RESEARCH ARTICLE
Comparative Analysis of Phytochemical Composition of Aqueous Extracts from Azadirachta indica and Vernonia amygdalina
Sciences of Phytochemistry|Vol. 3, Issue 2, pp. 91-97 (2024)
Received
Aug 11, 2024Revised
Oct 5, 2024Accepted
Oct 20, 2024Published
Oct 24, 2024
Abstract
Introduction
Medicinal plants, such as Azadirachta indica (A. indica) and Vernonia amygdalina (V. amygdalina), have garnered significant attention due to their traditional use and the broad range of bioactive compounds they contain, making them valuable in pharmaceutical applications (1, 2). These plants are rich sources of phytochemicals, which are secondary metabolites that provide defense against environmental threats. Natural products derived from these compounds often exhibit better efficacy and fewer side effects than synthetic ones (3). In many developing countries, especially in rural areas, medicinal plants are vital in providing primary healthcare (4-6). A. indica, belonging to the Meliaceae family, is native to the Indian subcontinent but is also found in tropical regions such as Nigeria, commonly known as "dongoyaro" (7, 8). The neem tree leaves, roots, bark, and seeds are utilized in traditional medicine for their wide-ranging medicinal properties (9). Similarly, V. amygdalina, known as the bitter leaf, is a small perennial shrub of the Asteraceae family, commonly found across Africa, especially Nigeria (2). Its distinct bitter taste, lasting for several min, indicates its rich phytochemical content.
Neem leaves have demonstrated antimicrobial properties against dental pathogens and are widely used in dental treatments (10). In Ayurvedic medicine, neem has been utilized to treat common tropical diseases such as fever and malaria (11). Additionally, neem oil is an effective ingredient in mosquito repellents (12). The phytochemical profile of neem includes antimicrobial (13), antifungal (14), immunomodulatory (15), antiviral (16), antidiabetic (17), and anti-inflammatory activities (18), which make it effective against conditions such as cancer, digestive disorders, and skin infections (19). V. amygdalina is also valued for its medicinal properties, particularly for treating diabetes and fever (20, 21). Its roots are used for toothache relief (22), and the aqueous extract has shown significant blood sugar-lowering effects in rats, comparable to standard diabetic drugs (23). Bitter leaf is also recognized as a fat-burning tonic, with quercetin, a compound found in the plant, demonstrated to alleviate inflammatory bowel disease (24). Furthermore, it is a rich source of dietary fiber and has shown potential in cancer management through its bioactive peptides and chloroform extracts, which target cancer-related cellular pathways (25, 26).
Despite advancements in treating tropical diseases, illnesses such as malaria, typhoid fever, yellow fever, and scabies continue to pose significant public health risks in regions where they are endemic (27). The growing resistance of parasites to current drugs underscores the need for novel antimicrobial agents (28, 29). Traditional herbal medicine remains a crucial resource for managing tropical diseases, particularly in Africa, where access to modern healthcare can be limited (29). Given the importance of A. indica and V. amygdalina in traditional medicine and their similar uses, this study aims to assess and compare the phytochemical constituents of both plants. Using qualitative and quantitative analysis, the study will explore the phytochemical profiles of the aqueous extracts of these plants, providing a foundation for future research on their potential as sources of new antimicrobial and therapeutic agents.
Materials and Methods
Collection and Preparation of Plants
A. indica and V. amygdalina(leaves)were collected from a garden around Poly-road in Auchi community, Edo, Nigeria, at coordinates 7°04'3.22"N 6°15'48.96"E, situated at an altitude of 205.42 m. Mr. Ogbemudia O. Murphy, a plant biology and biotechnology graduate at the University of Benin, identified and authenticated the specimens, and voucher numbers UBH-V342 and UBH-A184 were deposited. Subsequently, all samples were harvested on 13 June 2024. Specimens were air-dried at room temperature for 15 d until they became crispy, preserving all the nutrients and phytochemicals.
Preparation of Crude Powder
After 15 d of air drying, properly dried specimens' leaves were powdered using an electric grinder. The products were sieved to extract fine powder. The powder was stored in an airtight plastic container in a dark room at 25 °C before use.
Preparation of Crude Stock Solutions
For every specimen, 100 g of crude powder was soaked in 1000 mL of distilled water in a separate step and left in an airtight plastic bottle overnight for maceration. The mixture was filtered in a Whiteman filter paper No. 42, boiled for about 4 mins in a heating mantle, and left to cool. Stock solutions were refrigerated at 4 °C for future use.
Qualitative Phytochemical Analysis
Phytochemical screening of the aqueous extract of A. indica and V. amygdalina was conducted to qualitatively determine the presence or absence of the following secondary metabolites that are: alkaloids, tannins, flavonoids, saponins, glycosides, saponins, and phenolic, using the method outlined by Shrestha et al. (2015) (30) and Sofowora (1993) (31).
Saponins
A solution of 2 mL of crude extract was diluted with 20 mL of distilled water and shaken in a test tube for 10 min. The accumulation of a 2 cm layer of foam indicates saponin's presence. After adding two drops of olive oil to the foam mixture and giving it a good shake, the formation of an emulsion was monitored (32).
Phenolic Compounds
A solution of 2 mL of crude extract was treated and mixed with 3 mL of 10% ferric chloride (FeCl3) solution. The formation of a bluish-black, bluish-green, or violet coloration indicates the presence of phenolic compounds or a phenolic hydroxyl group (33).
Flavonoids
A few drops of 10% lead acetate (Lead (II) ethanoate) solution were combined with 2 mL of the treated crude extract solution. The formation of a yellow-colored precipitate indicates the presence of flavonoids (33).
Alkaloids
Hager's Reagent (Saturated Picric Acid Solution)
The crude extract solution was combined with 2 mL of picric (carbazolic) acid. A yellowish precipitate's formation indicates alkaloids' presence (34).
Mayer's Reagent
A 2 mL crude extract was taken in a test tube, and 3 drops of Mayer's reagent were added. The formation of a green-colored precipitate indicates the presence of alkaloids in the solution, thereby confirming the test positive (34).
Tannins
A 1% gelatin containing sodium chloride (NaCl) solution was mixed with 2 mL of crude extract solution. The formation of a white-colored precipitate indicates the presence of tannins (33).
Glycosides
After mixing 2 mL of crude extract with 2 mL of 5% FeCl3 solution, the mixture was heated in a boiling water bath for 4–5 min. The mixture was allowed to cool and then extracted with an equal volume of benzene. After being separated, the benzene layer was treated using 2 mL of ammonia solution. The formation of a pink or rose-pink color in the ammoniacal later indicates the presence of glycosides (33).
Terpenoids
A 2 mL of crude extract was dissolved in 2 mL of chloroform and evaporated. 3 mL of concentrated sulfuric acid (H2SO4) was added carefully along the wall of the test tube. The formation of a reddish-brown coloration at the interface indicates the presence of terpenoids (35).
Quantitative Phytochemical Analysis
The quantitative phytochemical test was done using gravimetric methods as described by Harborne (1973) (36), Indumathi et al. (2014) (37), and Muhammed and Abubakar (2016) (35).
Terpenoids
Dried plant extract (crude) 10 g (initial weight, Wi) was taken and soaked in 90 mL of ethanol. The extract is then filtered, mixed with petroleum ether, and filtered again using a separating funnel. The extract was dried and weighed (final weight, Wf) (37). The following formula yields the percentage of total terpenoid (Eq. 1):
Flavonoids
Dried plant extract (crude) 6 g was placed in an Erlenmeyer flask with 100 mL of distilled water, and 2 mL of hydrochloric acid (HCl) solution was added. The solution is boiled for 20 min and allowed to cool, after which it is filtered into Whiteman No. 42 filter paper. The aqueous layer was discarded and filtered with a pre-weighed filter paper. The residue is dried for 25 mins at 50 ℃ (38). The percentage yield was calculated using Eq. 1.
Alkaloids
In an Erlenmeyer flask, 5 g of crude material was dissolved in 100 mL of 10% acetic acid. It was well combined and left to settle for four hours to ensure optimal extraction. After that, filter paper was used to filter the sample. Using a heated plate for evaporation, the filtrate was concentrated to a quarter of its initial volume. Concentrated ammonium hydroxide (NH4OH) was added in drops to precipitate the alkaloids in the filtrate. The solution was filtered and washed with 1.5% NH4OH. Filter paper containing residue was dried at 40 °C in an oven before being weighed (39). The percentage yield was calculated using Eq. 1.
Saponins
A solution of 50 mL plant extract was placed in a conical flask. 200 mL of 50% alcohol was added and boiled under reflux for 25 min and filtered while still hot through a filter paper. 2 g of charcoal was added and boiled, then filtered while hot. The filtrate was left to cool, and an equal amount of acetone was added, completely precipitating the saponins. The saponins were collected and dried (35). The percentage yield was calculated using Eq. 1.
Glycosides
A solution of 10 mL crude extract was transferred to a 100 mL Erlenmeyer flask, and 60 mL of distilled water and 10 mL of 12.5% lead acetate were added, mixed, and filtered. In a different flask, 50 mL of filtrate was transferred, and 10 mL of 47% Na2HPO4 was added to precipitate excess Pb2+ ions. This was mixed and completed to volume with distilled water. The mixture was filtered twice to eliminate excess lead phosphate. For treatment, 20 mL of purified filtrate was put in a clean flask and mixed with 20 mL of Baljet reagent. A blank titration was carried out using 20 mL of Baljet reagent and 20 mL of distilled water. This was allowed to stand for an hour for complete color development. The color intensity was measured calorimetrically at 495 nm (36). The percentage yield was calculated using Eq. 1.
Design and Analysis
Simple percentages were calculated to determine the occurrence of phytochemicals in the two plant samples. The data were subjected to variance and mean separation by DMRT at a 5% significance level.
Results and Discussion
Qualitative Phytochemical Analysis of Azadirachta indica and Vernonia amygdalina
The phytochemical constituents detected in the aqueous extract of A. indica and V. amygdalina are shown in Table 1. Existing phytochemicals include alkaloids, flavonoids, terpenoids, saponins, tannins, glycosides, and phenolic compounds. Recently, significant studies on phytochemistry have been conducted on plants, focusing on their leaves, bark, roots, seeds, and stems (40). Natural products or secondary metabolites present in plants have been reported to have many biological, pharmaceutical, and therapeutic properties. They are extensively used in drug production industries (41). Owning to the diversity between A. indica and V. amygdalina, Table 1 reveals that flavonoids, alkaloids, and terpenoids were most present in the aqueous extract of A. indica. Saponins and glycosides were moderately concentrated, while tannins and phenolic compounds were found in low concentration.
| Test types | Azadirachta indica | Vernonia amygdalina |
|---|---|---|
| Saponin test | ++ | ++ |
| Terpenoid test | +++ | ++ |
| Tannin test | + | + |
| Glycoside test | ++ | + |
| Alkaloid test | +++ | ++ |
| Flavonoid test | +++ | +++ |
| Phenolic test | + | + |
| Note: the signs are indications of color change and intensity. +++: Most present; ++: Moderately present; +: Least present | ||
Our finding agrees with a study by Krupa et al. (2023) identifying the presence of flavonoids, alkaloids, terpenoids, saponins, glycosides, tannins, and phenolic compounds in A. indica (42). In the qualitative phytochemical analysis of V. amygdalina, it was noted that flavonoids were most present, alkaloids, terpenoids, and saponins were moderately concentrated, and tannins, phenolic compounds, and glycosides were found in low concentration. The result agrees with a study on the aqueous extract of V. amygdalina by Usunomena et al. (2016) and Raimi et al. (2020), identifying the presence of flavonoids, alkaloids, terpenoids, saponins, and glycosides (32, 43). Flavonoids, alkaloids, terpenoids, saponins, glycosides, tannins, and phenolic compounds were present in the extract used, which contrasts with research by Ajayi et al. (2017) with no alkaloid present in aqueous extract of V. amygdalina (44).
Quantitative Phytochemical Analysis of Azadirachta indica and Vernonia amygdalina
The gravimetric method was used to qualitatively analyze the phytochemicals present in A. indica and V. amygdalina as described by Harborne (1973) (36), Indumathi et al. (2014) (37), and Muhammed and Abubakar (2016) (35). Quantitative screening of saponins, terpenoids, glycosides, alkaloids, and flavonoids was done with the leaves of plants (A. indica and V. amygdalina), and the data is shown in Figure 1. The highest percentage of flavonoid compound was obtained from the neem plant (13.80%), which is only marginally higher than the percentage present in bitter leaf (13.62%). The highest percentage of terpenoids was obtained from the neem plant (13.13%), greater than that of bitter leaf (8.21%). The highest percentage of Alkaloids was obtained from the neem plant (10.67%), slightly different from that of bitter leaf (9.68%). Glycosides and Saponins were observed to be of lower percentage. The glycoside in the neem plants (6.78%) was higher than in the bitter leaf (4.11%), and similarly, saponins were higher in the neem plants (7.43%) than in the bitter leaf (6.87%).
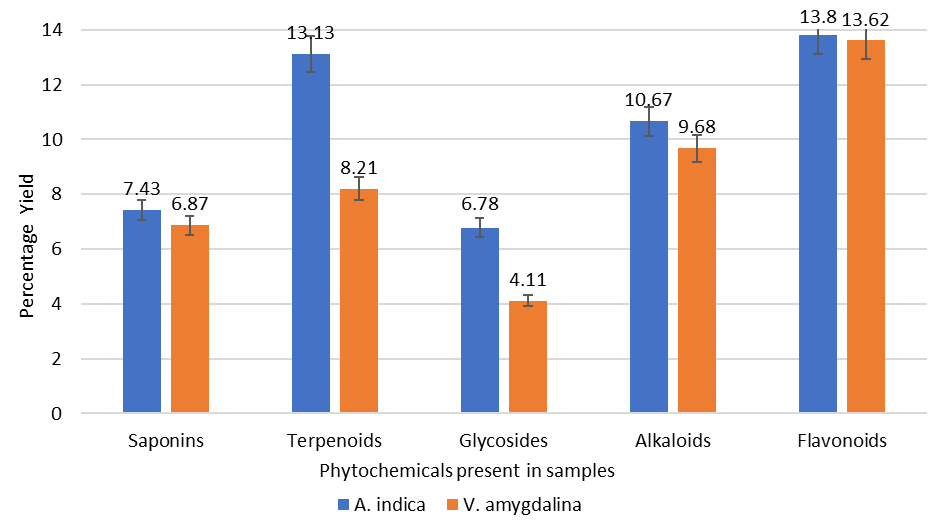
The plants employed in this study were discovered to contain a key component needed to fight human illnesses. Many substances scavenge free radicals, including flavonoids, tannins, alkaloids, phenolic acids, and other metabolites. Phenol derivatives are also used as ingredients in the manufacturing of sunscreens (45). Many biologically active terpenoids are used in combating inflammation, cancer, malaria, and infectious disorders (46). It is evident that the high percentage of alkaloids and saponins present in these plants has assisted in treating fever and serves as dietary supplements (35). The therapeutic potential of glycosides derived from medicinal plants cannot be overemphasized. We look at their functions in terms of cardiovascular, antibacterial, anticancer, and anti-inflammatory (47). Alkaloids, tannins, flavonoids, saponins, and phenolic compounds are some of the phytochemical components that give these plants their therapeutic action; each part of the plant has a complex active ingredient that is advantageous from a pharmacological standpoint (48). According to the nutritional composition of the plants, it's crucial to remember that these plant components can be genetically altered to boost output and enhance quality.
Conclusion
The results revealed the existence of medicinally important phytochemicals and bioactive compounds in A. indica and V. amygdalina leaves. Among the various qualitative tests done to determine the natural product found in A. indica and V. amygdalina leaves, it was observed that the two plant samples contained phenolic chemicals, alkaloids, flavonoids, terpenoids, saponins, tannins, and glycosides. The difference in phytochemical percentage for saponins, alkaloids, and flavonoids was relatively low, as the calculated percentage difference was less than 10%, and those of terpenoids and glycosides were greater than 40%. It is further observed from the study that the percentage yields of A. indica are higher than those of V. amygdalina. They can both serve the same medicinal purposes. This result supports the substitution of both plants in herbal medicine, as they are both used in place of one another. Previous studies have confirmed phytochemicals to be bioactive, so they may be suggested and recommended for further research to identify the active components of A. indica and V. amygdalina, serving as one of the alternatives to utilize secondary metabolites found in medicinal plants and replace harmful synthetic chemicals.
Declarations
Acknowledgment
The author would want to extend their gratitude to the Faculty of Physical Sciences, Faculty of Education, University of Benin; School of Applied Science and Technology, Biological Science Laboratory Technology Department, Auchi Polytechnic, Auchi, Edo, Nigeria, and Laboratory staff for their valuable assistance and support in carrying out this study.
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
Not applicable.
Funding Information
Not applicable.
References
- Wylie MR, Merrell DS. The Antimicrobial Potential of the Neem Tree Azadirachta indica. Front Pharmacol. 2022 May 30;13.
- Ogwu MC, Ikhajiagbe B. Vernonia amygdalina Delile (Asteraceae): An Overview of the Phytochemical Constituents, Nutritional Characteristics, and Ethnomedicinal Values for Sustainability. In 2024. p. 1–29.
- Atanasov AG, Zotchev SB, Dirsch VM, Supuran CT. Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov. 2021 Mar 28;20(3):200–16.
- Maroyi A. Traditional use of medicinal plants in south-central Zimbabwe: review and perspectives. J Ethnobiol Ethnomed. 2013 Dec 4;9(1):31.
- Yebirzaf Y, Esubalew T, Workinesh T. The dynamics of medicinal plants utilization practice nexus its health and economic role in Ethiopia: A review paper. Int J Biodivers Conserv. 2019 Jan 31;11(1):31–47.
- Sofowora A, Ogunbodede E, Onayade A. The role and place of medicinal plants in the strategies for disease prevention. African J Tradit Complement Altern Med. 2013 Aug 14;10(5).
- Liauw MY, Natan FA, Widiyanti P, Ikasari D, Indraswati N, Soetaredjo FE. Extraction of Neem Oil (Azadirachta Indica A. Juss) Using N-hexane and Ethanol: Studies of Oil Quality, Kinetic and Thermodynamic. ARPN J Eng Appl Sci. 2008;3(3):49–54.
- Olaniyi AM, Oshiobugie MJ, Raphael AO. Experimental and Mathematical Model for the Antimalarial Activity of Methanolic Root Extract of Azadirachta indica (Dongoyaro) in Mice Infected with Plasmodium berghei NK65. J Adv Math Comput Sci. 2020 Aug 3;68–82.
- Tesfaye B, Tefera T, Misikir O, Tsegaye G. Extraction and Comparison of Essential Oil from Neem Seed by Using Soxhlet Extraction and Simple Distillation Methods. Int J Eng Technol Manag Res. 2018;S(9):74–81.
- Chaturvedi P, Bag A, Rawat V, Jyala SN, Satyavali V, Jha PK. Antibacterial effects of Azadirachta indica leaf and bark extracts in clinical isolates of diabetic patients. Natl J Integr Res Med. 2011;2(1):5–9.
- Owusu-Boadi E, Akuoko Essuman M, Mensah G, Ayamba Ayimbissa E, Boye A. Antimicrobial activity against oral pathogens confirms the use of Musa paradisiaca fruit stalk in ethnodentistry. Evidence-Based Complement Altern Med. 2021;2021:8663210.
- Al-Hashemi ZSS, Hossain MA. Biological activities of different neem leaf crude extracts used locally in Ayurvedic medicine. Pacific Sci Rev A Nat Sci Eng. 2016;18:128–31.
- Altayb HN, Yassin NF, Hosawi S, Kazmi I. In-vitro and in-silico antibacterial activity of Azadirachta indica (Neem), methanolic extract, and identification of Beta. d-Mannofuranoside as a promising antibacterial agent. BMC Plant Biol. 2022;22(1):1–4.
- Singh UP, Maurya S, Singh A, Singh M. Antifungal activity of neem ( Azardirachta indica ) toddy. Arch Phytopathol Plant Prot. 2010 Jan;43(2):133–9.
- Sarkar S, Singh RP, Bhattacharya G. Exploring the role of Azadirachta indica (neem) and its active compounds in the regulation of biological pathways: An update on molecular approach. 3 Biotech. 2021;11(4):178.
- Hemdan BA, Mostafa A, Elbatanony MM, El-Feky AM, Paunova-Krasteva T, Stoitsova S, et al. Bioactive Azadirachta indica and Melia azedarach leaves extracts with anti-SARS-CoV-2 and antibacterial activities. PLoS One. 2023;18(3):e0282729.
- Ghodesawar MG, Jadaramkunti UC, Aladakatti RH. Assessment of Antidiabetic and Antioxidant Potential of Lyophilized Neem Leaf Powder (LNLP) Supplementation in Alloxan-Induced Diabetic Male Albino Rats. J Pharm Sci Res. 2023;15(5):1118–30.
- Schumacher M, Cerella C, Reuter S, Dicato M, Diederich M. Anti-inflammatory, pro-apoptotic, and anti-proliferative effects of a methanolic neem (Azadirachta indica) leaf extract are mediated via modulation of the nuclear factor-κB pathway. Genes Nutr. 2011 May 14;6(2):149–60.
- Wylie MR, Merrell DS. The antimicrobial potential of the neem tree Azadirachta indica. Front Pharmacol. 2022;13:891535.
- Crellin JK, Philport J, Tommie B. A Reference Guide to Medicinal Plants: Herbal Medicine, Past and Present. Duke University Press; 1989. 205 p.
- Imaga NOA, Bamigbetan DO. In vivo biochemical assessment of aqueous extracts of Vernonia amygdalina (Bitter leaf). Int J Nutr Metab. 2013;5(2):22–7.
- Ademola IO, Eloff JN. Anthelminthic activity of acetone extract and fractions of Vernonia amygdalina against Haemonchus contortus eggs and larvae. Trop Anim Health Prod. 2011;43(2):521–7.
- Onyibe PN, Edo GI, Nwosu LC, Ozgor E. Effects of Vernonia amygdalina fractionate on glutathione reductase and glutathione-S-transferase on alloxan induced diabetes wistar rat. Biocatal Agric Biotechnol. 2021;36:102118.
- Ugbogu EA, Emmanuel O, Dike ED, Agi GO, Ugbogu OC, Ibe C, et al. The phytochemistry, ethnobotanical, and pharmacological potentials of the medicinal plant-Vernonia amygdalina L. (bitter leaf). Clin Complement Med Pharmacol. 2021;1(1):100006.
- Iruoghene Edo G. Analysis of phytochemical constituents and antioxidant potential of bitter kola leaf extract towards bioactive food, nutrition and health resources. Org Med Chem Int J. 2022;5(11).
- Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024 May 4;74(3):229–63.
- Baral B, Mamale K, Gairola S, Chauhan C, Dey A, Kaundal RK. Infectious diseases and its global epidemiology. In: Nanostructured Drug Delivery Systems in Infectious Disease Treatment. Elsevier; 2024. p. 1–24.
- Ogbeide OK, Sajere O, Obukuru IS, Akedayi P, Omorefe NO, Unachukwu CI, et al. Comparative Studies on the Bioactive Chemical Composition, Antioxidant and Antimalarial potentials of Picralima nitida Leaf and Stem Bark Extracts. J Med Plants Res. 2023;
- Oghogho UI, Ekugum E, Ogbeide OK, Idagan MI, Uadia JO, Falodun A. Phytochemical Assessment, Anti-inflammatory and Antimalarial Activities of Beta vulgaris (Chenopodiaceae) Root Extract. Trop J Phytochem Pharm Sci. 2022;1(1):3–8.
- Shrestha P, Adhikari S, Lamichhane B, Shrestha BG. Phytochemical screening of the medicinal plants of Nepal. IOSR J Environ Sci Toxicol Food Technol. 2015;6:11–7.
- Sofowora A. Medicinal plants and traditional Medicine in Africa. 2nd ed. Ibadan, Nigeria: Spectrum Books LTD.; 1993. 191-289 p.
- Usunomena U, Ngozi OP. Phytochemical analysis and proximate composition of Vernonia amygdalina. Int J Sci World. 2016;4(1):11–4.
- Oligie MHA, Eduwuirofo LO, Iyekekpolor RM, Oghomwenrhiere O, Ejimadu CM, Obasuyi EI, et al. Phyto-Constituents, GC-MS Analysis and Antifungal Activity of Methanol Extract of Sweet Orange Peels. NIPES-Journal Sci Technol Res. 2023;5(2).
- Hashmi HF, Bibi S, Anwar M, Rashid Khan M. Qualitative and quantitative analysis of phytochemicals in Lepidium Pinnatifidum Ledeb. Sch Int J Tradit Complement Med. 2021;4(5):67–75.
- Muhammad SA, Abubakar SM. Qualitative and Quantitative Determination of Phytochemicals in Aqueous Extract of Chrysophyllum albidum Seed Kernel. Biosci Biotechnol Res Asia. 2016;13(2):1201–6.
- Harborne JB. Phenolic compounds. In: Phytochemical Methods. Dordrecht: Springer; 1973. p. 33–88.
- Indumathi C, Durgadevi G, Nithyavani S, Gayathri PK. Estimation of terpenoid content and its antimicrobial property in Enicostemma litorrale. Int J ChemTech Res. 2014;6(9):4264–7.
- Khanal S. Qualitative and quantitative phytochemical screening of Azadirachta indica Juss. plant parts. J Med Plants. 2021;122–7.
- Mokogwu ATH, Okorie UP, Digban KA, Abubakar A, Mokogwu EE. Phytochemical and trace element analysis of Vernonia amygdalina (Bitter leaf) in different locations in Nigeria. African J Cell Pathol. 2014;3(12):44–50.
- Kala S, Johnson M, Raj I, Bosco D, Jeeva S. Preliminary Phytochemical Analysis of Some Medicinal Plants of South India. J Nat Conscientia. 2011;2:478–81.
- Chaachouay N, Zidane L. Plant-derived natural product: A source for drug discovery and development. Drugs Drug Candidates. 2024;3(1):184–207.
- Krupa RJ, Vijayakumar K, Sujith S, Vergis J, Deepa PM, George A, et al. Phytochemical analysis of aqueous extract of Azadirachta indica leaves. Pharma Innov J. 2023;SP-12(10):1505–11.
- Raimi CO, Oyelade AR, Adesola OR. Phytochemical screening and in-vitro antioxidant activity on Vernonia amygdalina (ewuro-bitter leaf). Eur J Agric For Res. 2020;8(2):12–7.
- Ajayi A, Ude AN, Balogun OJ. Qualitative and quantitative phytochemical analysis of Moringa oleifera and Vernonia amygdalina. Fulafia J Sci Technol. 2017;3(2):51–7.
- Svobodová A, Psotová J, Walterová D. Natural Phenolics in the Prevention of UV-induced Skin Damage. Biomed Pap. 2003;147:137–45.
- Mbaveng AT, Hamm R, Kuete V. Harmful and protective effects of terpenoids from African medicinal plants. In: Toxicological Survey of African Medicinal Plants. Elsevier; 2014. p. 557–76.
- Riaz T, Akram M, Laila U, Zainab R, Khalil MT, Iftikhar M, et al. Therapeutic applications of glycosides obtained from medicinal plants. Int Arch Integr Med. 2023;10(8).
- Georgewill OA, Georgewill UO. Evaluation of the Anti-inflammatory Activity of Extract of Vernonia amygdalina. Asian Pac J Trop Med. 2010;150–1.