophthalmic toxicity of PHMG by tissue viability assay. | Aqueous solutions of PHMG (≤0.13%) had no irritant effect. |
| Note: 1IL – interleukin; 2SG – stress granule; 3DNA - deoxyribonucleic acid; 4CV – cell viability; 5LDH - lactate dehydrogenase; 6SA-β-gal - senescence-associated β-galactosidase; 7IC50 - half maximal inhibitory concentration; 8MIT - minimal induction threshold; 9h-CLAT - human cell line activation test; 10HGF - human gingival fibroblasts; 11PGE2 - prostaglandin E2; 12MMP-1 - matrix metalloproteinase-1; 13RHClE - reconstructed human cornea-like epithelium. | |||
| Study | Subject | Outcome parameters | Results |
|---|---|---|---|
| Acute / Subchronic toxicity | |||
| Kovalenko et al. (2011) (37) | M14 | Acute toxicity study of PHMG-H (0.05-0.1%) intragastrically. | The detected LD5021 of PHMG-H was 1434 mg/kg. The substance was low toxic. |
| Kim, H. R. et al. (2015) (38) | ME15RAW264.7 | To study the inflammatory effect induced by PHMG-P at concentrations from 0.14 mg/ml to 35.10 mg/ml for 6 and 24 h. | PHMG-P caused dose-dependent cytotoxicity LC5022 of 11.15-0.99 mg/ml at 6 and 24 h, respectively. PHMG-P induced pro-inflammatory cytokines including IL-1β, IL-6 and IL-8. |
| Asiedu-Gyekye, I. J. et al. (2014) (39) | RSD16 | Study of the possible effects of subchronic toxicity of PHMG-H administered intragastrically. | The detected LD50 of PHMG-H was 600 mg/kg. |
| Dias, F. G. G. et al. (2021) (40) | RW17 | Assessment of acute oral toxicity of 0.5% PHMG-H solution. | PHMG-H 5% is classified as Acute Toxicity Category 5 (LD50 > 2000–5000 mg/kg). |
| Lee, Y. H. et al. (2020) (41) | RF34418 | Assess subacute inhalation toxicity of PHMG-H (1, 5, or 25 mg/m3 over 6 h per day, 5 days per week for two weeks). | NOAEL23 PHMG-H < 1 mg/m3. |
| Skin toxicity | |||
| Kovalenko et al. (2011) (37) | GP19 | Study of the sensitizing properties of PHMG-H (1% and 3%) when applied to the skin twice a day. | The drug did not have a cumulative, sensitizing or irritating effect at the indicated concentrations. |
| Dias, F. G. G. et al. (2021) (40) | RW, M | Evaluation of possible side effects of cutaneous PHMG-H 0.5%. | PHMG-H reduced the area of skin lesions and increased the number of fibroblasts, with no side effects. At a concentration of 5%, PHMG-H exhibited neither genotoxicity nor cytotoxicity at doses up to 1500 mg/kg by micronucleus assay. |
| Oral toxicity | |||
| Sklyanova et al. (2006) (42) | R20 | To determine the toxicity of PHMG-P injection (0.25%) in a histomorphological study when exposed to cheek tissue. | PHMG-P (0.25%) caused aseptic inflammation at the injection site. The least irritating effect on cheek tissue and subcutaneous connective tissue was produced by 1.0 ml of injection. |
| Pulmonary toxicity | |||
| Jeong S. H. et al (2024) (43) | R | To study the severity of lung injury resulting from intratracheal instillation of PHMG-P (0.2, 1.0, and 5.0 mg/kg). | The severity of lung damage, as well as the number, size and malignancy of tumors, increased as the dose of PHMG-P was increased. Bronchiolar-alveolar hyperplasia developed in all groups. |
| Song, J. et al. (2022) (44) | M | Study of the lung injury process of PHMG-P when administered intravenously (0.9 or 7.2 mg/kg) or intratracheally (0.9 mg/kg). | PHMG-P promoted the production of pro-inflammatory cytokines and also caused fibrotic changes in the lungs when administered intratracheally (0.9 mg/kg). |
| Li, X. Et al. (2021) (45) | M C57BL/6J | Study of the mechanism of PHMG-induced (0.05, 0.1, 1, and 2 mg/ml) pulmonary fibrosis based on increased surface tension mediated by pulmonary surfactant inhibition. | PHMG-H induced pulmonary fibrosis along with increased surface tension. |
| Lee, J. D. et al (2020) (46) | RW | Determination of pulmonary toxicity caused by intratracheal PHMG-P (single dose 1.5 mg/kg or 0.1 mg/kg, 2 times a week, for 4 weeks). | Upon single administration, PHMG-P induced alveolar macrophage aggregation and granulomatous inflammation. Pulmonary fibrosis, chronic inflammation, bronchiole-alveolar fibrosis, and squamous cell metaplasia were observed in the repeated administration group. |
| Lee, Y. H. et al. (2019) (47) | Males RF344 | Assessment of oxidative stress in the lungs induced by inhaled exposure to PHMG-H (0.13, 0.40 or 1.20 mg/m3 6 h per day, 5 days per week for 13 weeks). | The number of oxidative stress markers in the bronchial epithelium of rats increased in a dose-dependent manner. At 1.20 mg/m3 an increase in respiratory rate and a decrease in body weight were observed. At 0.13 mg/m3, no deviations in the lung structure were observed. |
| Lee, Y. H. et al. (2020) (41) | RF344 | Subacute inhalation toxicity study of PHMG-H (1, 5, or 25 mg/m3 for 6 h per day, 5 days per week for 2 weeks). | The severity of lung damage increased in a dose-dependent manner. Exposure to PHMG-H (5 and 25 mg/m3) caused squamous metaplasia of the bronchial and bronchiolar epithelium, as well as alveolar emphysema and necrosis with inflammation. Lymphoid hyperplasia of broncho-associated lymphoid tissue was observed in rats exposed to 1, 5, and 25 mg/m3. Alveolar macrophage aggregation was observed in male rats exposed to 0, 1, 5, and 25 mg/m3 and female rats exposed to 1 and 25 mg/m3. |
| Park, S. et al. (2014) (48) | RSD | Assessment of lung injury resulting from nasal inhalation of PHMG-P (1.6 mg/m3) aerosol 6 h per day, 5 days per week, for 4 weeks. | PHMG-P (1.6 mg/m3) caused typical bronchiolocentric destruction with inflammation and fibrosis. |
| Cardiac toxicity | |||
| Choi, J. H. et al. (2024) (49) | RSD | Study of platelet procoagulant activity induced by PHMG-P (2.5 μg/ml) | PHMG-P causes procoagulant platelet activation, which may contribute to prothrombotic risk and cardiovascular disease. |
| Ocular toxicity | |||
| Lee, H. , et al. (2021) (50) | Rabbit cornea cells | Study of the adverse ocular effects of PHMG-H (1, 5, 10 and 25 μg/ml for 24, 48, 72 and 96 h). | PHMG-H can cause fibrosis. IC5024 of PHMG-H at 24, 48, 72 and 96 h: 20.8 µg/ml, 13.8 µg/ml, 8.5 µg/ml and 6.2 µg/ml, respectively. PHMG-H induced cyclooxygenase-2 at 25 μg/ml and hemeoxygenase-1 at all concentrations. |
| Hepatic toxicity | |||
| Choi, Y. J. et al (2022) (51) | M | Study of the effect of PHMG (0, 60 and 200 μg/kg) on the pathophysiology and metabolism of the liver when administered intratracheally 3 times a week, 12 times in total. | PHMG significantly reduced liver cholesterol levels. mRNA-seq25 analysis revealed changes in the expression of genes associated with cholesterol biosynthesis and metabolism to bile acids. |
| Asiedu-Gyekye, I. J. et al. (2014) (39) | RSD | To study the possible subchronic toxicity effects of PHMG-H (0.006 mg/kg, 0.012 mg/kg, and 0.036 mg/kg) administered intragastrically. | In 10% of animals at all doses, local areas of mild pericentral degeneration of hepatocytes were observed in the liver tissue. |
| Kim, M. et al (2022) (52) | M C57/BL6 male | To study the pathophysiology of liver fibrosis induced by intraperitoneal administration of PHMG-P (0.03% and 0.1%) twice a week for 5 weeks. | Diffuse fibrotic lesions of the liver were revealed without affecting the lungs. PHMG-P induces liver fibrosis in the pericentral, periportal, and capsular regions. |
| Dias, F. G. G. et al. (2021) (40) | RW | Investigate the potential for liver injury when 0.5% PHMG-H topical solution is administered orally by gavage. | Alanine aminotransferase/aspartate aminotransferase and urea/creatinine did not differ significantly from the control group. |
| Hematological toxicity | |||
| Sung HJ et al. (2022) (53) | RSD males | Study of possible disorders of hematopoietic function 20 weeks after intratracheal instillation of PHMG-P (1-5 mg/kg). | PHMG-P affects hematopoiesis involved in monocyte differentiation and platelet production. |
| Asiedu-Gyekye, I. J. et al. (2014) (39) | RSD (males and females) | Study of hematological parameters during intragastric administration of PHMG-H (0.006 mg/kg, 0.012 mg/kg, and 0.036 mg/kg). | PHMG-H did not have any harmful effects on the hematopoietic system of animals. |
| Lee, J. et al. (2021) (54) | Females RSD | Study of hematological parameters in pregnant female rats by inhalation of PHMG-P aerosol (0.14, 1.60, and 3.20 mg/m3). | PHMG-P (3.20 mg/m3) increased the total number of red blood cells, hematocrit, hemoglobin and neutrophils, decreased the number of reticulocytes, lymphocytes, eosinophils, basophils, decreased alanine aminotransferase, alkaline phosphatase, total bilirubin, total cholesterol, sodium, phospholipids, and chloride. PHMG-P (1.60 mg/m3) also increased the total number of red blood cells, hematocrit, and hemoglobin, and a decrease in alkaline phosphatase and chloride was observed. |
| Lee, Y. H. et al. (2020) (41) | RF344 (males and females) | Study of hematological parameters as part of a study of subacute toxicity of PHMG-H in the form of an aerosol (1 mg/m3, 5 mg/m3, or 25 mg/m3 for 6 h a day, 5 days a week for two weeks). | PHMG-H (1, 5, and 25 mg/m3) increased the total number of red blood cells, hematocrit, and hemoglobin. Hemoglobin increased significantly at 25 mg/m3. Reticulocytes were significantly reduced at 5 and 25 mg/m3. Platelets decreased at 25 mg/m3. Monocytes and neutrophils increased at 25 mg/m3. Lymphocytes were reduced in males at 25 mg/m3, and in females at 5 and 25 mg/m3. Eosinophils were significantly reduced in females at 25 mg/m3. Alanine aminotransferase increased in males at 25 mg/m3, and in females at 5 and 25 mg/m3. Aspartate aminotransferase increased significantly in all at 25 mg/m3. |
| Reproductive toxicity | |||
| Lee, J. et al. (2022) (55) | R | To study the postnatal development of offspring after exposure to PHMG-P (0, 0.14, 1.60, and 3.20 mg/m3). | PHMG-P (1.60 and 3.20 mg/m3) increased perinatal mortality and decreased the viability index; F1 offspring had lower birth weight. Pregnant rats had severe systemic toxicity and a prolonged gestation period. |
| Lee, J. et al (2019) (56) | RSD | To study the toxic effects of orally administered PHMG-P (0, 13, 40, and 120 mg/kg) | PHMG-P (120 mg/kg) exhibited toxicity including depressed behavior, thinness, decreased body weight, decreased food intake, and decreased body weight of F1 offspring. NOAEL was 40 mg/kg/day. |
| Nephrotoxicity | |||
| Asiedu-Gyekye, I. J. et al. (2014) (39) | RSD | To study the possible subchronic toxicity effects of PHMG-H (0.006 mg/kg, 0.012 mg/kg, and 0.036 mg/kg) administered intragastrically. | At 0.006 mg/kg and 0.036 mg/kg, 20% of animals showed mild degrees of hydropic changes in the proximal tubules. |
| Note: 14M – mice; 15ME - Mice macrophage; 16RSD – Rats Sprague-Dawley; 17RW – Rats Wistar; 18RF344 – Rats F344; 19GP - guinea pig; 20R – Rats; 21LD50 - median lethal dose; 22LC50 – median lethal concentration; 23NOAEL - No-observed-adverse-effect level; 24IC50 - half maximal inhibitory concentration; 25mRNA-seq - messenger ribonucleic acid sequencing. | |||
The majority of articles (67.3%) were published within the last 5 years, indicating a growing interest and research focus on the topic. The peak in publication volume occurred in 2021, with 10 studies published, followed by 2019, which saw the release of 8 studies. Geographically, South Korea emerged as the leading contributor to the literature, likely due to a mass poisoning event involving HD, which spurred significant research and analysis in the country.
Quality Evaluation
Results of Toxicity Assessment of PHMG in In Vitro studies using Human Cell Lines as Models
The toxicity results of PHMG from in vitro studies are presented exclusively using human cell models. These results are summarized in Table 1, which provides basic information about the studies conducted.
| Study | Subject | Outcome parameters | Results |
|---|---|---|---|
| Pulmonary toxicity | |||
| Ryu et al (2019) (57) | Patients with lung damage (n=453), control group (n=700). | Assessment of inhalation exposure to HD26. | In 259 (57.2%) patients with ILD27, it developed within one year of HD use, where the average level of inhaled PHMG was 145.1 µg/m3. |
| Lamichhane et al (2019) (58) | Patients with IIP (n=244), control group (n=244). | Frequency distribution of characteristics associated with HD exposure. | The use of HD-containing PHMG was associated with a higher risk of lung damage compared to those who used other disinfectants. |
| Lee et al (2021) (59) | Patients (n=362). | Study of asthma induced by PHMG-based HD exposure using data from a panel study of South Korean children. | Asthma associated with PHMG exposure was characterized by decreased lung function, less positive bronchial hyperresponsiveness scores, and different plasma protein distribution. |
| Park et al (2014) (60) | Patients with lung damage (n=38). | Assessing PHMG-based HD exposure through medical record review. | Three pregnant women and six preschool-aged children died as a result of lung damage. Another six pregnant women and 22 preschool-aged children experienced non-lethal lung injury. Three adult male office workers did not suffer fatal lung damage. |
| Ju et al (2021) (61) | Patients with fatal injuries (n=1413) | Analysis of the distribution of causes of death among applicants according to medical records. | 43.0% of the affected individuals had more than one case of ILD identified. Among the causes of mortality in the deceased, respiratory organ diseases accounted for 54.4%. Among those who died from respiratory diseases, ILD were the most common cause of death (65.5%). |
| Hepatic toxicity | |||
| Kim et al (2023) (62) | Patients (n=3) who used HD. | To examine the likelihood of developing toxic hepatitis due to the inhalation of HD-containing PHMG by analyzing patients' medical records. | Patients exhibited an increase in aspartate aminotransferase and alanine aminotransferase levels up to 2000-4000 IU/L. One fatality was recorded, while two patients were discharged after treatment. Hepatotoxicity due to inhalation was not confirmed. |
| Makarov et al (2009) (63) | Blood serum of patients (n=40) with toxic hepatitis and healthy volunteers (n=50). | Determining the impact of consuming alcohol surrogates containing PHMG-H on the lipid profile of blood. | Patients differed from healthy volunteers by a twofold decrease in the relative content of total phospholipids, free fatty acids, cholesterol esters, and phosphatidylcholine, but had higher levels of free cholesterol and total lysophospholipids. The overall lipid levels in patients were three times higher than the norm. The development of toxic hepatitis has been confirmed. |
| Ostapenko et al (2011) (22) | Patients with poisoning from surrogate alcohol (n=579). | Study of the clinical presentation and outcomes of poisoning with surrogate alcohol containing PHMG-H. | Jaundice was observed in 99.7%, with detected foci of cholestasis, fibrosis progressing to cirrhosis and inflammatory infiltration. The development of cholestatic hepatitis has been confirmed. |
| Skin Toxicity | |||
| Pummi et al (2012) (64) | Patient with skin sensitization (n=1) and previously observed dermatitis. | Assessing the risk of skin sensitization from a PHMG-containing antiseptic. | Sensitization developed within 1-2 months with frequent use of a non-alcoholic disinfectant containing PHMG at concentrations of 0.1-1%. |
| Oral Toxicity | |||
| Vitt et al (2019) (65) | Patients with severe chronic periodontitis (n=19). | Evaluation of side effects of irrigation with antiseptic PHMG-P (1%) in periodontal treatment. | No adverse effects were observed with PHMG-P (1%) during the study. |
| Note: 26HD - humidifier disinfectants; 27ILD - interstitial lung diseases. | |||
Results of Toxicity Assessment of PHMG in Trials Involving Animals
The toxicity results of PHMG from animal studies are summarized in Table 2, which provides basic information about the studies conducted.
Results of Toxicity Assessment of PHMG in Trials Involving Human
The results of PHMG toxicity studies in trials involving people are summarized in Table 3, which provides basic information about the studies conducted.
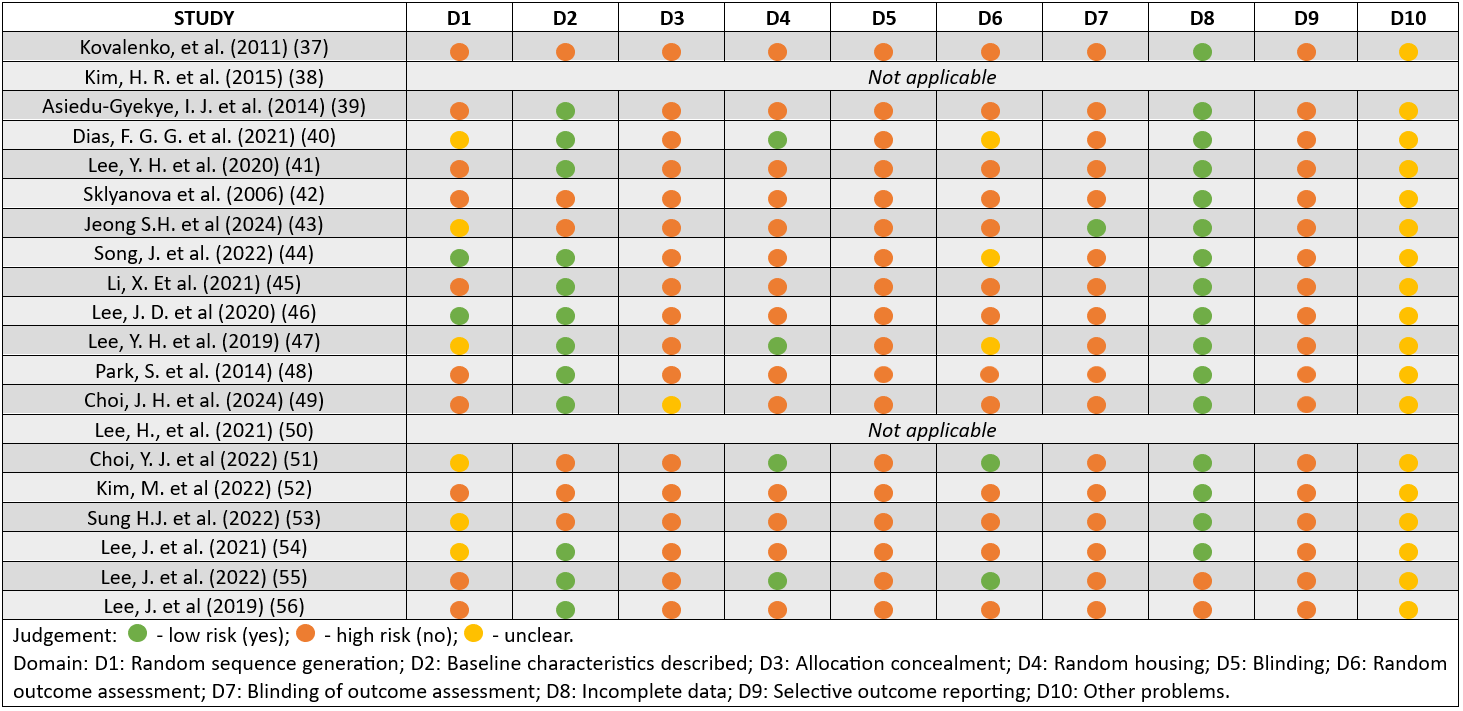
SYRCLE's Risk of Bias Tool
The results of the attribution of bias based on each domain of SYRCLE's tool are shown in Figure 2.
Notably, all studies failed to clearly define whether the animals were randomly housed during the experiment if proper blinding of the caregivers/investigators concerning which intervention each animal received during the experiment was performed, or if there was a random selection of the animals for outcome assessment.
Conclusion
In conclusion, the systematic review indicates that inhalation exposure to PHMG poses significant health risks, including lung fibrosis and malignant tumors, making its use in aerosol medications unacceptable. Additionally, PHMG negatively impacts liver function, heart activity (inducing procoagulant platelet activation), kidneys (causing mild hydropic changes in proximal tubules), and hematopoietic function, leading to changes in erythrocyte counts, hematocrit, and hemoglobin levels. Although one animal study showed that a 0.5% aqueous solution of PHMG-H did not significantly alter liver enzyme levels or kidney function, the overall evidence suggests caution.
The clinical implications of these findings are significant. Given the severe respiratory consequences, PHMG should not be used in any form that could lead to inhalation exposure, especially in aerosolized medications. For dermal applications, PHMG appears safe at concentrations up to 3%, though practical use might limit this to 1% due to potential discomfort. Clinicians should be aware of the possibility of sensitization, particularly in patients with pre-existing skin conditions. The use of PHMG in wound care, particularly in combination with chitosan, could offer benefits, but close monitoring for allergic reactions is advisable. In oral health, 1% PHMG-P has been used safely in periodontal treatment, suggesting its potential in dental care. For ophthalmic use, concentrations should be carefully controlled. Solutions of PHMG-P below 0.13% appear safe for human corneal epithelium, however, lower concentrations still pose a risk of corneal fibrosis, as shown in animal studies. Clinicians should prefer lower concentrations and consider alternatives or formulations with mitigated toxicity for sensitive applications like eye drops.
Overall, while PHMG and its derivatives show promise in various medical applications, their use must be reasonable, with careful consideration of the associated risks. The potential benefits in a post-antibiotic era are significant, but only with strict adherence to safety protocols can PHMG be effectively integrated into clinical practice.
Abbreviations
PHMG=polyhexamethylene guanidine; HD=humidifier disinfectants; ILD=interstitial lung diseases; ppm=parts per million; PHMG-H=polyhexamethylene guanidine hydrochloride; PHMG-P=polyhexamethylene guanidine phosphate; PHMB=polyhexamethine biguanidine; IL=interleukin; SG=stress granule; DNA=deoxyribonucleic acid; CV=cell viability; LDH=lactate dehydrogenase; SA-β-gal=senescence-associated β-galactosidase; IC50=half maximal inhibitory concentration; MIT=minimal induction threshold; h-CLAT=human cell line activation test; HGF=human gingival fibroblasts; PGE2=prostaglandin E2; MMP-1=matrix metalloproteinase-1; RHClE=reconstructed human cornea-like epithelium; M=mice; ME=mice macrophage; RSD=rats Sprague-Dawley; RW=rats Wistar; RF344=rats F344; GP=guinea pig; R=rats; LD50=median lethal dose; LC50=median lethal concentration; NOAEL=no-observed-adverse-effect level; IC50=half maximal inhibitory concentration; mRNA-seq=messenger ribonucleic acid sequencing.
Declarations
Conflict of Interest
The authors report there are no competing interests to declare.
Data Availability
Not applicable.
Ethics Statement
Not applicable.
Funding Information
Not applicable.
References
- Boyd NK, Teng C, Frei CR. Brief Overview of Approaches and Challenges in New Antibiotic Development: A Focus On Drug Repurposing. Front Cell Infect Microbiol. 2021; 11: 684515. https://doi. org/10.3389/fcimb.2021.684515
- Jackson N, Czaplewski L, Piddock LJV. Discovery and development of new antibacterial drugs: learning from experience? J Antimicrob Chemother. 2018; 73 (6): 1452-1459. https://doi. org/10.1093/jac/dky019. PMID: 29438542
- Jenull S, Laggner H, Hassl I, Velimirov B, Huettinger M, Zemann N. Cooperativity between antibiotics and antiseptics: testing the bactericidal effect. J Wound Care. 2017; 26 (12): 720-726. https://doi. org/10.12968/jowc.2017.26.12.720
- Ferrara M, Gatti F, Lockington D, Iaria A, Kaye S, Virgili G, et al. Antimicrobials and antiseptics: Lowering effect on ocular surface bacterial flora – A systematic review. Acta ophthalmol. 2023; 102 (3). https://doi. org/10.1111/aos.15732
- Mencucci R, Favuzza E, Bottino P, Mazzantini C, Zanotto E, Pellegrini-Giampietro DE, et al. A new ophthalmic formulation containing antiseptics and dexpanthenol: In vitro antimicrobial activity and effects on corneal and conjunctival epithelial cells. Exp Eye Res. 2020; 201: 108269. https://doi. org/10.1016/j. exer.2020.108269
- Nur Kertmen Kurtar S, Kertmen M, Kose M. Design of a new porphyrin-based compound and investigation of its photosensitive properties for antibacterial photodynamic therapy. Spectrochim Acta A Mol Biomol Spectrosc. 2024; 319: 124529. https://doi. org/10.1016/j. saa.2024.124529
- Moshynets OV, Baranovskyi TP, Iungin OS, Kysil NP, Metelytsia LO, Pokholenko I, et al. eDNA Inactivation and Biofilm Inhibition by the PolymericBiocide Polyhexamethylene Guanidine Hydrochloride (PHMG-Cl). Int J Mol Sci. 2022; 23 (2): 731. https://doi. org/10.3390/ijms23020731
- Vitt A, Sofrata A, Slizen V, Sugars RV, Gustafsson A, Gudkova EI, et al. Antimicrobial activity of polyhexamethylene guanidine phosphate in comparison to chlorhexidine using the quantitative suspension method. Ann Clin Microbiol Antimicrob. 2015; 14 (36). https://doi. org/10.1186/s12941-015-0097-x
- Guseva MN, Doronin MI, Shevchenko MA, Mikhalishin DV, Gochmuradov YM, Mikhalishin VV, et al. Determination of indicators for tests of polysept (polyhexamethylene guanidine hydrochloride) for flocculation properties. Veterinary Science Today. 2022; 11 (3): 254-261. https://doi. org/10.29326/2304-196X-2022-11-3-254-261
- Dias FGG, Pereira LF, Parreira RLT, Veneziani RCS, Bianchi TC, Fontes VFNP, et al. Evaluation of the antiseptic and wound healing potential of polyhexamethylene guanidine hydrochloride as well as its toxic effects. Eur J Pharm Sci. 2021; 160: 105739. https://doi. org/10.1016/j. ejps.2021.105739
- Mathurin YK, Koffi-Nevry R, Gué hi ST, Tano K, Oulé MK. Antimicrobial activities of polyhexamethylene guanidine hydrochloride-based disinfectant against fungi isolated from cocoa beans and reference strains of bacteria. J Food Prot. 2012; 75 (6): 1167-71. https://doi. org/10.4315/0362-028X. JFP-11-361
- Wang J, Yu Y, Dong Y. Disinfection of Ready-to-Eat Lettuce Using Polyhexamethylene Guanidine Hydrochloride. Microorganisms. 2020; 8 (2): 272. https://doi. org/10.3390/microorganisms8020272
- Oulé MK, Quinn K, Dickman M, Bernier AM, Rondeau S, De Moissac D, et al. Akwaton, polyhexamethylene-guanidine hydrochloride-based sporicidal disinfectant: a novel tool to fight bacterial spores and nosocomial infections. J Med Microbiol. 2012; 61 (Pt 10): 1421-1427. https://doi. org/10.1099/jmm.0.047514-0
- Aviv O, Amir N, Laout N, Ratner S, Basu A, Domb AJ. Poly (hexamethylene guanidine)-poly (ethylene glycol) solid blend for water microbial deactivation. Polymer Degradation and Stability, 2016; 129: 239– 245. https://doi. org/10.1016/j. polymdegradstab.2016.04.020
- Liu N, Ren P, Saleem A, Feng W, Huo J, Ma H, et al. Simultaneous Efficient Decontamination of Bacteria and Heavy Metals via Capacitive Deionization Using Polydopamine/Polyhexamethylene Guanidine Co-deposited Activated Carbon Electrodes. ACS Appl Mater Interfaces. 2021; 29; 13 (51): 61669-61680. https://doi. org/10.1021/acsami.1c20145
- Mogilevskaya IV Antimicrobial effect of preparations based on polyhexamethyleneguanidine hydrochloride on Calligonum Aphyllum (pall. ) gurke in vitro // Scientific Review. Biol. Sci. 2023 [cited 2024 Aug 9]; 4: 11-16. https://doi. org/10.17513/srbs.1336 Available from: https://science-biology. ru/en/article/view? id=1336 Russian
- Vointseva II, Gembitsky PA. Polyguanidines - disinfectants and polyfunctional additives in composite materials. Russian
- Kha KA, Grammatikova NÉ, Vasilinko IA, Kedik SA. Comparative in vitro antibacterial activity of polyhexamethylene guanidine hydrochloride and polyhexamethylene guanidine succinate. Antibiot Khimioter. 2013; 58 (1-2): 3-7. Russian
- Lee E, Lee SY, Hong SJ. The past, present, and future of humidifier disinfectant-associated interstitial lung diseases in children. Clin Exp Pediatr. 2020; 63 (7): 251-258. https://doi. org/10.3345/cep.2019.01326
- Park DU, Park J, Yang KW, Park JH, Kwon JH, Oh HB. Properties of Polyhexamethylene Guanidine (PHMG) Associated with Fatal Lung Injury in Korea. Molecules. 2020; 25 (14): 3301. https://doi. org/10.3390/molecules25143301
- Park DU, Yang KW, Kim J, Park JH, Lee SY, Zoh KE, et al. Characteristics of the Molecular Weight of Polyhexamethylene Guanidine (PHMG) Used as a Household Humidifier Disinfectant. Molecules. 2021; 26 (15): 4490. https://doi. org/10.3390/molecules26154490
- Ostapenko YN, Brusin KM, Zobnin YV, Shchupak AY, Vishnevetskiy MK, Sentsov VG, et al. Acute cholestatic liver injury caused by polyhexamethyleneguanidine hydrochloride admixed to ethyl alcohol. Clin Toxicol (Phila). 2011; 49 (6): 471-7. https://doi. org/10.3109/15563650.2011.592837 Russian
- Kozoor OI, Nosikova LA, Shestakov KA, Kochetov AN. The determination of polymeric derivatives of guanidine in disinfectants by two-phase titration. Fine Chemical Technologies. 2015; 10 (2): 20-24. Russian
- Commission decision of 9 February 2012 concerning the non-inclusion of certain substances in Annex I, IA or IB to Directive 98/8/EC of the European Parliament and of the Council concerning the placing of biocidal products on the market (L 38/48 of 11.02.2012) [cited Jan 24, 2024]. Available from: https://eur-lex. europa. eu/LexUriServ/LexUriServ. do? uri=OJ: L: 2012: 038: 0048: 0050: EN: PDF
- Hooijmans CR, Rovers MM, de Vries RB, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE's risk of bias tool for animal studies. BMC Med Res Methodol. 2014; 14: 43. https://doi. org/10.1186/1471-2288-14-43
- Wei L, Guo Y, Ding X, Guo C, Ge N, Ren D, et al. The liver X receptor agonist T0901317 reduces the inflammation of alveolar epithelial cells induced by polyhexamethylene guanidine through inhibition of the NFκ B signaling pathway. Ann Transl Med. 2021; 9 (24): 1795. https://doi. org/10.21037/atm-21-6501
- Lee H, Jeong SH, Lee H, Kim C, Nam YJ, Kang JY, et al. Analysis of lung cancer-related genetic changes in long-term and low-dose polyhexamethylene guanidine phosphate (PHMG-p) treated human pulmonary alveolar epithelial cells. BMC Pharmacol Toxicol. 2022; 23 (1): 19. https://doi. org/10.1186/s40360-022-00559-5
- Choi S, Choi S, Choi Y, Cho N, Kim SY, Lee CH, et al. Polyhexamethylene guanidine phosphate increases stress granule formation in human 3D lung organoids under respiratory syncytial virus infection. Ecotoxicol Environ Saf. 2022; 229: 113094. https://doi. org/10.1016/j. ecoenv.2021.113094
- Jin SW, Lee GH, Pham HT, Choi JH, Jeong HG. Polyhexamethylene Guanidine Phosphate Damages Tight Junctions and the F-Actin Architecture by Activating Calpain-1 via the P2RX7/Ca2+ Signaling Pathway. Cells. 2019; 9 (1): 59. https://doi. org/10.3390/cells9010059
- Jung HN, Zerin T, Podder B, Song HY, Kim YS. Cytotoxicity and gene expression profiling of polyhexamethylene guanidine hydrochloride in human alveolar A549 cells. Toxicol In Vitro. 2014; 28 (4): 684-92. https://doi. org/10.1016/j. tiv.2014.02.004
- Song J, Jung KJ, Yoon SJ, Lee K, Kim B. Polyhexamethyleneguanidine phosphate induces cytotoxicity through disruption of membrane integrity. Toxicology. 2019; 414: 35-44. https://doi. org/10.1016/j. tox.2019.01.001
- Kim JY, Kim HH, Cho KH. Acute cardiovascular toxicity of sterilizers, PHMG, and PGH: severe inflammation in human cells and heart failure in zebrafish. Cardiovasc Toxicol. 2013; 13 (2): 148-60. https://doi. org/10.1007/s12012-012-9193-8
- Kim SH, Kwon D, Lee S, Ki SH, Jeong HG, Hong JT, et al. Polyhexamethyleneguanidine Phosphate-Induced Cytotoxicity in Liver Cells Is Alleviated by Tauroursodeoxycholic Acid (TUDCA) via a Reduction in Endoplasmic Reticulum Stress. Cells. 2019; 8 (9): 1023. https://doi. org/10.3390/cells8091023
- Yang S, Heo Y, Gautam R, Lee J, Maharjan A, Jo J, et al. Prediction of the skin sensitization potential of polyhexamethylene guanidine and triclosan and mixtures of these compounds with the excipient propylene glycol through the human Cell Line Activation Test. Toxicol Ind Health. 2021 Jan; 37 (1): 1-8. https://doi. org/10.1177/0748233720974131
- Vitt A, Slizen V, Boströ m EA, Yucel-Lindberg T, Kats A, Sugars RV, et al. Effects of polyhexamethylene guanidine phosphate on human gingival fibroblasts. Acta Odontol Scand. 2017; 75 (7): 524-529. https://doi. org/10.1080/00016357.2017.1350993
- Park J, Lee H, Park K. Eye irritation tests of polyhexamethylene guanidine phosphate (PHMG) and chloromethylisothiazolinone/methylisothiazolinone (CMIT/MIT) using a tissue model of reconstructed human cornea-like epithelium. Environ Health Toxicol. 2019; 34 (2): e2019004. https://doi. org/10.5620/eht. e2019004
- Kovalenko VL, Chehun AI, Yarokhno YM, Hnatenko AV, Savchenko LG. Effect of a disinfectant based on polyhexamethyleneguanidine hydrochloride on the body of laboratory animals. Scientific Bulletin of S. Z. Lviv National University of Veterinary Medicine and Biotechnology Gzhitskyi. 2011 [cited 2024 May 19]; 13 (4-4 (50)): 247-251. Available from: https://cyberleninka. ru/article/n/vpliv-dezinfektantu-na-osnovi-poligeksametilenguanidin-gidrohloridu-na-organizm-laboratornih-tvarin Russian
- Kim HR, Shin DY, Chung KH. The role of NF-κ B signaling pathway in polyhexamethylene guanidine phosphate induced inflammatory response in mouse macrophage RAW264.7 cells. Toxicology letters. 2015; 233 (2): 148– 155. https://doi. org/10.1016/j. toxlet.2015.01.005
- Asiedu-Gyekye IJ, Mahmood SA, Awortwe C, Nyarko AK. A preliminary safety evaluation of polyhexamethylene guanidine hydrochloride. International journal of toxicology. 2014; 33 (6): 523– 531. https://doi. org/10.1177/1091581814553036
- Dias FGG, Pereira LF, Parreira RLT, Veneziani RCS, Bianchi TC, Fontes VFNP, et al. Evaluation of the antiseptic and wound healing potential of polyhexamethylene guanidine hydrochloride as well as its toxic effects. European journal of pharmaceutical sciences: official journal of the European Federation for Pharmaceutical Sciences. 2021; 160: 105739. https://doi. org/10.1016/j. ejps.2021.105739
- Lee YH, Seo DS. Toxicity of humidifier disinfectant polyhexamethylene guanidine hydrochloride by two-week whole body-inhalation exposure in rats. Journal of toxicologic pathology. 2020; 33 (4): 265– 277. https://doi. org/10.1293/tox.2020-0043
- Sklyanova YuA, Ushakov RV, Kazimirsky VA, Izatulin VG. Experimental substantiation of the use of fogucid (anavidin) in dentistry. Acta Biomedica Scientifica. 2006 [cited 2024 May 19]; 4: 344-346. Available from: https://cyberleninka. ru/article/n/eksperimentalnoe-obosnovanie-primeneniya-fogutsida-anavidina-v-stomatologii Russian
- Jeong SH, Lee H, Nam YJ, Kang JY, Lee H, Choi JY, et al. Longitudinal long term follow up investigation on the carcinogenic impact of polyhexamethylene guanidine phosphate in rat models. Scientific reports. 2024; 14 (1): 7178. https://doi. org/10.1038/s41598-024-57605-x
- Song J, Jung KJ, Yang MJ, Kim W, Lee BS, Choe SK, et al. Disruption of Membrane Integrity as a Molecular Initiating Event Determines the Toxicity of Polyhexamethylene Guanidine Phosphate Depending on the Routes of Exposure. International journal of molecular sciences. 2022; 23 (6): 3289. https://doi. org/10.3390/ijms23063289
- Li X, Zhang J, Du C, Jiang Y, Zhang W, Wang S, et al. Polyhexamethylene guanidine aerosol triggers pulmonary fibrosis concomitant with elevated surface tension via inhibiting pulmonary surfactant. Journal of hazardous materials. 2021: 420: 126642. https://doi. org/10.1016/j. jhazmat.2021.126642
- Lee JD, Kim HY, Kang K, Jeong HG, Song MK, Tae IH, et al. Integration of transcriptomics, proteomics and metabolomics identifies biomarkers for pulmonary injury by polyhexamethylene guanidine phosphate (PHMG-p), a humidifier disinfectant, in rats. Archives of toxicology. 2020; 94 (3): 887– 909. https://doi. org/10.1007/s00204-020-02657-x
- Lee YH, Seo DS, Lee M J, Cha HG. Immunohistochemical characterization of oxidative stress in the lungs of rats exposed to the humidifier disinfectant polyhexamethylene guanidine hydrochloride. Journal of toxicologic pathology. 2019; 32 (4): 311– 317. https://doi. org/10.1293/tox.2019-0049
- Park S, Lee K, Lee EJ, Lee SY, In KH, Kim HK, et al. Humidifier disinfectant-associated interstitial lung disease in an animal model induced by polyhexamethylene guanidine aerosol. American journal of respiratory and critical care medicine. 2014; 190 (6): 706– 708. https://doi. org/10.1164/rccm.201404-0710LE
- Choi JH, Kim K. Polyhexamethylene Guanidine Phosphate Enhanced Procoagulant Activity through Oxidative-Stress-Mediated Phosphatidylserine Exposure in Platelets. Toxics. 2024; 12 (1): 50. https://doi. org/10.3390/toxics12010050
- Lee H, Park J, Park K. Fibrosis as a result of polyhexamethylene guanide exposure in cultured Statens Seruminstitut Rabbit Cornea (SIRC) cells. Environmental analysis, health and toxicology. 2021; 36 (2): e2021009. https://doi. org/10.5620/eaht.2021009
- Choi YJ, Yang HS, Zhang Y, Lee W, Yun SH, Nam YA, et al. Intratracheal exposure to polyhexamethylene guanidine phosphate disrupts coordinate regulation of FXR-SHP-mediated cholesterol and bile acid homeostasis in mouse liver. Ecotoxicology and environmental safety. 2022; 247: 114213. https://doi. org/10.1016/j. ecoenv.2022.114213
- Kim M. , Hur S, Kim KH, Cho Y, Kim K, Kim HR, et al. A New Murine Liver Fibrosis Model Induced by Polyhexamethylene Guanidine-Phosphate. Biomolecules & therapeutics. 2022; 30 (2): 126– 136. https://doi. org/10.4062/biomolther.2021.120
- Sung HJ, Jeong SH, Kang JY, Kim C, Nam YJ, Kim JY, Choi JY, Lee HJ, Lee YS, Kim EY, Baek YW, Lee H, Lee JH. Hematotoxic Effect of Respiratory Exposure to PHMG-p and Its Integrated Genetic Analysis. Toxics. 2022 Nov 16; 10 (11): 694. doi: 10.3390/toxics10110694
- Lee J, Choi SJ, Jeong JS, Kim SY, Lee SH, Yang MJ, Lee SJ, et al. A humidifier disinfectant biocide, polyhexamethylene guanidine phosphate, inhalation exposure during pregnancy induced toxicities in rats. Journal of hazardous materials. 2021; 404 (Pt B): 124007. https://doi. org/10.1016/j. jhazmat.2020.124007
- Lee J, Choi SJ, Jeong JS, Kim SY, Lee SJ, Baek SK, et al. Adverse postnatal developmental effects in offspring from humidifier disinfectant biocide inhaled pregnant rats. Chemosphere. 2022; 286 (Pt 2): 131636. https://doi. org/10.1016/j. chemosphere.2021.131636
- Lee J, Jeong JS, Kim SY, Im WJ, Shin YJ, Lee K, et al. Reproductive and developmental toxicity screening of polyhexamethylene guanidine phosphate by oral gavage in rats. Regulatory toxicology and pharmacology: RTP. 2016; 108: 104440. https://doi. org/10.1016/j. yrtph.2019.104440
- Ryu SH, Park DU, Lee E, Park S, Lee SY, Jung S, et al. Humidifier disinfectant and use characteristics associated with lung injury in Korea. Indoor Air. 2019; 29 (5): 735-747. https://doi. org/10.1111/ina.12585
- Lamichhane DK, Leem JH, Lee SM, Yang HJ, Kim J, Lee JH, et al. Family-based case-control study of exposure to household humidifier disinfectants and risk of idiopathic interstitial pneumonia. PLoS One. 2019; 14 (9): e0221322. https://doi. org/10.1371/journal. pone.0221322
- Lee SY, Ahn HS, Kim EM, Kim KK, Kang MJ, Park MJ, et al. Different Characteristics of Childhood Asthma Related to Polyhexamethylene Guanidine Exposure. Ann Am Thorac Soc. 2021; 18 (9): 1523-1532. https://doi. org/10.1513/AnnalsATS.202007-807OC
- Park D, Leem J, Lee K, Lim H, Choi Y, Ahn JJ, et al. Exposure characteristics of familial cases of lung injury associated with the use of humidifier disinfectants. Environ Health. 2014; 13: 70. https://doi. org/10.1186/1476-069X-13-70
- Ju YJ, Lee S, Sheen S, Choi DW, Leem JH, Lee SY. A comprehensive study of deaths due to exposure to humidifier disinfectant in Korea: focusing on medical records, assessment of exposure to humidifier disinfectants, and causes of death. Epidemiol Health. 2021; 43: e2021091. https://doi. org/10.4178/epih. e2021091
- Kim HD, Kim HC, Leem JH. Toxic hepatitis after exposure to humidifier disinfectant: A case series report. Environ Anal Health Toxicol. 2023; 38 (1): e2023002-0. https://doi. org/10.5620/eaht.2023002
- Makarov VK, Ryasensky DS. Assessment of the influence of polyhexamethylene guanidine hydrochloride consumption on the lipid composition of blood serum // Toxicological Bulletin. 2009; 3 (96). Russian
- Pummi K, Kemppi E, Lammintausta K. Occupational sensitization to polyhexamethylene guanidine hydrochloride in a non-alcoholic hand rub. Contact Dermatitis. 2012; 66 (6): 348-9. https://doi. org/10.1111/j.1600-0536.2012.02017.x
- Vitt A, Gustafsson A, Ramberg P, Slizen V, Kazeko LA, Buhlin K. Polyhexamethylene guanidine phosphate irrigation as an adjunctive to scaling and root planing in the treatment of chronic periodontitis. Acta Odontol Scand. 2019; 77 (4): 290-295. https://doi. org/10.1080/00016357.2018.1541099
