RESEARCH ARTICLE
Potential of Ciplukan Leaf (Physalis Angulate L.) Infusion as A Bronchodilator in Pulmonary Histopathology of Sprague Dawley White Rats
Sciences of Pharmacy|Vol. 3, Issue 1, pp. 61-69 (2024)
Received
Jan 29, 2024Revised
Mar 11, 2024Accepted
Mar 12, 2024Published
Mar 20, 2024
Abstract
Introduction
Indonesia stands as a nation endowed with abundant natural resources. Presently, these resources are not only harnessed for sustenance but also serve as raw materials for crafting traditional remedies, envisioned to address various ailments. Moreover, natural substances can be used effectively and safely. One of the extensively employed traditional medicinal practices, based on empirical usage, involves the utilization of Ciplukan leaves (1).
Ciplukan ( Physalis angulate L), a plant belonging to the Solanaceae family, is characterized by hollow stems and reaches a height of approximately one meter. Originating from tropical and subtropical regions, notably Central America. Ciplukan exists in three distinct variations in Indonesia, particularly within the Javanese region: (P. angulate L), (Physalis minima L), and (Physalis peruviana L) (2). Within Indonesian communities, the leaves of Ciplukan (P. angulate L), are employed as a traditional remedy for asthma. The preparation involves boiling the leaves in water and subsequently consuming the decoction (3). According to existing research, the Ciplukan plant demonstrates a range of pharmacological effects, including antimicrobial, antiparasitic, anti-inflammatory, antinociceptive, antimalarial, immunosuppressive, diuretic, antitumor, and anti-asthmatic activities (4).
Asthma is a respiratory disorder that poses a global health challenge in both developed and developing nations. It is estimated that there are 339 million asthma sufferers worldwide (5). Asthma constitutes a chronic inflammatory ailment of the airways, involving various types of cells and cellular components, particularly mast cells, eosinophils, T lymphocytes, macrophages, neutrophils, and epithelial cells. In susceptible individuals, this inflammation leads to recurring symptoms such as shortness of breath, chest tightness, and coughing, particularly during the night and early morning. These symptoms are associated with widespread airway obstruction, which can either spontaneously resolve or be managed through treatment. The inflammation also triggers an augmented bronchial hypersensitivity to various stimuli that existed previously. The reversibility of airflow limitation may be incomplete in some asthma patients (6).
Asthma, a chronic respiratory ailment, is prevalent in 1-18% of populations across various developed and developing countries, emerging as a global health concern that spans all age groups. The prevalence of asthma in adults is lower than in children. Approximately 300 million people worldwide suffer from asthma, and this number is projected to rise to 400 million by 2025 (7).
Fundamentally, asthma treatment comprises two distinct categories: long-term control medication (controller) and quick-relief medication (reliever). Controllers are administered to mitigate chronic airway inflammation, manage symptoms, reduce the risk of exacerbations, and mitigate lung function decline. Relievers, on the other hand, are dispensed when asthma symptoms deteriorate (8).
Ciplukan leave has been used by the Indonesian people for various diseases, such as asthma. It contains chemical compounds including saponins, flavonoids, polyphenols, chlorogenic acid, sugars, elaic acid, and fisalin. The anti-asthmatic effect of ciplukan could be because this plant contains alkaloids. Due to the presence of an alkaloid compound in the form of atropine which has a relaxing effect on the smooth muscles of the respiratory tract, ciplukan leaves can be used as an alternative asthma therapy. In vivo, research is frequently employed as a basis for determining effectiveness through the administration of substances or compounds to experimental animals. Testing using in vivo methods offers advantages as the data acquired can be extrapolated to establish effective dosages for humans (9). Building on the aforementioned premise, the researcher intends to conduct an efficacy assessment of Ciplukan leaf (P. angulate L. ) infusion as an anti-asthmatic bronchodilator through in vivo experimentation. The specific aim of this study was to examine the bronchodilator effect of ciplukan leaf infusion (P. angulata L. ) based on reducing the thickness of the bronchiolar epithelium, decreasing the number of eosinophils and the number of mast cells in experimental animals of white rats.
Experimental Section
Materials
Animal cages, wire mesh, feeding and drinking vessels for the experimental animals, digital scale, Omron nebulizer (MSG NB 0420), oral syringe cannula, Olympus light microscope (Zeiss Primo Star Brightfield), histopathology slide preparation tools.
The reagents used encompassed ovalbumin (Worthington Biochemical Corporation), ventolin nebules containing salbutamol, aluminum hydroxide, sodium chloride, and 10% formalin buffer.
Animals Preparation
Sprague Dawley white rats weighing between 140-200 grams and aged approximately 2-3 months were utilized as experimental animals. The rats were procured from the Breeding Animal Veterinarian Laboratory. The selection of experimental animals was randomized, and their weights were measured one day before the commencement of the experiment to ensure homogeneity in body weight within groups. Ethical approval was sought from the Ethics Commission at the University of Indonesia with approval number of 134/UN2. F3.11/PDP.02.00/2023. The experimental animals used in this study were obtained from the Breeding Animal Veterinary Laboratory. Prior to subjecting the experimental animals to treatments, a 7-day acclimatization period was implemented in the testing area, during which the rats were provided with ad libitum access to food and water. This acclimatization aimed to familiarize the rats with their new environment and minimize stress. Adaptation encompassed factors such as lighting, room conditions, feeding, and drinking. To ascertain the required number of experimental subjects per treatment group, the Federer formula was employed. A total of 24 male rats were chosen as the experimental subjects for this study, and they were randomly allocated into 6 groups, each containing 4 rats.
Plant Identification
The material used was Ciplukan leaves (P. angulata L. ) obtained from the Research Institute for Spice and Medicinal Crops (BALITTRO) in Bogor. The Ciplukan plant (P. angulate L. ) used in this research was determined at the Laboratory of the Department of Biology, Collection Room of Biota at the University of Indonesia, Jakarta.
Preparation of Ciplukan Leaf Infusion
The infusion of Ciplukan leaves was prepared by weighing approximately 2 g of the powdered leaves and placing them in an infusion pot. The leaves were soaked in 100 mL of distilled water and heated over a water bath for 15-30 minutes from the point at which the temperature within the pot reached 90°C. The mixture was occasionally stirred during this process. The infusion was then squeezed while hot through flannel cloth and filtered using a Büchner funnel. The residue was further combined with 500 mL of water, and the extraction process was repeated. This process continued until the desired volume of the Ciplukan leaf infusion was obtained. Phytochemical screening was conducted for alkaloids, flavonoids, steroids/terpenoids, tannins, and saponins.
Administration of Experimental Treatment to Test Animals
The testing process is conducted over 29 days. On the testing day, rats are individually weighed, and subsequently, each rat is appropriately marked on its tail to distinguish among the subjects. The specific treatments corresponding to the group allocations outlined in Table 1 will be implemented:
| No. | Experimental groups | Types of treatments |
|---|---|---|
| K1. | Normal | No treatment administered |
| K2. | Negative control (-) | Ovalbumin 0.35 mg (intraperitoneal injection) |
| K3. | Positive control (+) | Ovalbumin 0.35 mg+ salbutamol |
| K4. | Dose I | Ovalbumin 0.35 mg + Ciplukan leaf infusion at a dose of 200 mg/kg body weight |
| K5. | Dose II | Ovalbumin 0.35 mg + Ciplukan leaf infusion at a dose of 400 mg/kg body weight |
| K6. | Dose III | Ovalbumin 0. 35 mg + Ciplukan leaf infusion at a dose of 800 mg/kg body weight |
| Note: Each group consists of four rat specimens | ||
In the normal control group, white rats are provided only with food and water. Apart from the normal group, allergen induction with ovalbumin (OVA) will be conducted in two stages. For the first stage, sensitization will be performed using OVA 0.35 mg + 1 mg AlOH3 in 0.9% NaCl intraperitoneally, administered twice on days 1 and 8. The second stage involves re-sensitization with OVA 0.35 mg + 1 mg AlOH3 in 0.9% NaCl through inhalation, administered three times on days 15, 17, and 19. The positive control group will receive inhalation treatment using salbutamol and Ciplukan leaf infusion at dose levels 1 (200 mg/kgBW), 2 (400 mg/kgBW), and 3 (800 mg/kgBW) for 1 week from day 20 to day 28, concurrently. The inhalation process will be performed by placing the test animals inside a chamber connected to an Omron CompAir Compressor Nebulizer, allowing for nebulization over 20 minutes. On day 29, euthanasia will be carried out by cervical dislocation, followed by dissection to obtain lung organ samples from all treatment groups.
Data Collection
Histopathological Preparation and Hematoxylin Eosin Staining
The histopathological preparations were conducted sequentially as follows: euthanasia, fixation, dehydration, clearing, sectioning, Hematoxylin-Eosin (HE) staining, and mounting.
Measurement of Bronchiolar Epithelium Thickness, Eosinophil Count, and Mast Cell Count
The lung tissue sections stained with Hematoxylin Eosin were observed under a microscope at 400x magnification with five fields of view. Epithelial thickness of the bronchioles was measured five times using pixel-based measurements in micrometers (μm). The eosinophil count was performed by observing and enumerating cells with characteristic bilobed nuclei stained reddish. Eosinophil counts were obtained from five fields of view in lung tissue specimens from each rat in the six treatment groups. Meanwhile, mast cell counts were conducted by observing and enumerating cells with the darkest staining and numerous granules. Microscopic images of the preparations were captured using image software. The collected data were subsequently subjected to descriptive analysis by comparing microscopic observations of lung tissue from each treatment group to the control group. The data obtained from the measurements of bronchiolar epithelium thickness, eosinophil count, and mast cell count will be subjected to data processing using Microsoft Excel software and statistically analyzed using Statistical Product and Service Solution (SPSS) version 26.
Results and Discussion
Phytochemical Screening
Phytochemical screening was conducted using a qualitative method involving chemical reagents. The compounds tested included alkaloids, saponins, tannins, phenolics, flavonoids, glycosides, triterpenoids, and steroids. The results of phytochemical screening are presented in Table 2.
| Identification | Test Results |
|---|---|
| Alkaloids Saponins Tannins Phenolics Flavonoids Glycosides Triterpenoids/Steroids | + + + + + - + |
Epithelial Bronchioles Thickness Measurement Results
The parameter employed in this research is the epithelial bronchioles thickness, aiming to assess whether the administration of P. angulate leaf infusion has the potential to ameliorate inflammation or swelling in the bronchiolar epithelial tissue. The following are the results of the epithelial bronchioles thickness measurements presented in Table 3. It's evident that there are fluctuations in the epithelial bronchioles thickness measurements across different groups (K1 to K6), each subjected to various treatments or conditions. These measurements serve as indicators of potential changes in inflammation or swelling within the bronchiolar epithelial tissue following treatment with P. angulate leaf infusion.
Comparing the mean thickness values across the groups, it appears that Group K3 exhibits the lowest mean thickness at 334.69, followed by Group K4 with a mean thickness of 421.70. On the other hand, Group K2 has a mean thickness of 584.95, making it the highest among the groups. This suggests that Group K3 and K4 experienced the most significant reduction in bronchioles thickness, indicating a potential amelioration of inflammation or swelling.
As depicted in Figure 1, Image K1 represents the microscopic view of lung tissue that did not receive any treatment (normal control), exhibiting no signs of inflammation or thickening in the epithelial tissue. In image K2, lung tissue is depicted with allergen ovalbumin sensitization and no therapeutic intervention, resulting in inflammation and tissue thickening. The microscopic depiction of image K3 shows lung tissue with allergen ovalbumin sensitization and treated with salbutamol therapy, demonstrating a reduction in epithelial thickening diameter. Image K4 displays the epithelial microscopic view of the dose 1 (200 mg/kgBW) treatment group of Ciplukan leaf infusion. The administration of a 200 mg/kgBW dose of Ciplukan leaf infusion indicates results below those of the dose 2 (400 mg/kgBW) and dose 3 (800 mg/kgBW) treatments, suggesting that the 200 mg/kgBW dose has a more significant impact compared to the 400 mg/kgBW and 800 mg/kgBW doses. Treatment with the 200 mg/kg BW dose, under microscopic observation, shows a reduction in epithelial tissue diameter and a potential reduction in epithelial tissue inflammation. This result is not in line with similar research conducted by Luliana S. , et al, which stated that increasing the dose of the infusion of P. angulata L. caused an increasing in the percentage of anti-inflammatory effect (1).
| Treatment Group | Rat No. | Mean ± SD | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| K1 | 323.38 | 452.09 | 555.85 | 648.89 | 495.05 ±139.86 |
| K2 | 645.74 | 574.72 | 561.76 | 557.56 | 584.95 ±41.18 |
| K3 | 406.30 | 200.96 | 407.57 | 323.95 | 334.69 ±97.36 |
| K4 | 302.16 | 440.53 | 362.14 | 581.97 | 421.70 ±120.94 |
| K5 | 393.57 | 575.57 | 579.7 | 382.38 | 482.81 ±109.61 |
| K6 | 472.33 | 530.65 | 389.58 | 547.24 | 484.95 ±71.23 |
| Note: The results of bronchioles epithelial thickness measurements are presented in micrometers (µm). | |||||
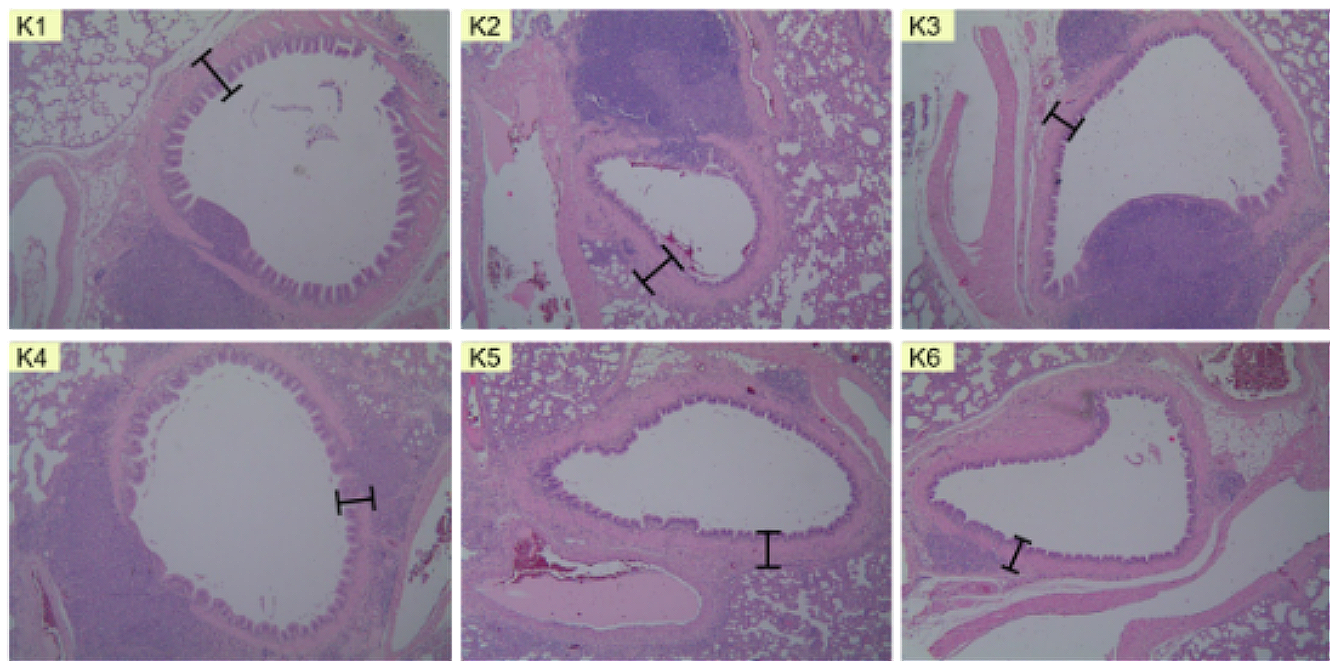
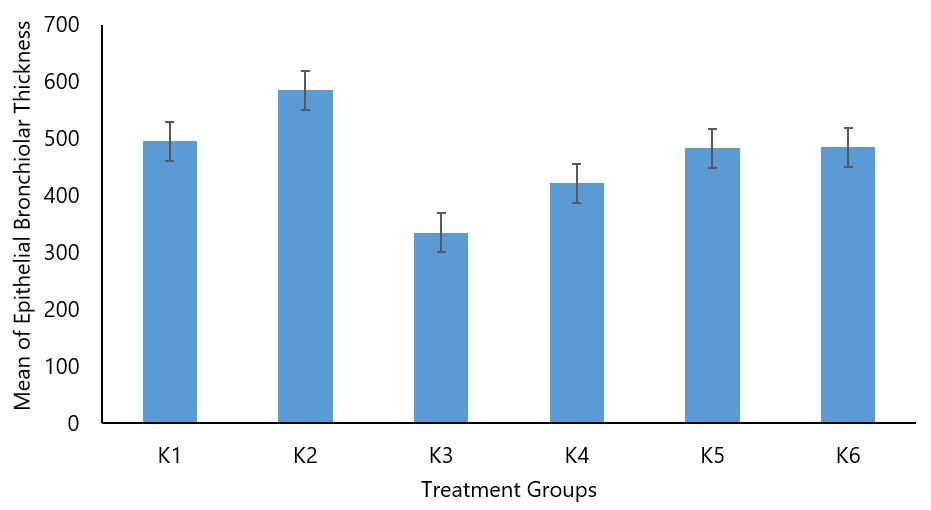
The results of histopathological observations showed that the highest value was found in the negative group (K2) with a mean of 584.95±41.18 µm. The study by Fehrenbach (2017) stated that bronchiolar epithelial damage is present in patients with mild to persistent asthma. This also indicates that mild inflammation caused by epithelial damage is the initial process of mucosal damage and the occurrence of airway remodeling, which consists of several components, including epithelial metaplasia with increased epithelial thickness (10). The comparison between the negative group (K2) with a mean of 584.95±41.18 µm and the salbutamol group (K3) with a mean of 334.69±97.36 µm yielded a p-value of 0.000, indicating a significant difference between the negative and salbutamol groups. The comparison between the negative group (K2) with a mean of 584.95±41.18 µm and the dose of 200 mg/kgBW group (K4) with a mean of 421.70±120.94 µm resulted in a p-value of 0.000, signifying a significant difference between the negative and 200 mg/kgBW dose of Ciplukan leaf infusion groups. The comparison between the negative group (K2) with a mean of 584.95±41.18 µm and the dose of 400 mg/kgBW group (K5) with a mean of 482.81±109.61 µm resulted in a p-value of 0.008, indicating a significant difference between the negative and 400 mg/kgBW dose of Ciplukan leaf infusion groups. The comparison between the negative group (K2) with a mean of 584.95±41.18 µm and the dose of 800 mg/kgBW group (K6) with a mean of 484.95±71.23 µm resulted in a p-value of 0.003, signifying a significant difference between the negative and 800 mg/kgBW dose of Ciplukan leaf infusion groups.
The comparison between the salbutamol group (K3) with a mean difference in bronchiolar epithelial thickness of 334.69±97.36 µm and the dose of 200 mg/kgBW group (K4) with a mean difference of 421.70±120.94 µm yielded a p-value of 0.042, signifying a significant difference. The comparison between the salbutamol group (K3) with a mean difference in bronchiolar epithelial thickness of 334.69±97.36 µm and the dose of 400 mg/kg BW group (K5) with a mean difference of 482.81±109.61 µm resulted in a p-value of 0.001, indicating a significant difference. In the comparison between the salbutamol group (K3) with a mean difference in bronchiolar epithelial thickness of 334.69±97.36 µm and the dose of 800 mg/kgBW group (K6) with a mean difference of 484.95±71.23 µm, the p-value was 0.000, demonstrating a significant difference between the salbutamol and 800 mg/kgBW dose of Ciplukan leaf infusion groups (K6).
Pulmonary Tissue Eosinophil Count
The parameter observed in this study is eosinophil count. Eosinophils are major mediators in asthma, contributing to severe inflammation in tissues. Representative images of eosinophils in lung tissue preparations from each group can be observed in the following figures.
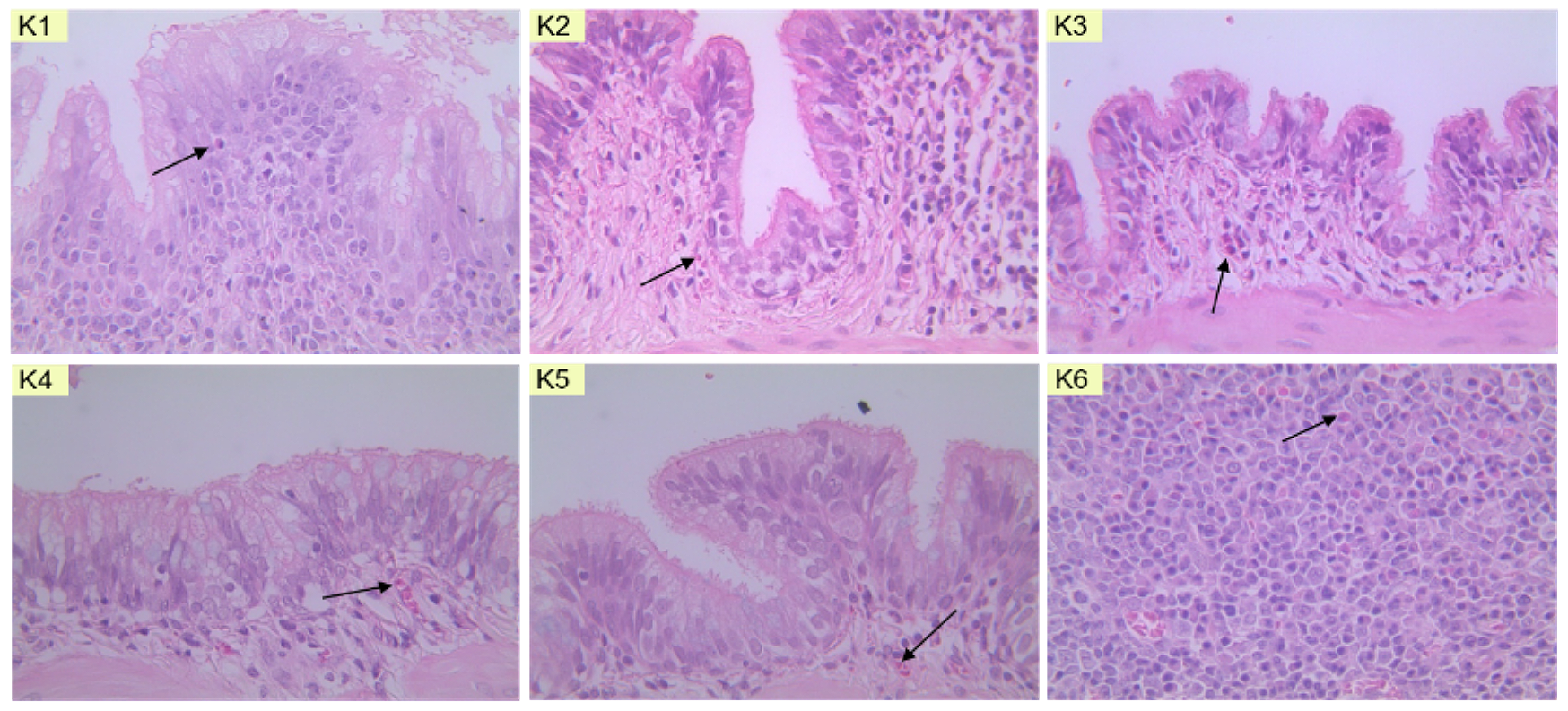
In Figure 3, eosinophils are depicted with a slightly bright reddish color and granulated appearance, with purple or blue staining in the cell nuclei. This coloration results from staining with Hematoxylin Eosin. After the staining process, the prepared samples were observed under a light microscope at 400x magnification, and the eosinophil count was determined in five fields of view. In addition to the microscopic depiction of eosinophil preparations in the lung tissue of rats, the results of the eosinophil count can be seen in Table 4
| Treatment Group | Rate No. | Eosinophil Count per Group | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| K1 | 6 | 4 | 4 | 5 | 19 |
| K2 | 8 | 13 | 10 | 13 | 44 |
| K3 | 8 | 10 | 10 | 9 | 37 |
| K4 | 6 | 6 | 5 | 6 | 23 |
| K5 | 5 | 4 | 5 | 3 | 17 |
| K6 | 8 | 5 | 7 | 7 | 27 |
In Table 4, it can be observed that groups of Dose I, Dose II, and Dose III exhibited a decrease in eosinophil count compared to both the negative control group and the positive control group. There was a notable increase in eosinophil count in the negative control group (ovalbumin allergen-induced group), indicating that the rats had undergone inflammation resulting in lung tissue inflammation. According to the research conducted by Palupi et al. (2020), inflammation is characterized by an elevated count of activated eosinophils, leading to respiratory epithelial damage through the mechanism of eosinophil action on immunoglobulin E (11). The negative control group serves as a comparative benchmark for the dosed Ciplukan leaf infusion treatment groups (200 mg/kgBW dose, 400 mg/kgBW dose, 800 mg/kgBW dose). In the positive control group (induced by ovalbumin with the administration of the anti-inflammatory drug salbutamol), a visibly lower eosinophil count is observed compared to the negative control group (induced by ovalbumin alone), suggesting the anti-inflammatory effect of salbutamol.
In the Dose 2 group (induced by ovalbumin with subsequent inhalation of Ciplukan leaf infusion at a dose of 400 mg/kgBW), a considerable reduction in eosinophil count is evident when compared to the negative control group (induced by ovalbumin alone). This implies that the administration of Ciplukan leaf infusion at a dose of 400 mg/kgBW is sufficiently effective in alleviating asthma symptoms in rats. In Table 4, the reduced eosinophil count values in the Dose 1 group (induced by ovalbumin with subsequent inhalation of Ciplukan leaf infusion at a dose of 200 mg/kgBW) and Dose 3 group (induced by ovalbumin with subsequent inhalation of Ciplukan leaf infusion at a dose of 800 mg/kgBW) in comparison to the negative control group (induced by ovalbumin alone) indicate a decrease in eosinophil count. This suggests that the administration of varying doses of Ciplukan leaf infusion (Dose 1 and Dose 3) has anti-inflammatory effects.
To ascertain whether there are significant differences in eosinophil counts within each group, the calculation of eosinophil numbers was performed across 5 fields of view on each rat lung tissue slide. The results of the mean eosinophil counts in this study are presented in Figure 4.
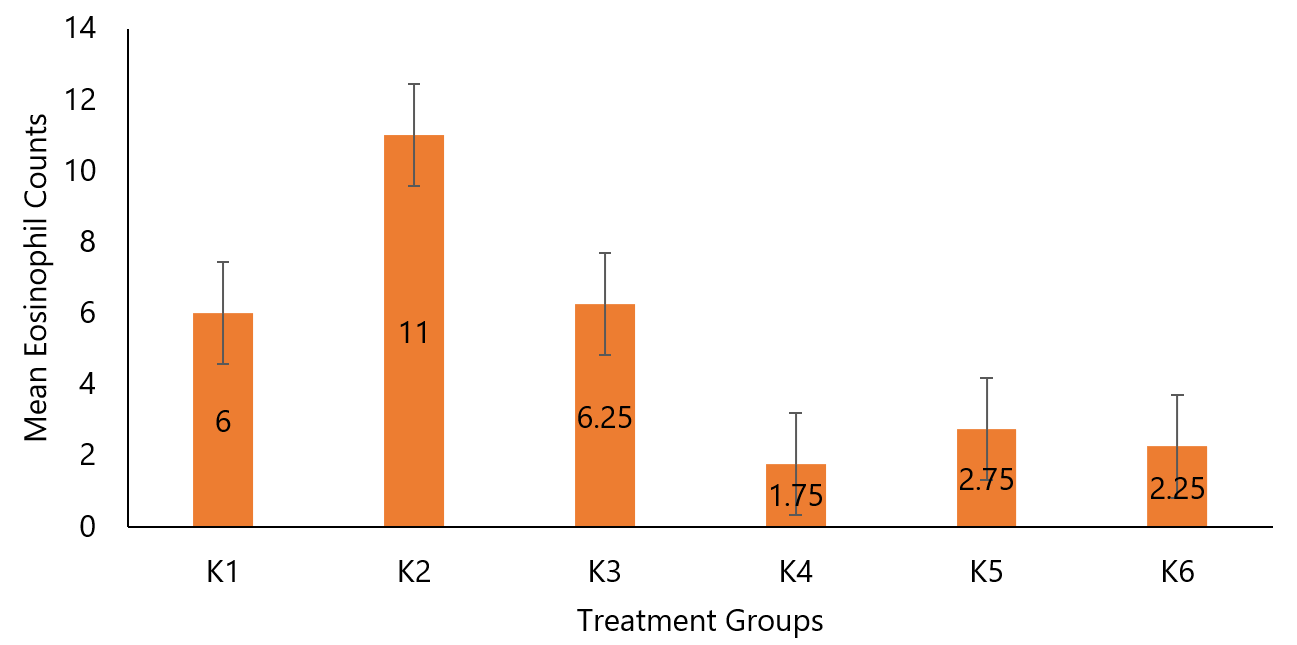
The comparison between the mean eosinophil count of the negative group (K2) with a mean difference of 11±2.45 and the salbutamol group (K3) with a mean difference of 9.25±0.96 yielded a p-value of 0.293 (p> 0.05), indicating no significant difference between the negative and salbutamol groups. The comparison between the negative group (K2) with a mean difference of 11±2.45 and the dose 200 mg/kgBB group (K4) with a mean difference of 5.75±0.5 yielded a p-value of 0.042 (p< 0.05), signifying a significant difference between the negative and dose 200 mg/kgBW Ciplukan leaf infusion groups. Similarly, the comparison between the negative group (K2) with a mean difference of 11±2.45 and the dose 400 mg/kgBB group (K5) with a mean difference of 4.25±0.96 yielded a p-value of 0.004 (p< 0.05), indicating a significant difference between the negative and dose 400 mg/ kgBW Ciplukan leaf infusion groups. However, the comparison between the negative group (K2) with a mean difference of 11±2.45 and the dose 800 mg/kgBB group (K6) with a mean difference of 6.75±1.26 yielded a p-value of 0.159 (p> 0.05), suggesting no significant difference between the negative and dose 800 mg/ kg BW Ciplukan leaf infusion groups.
Based on the research by Kim YH et al. , ovalbumin administration in mice can increase eosinophil recruitment in the lungs, making it a suitable negative control (12). According to the study conducted by Diding, one of the pro-inflammatory cytokines that influence eosinophil recruitment is IL-5 (13).
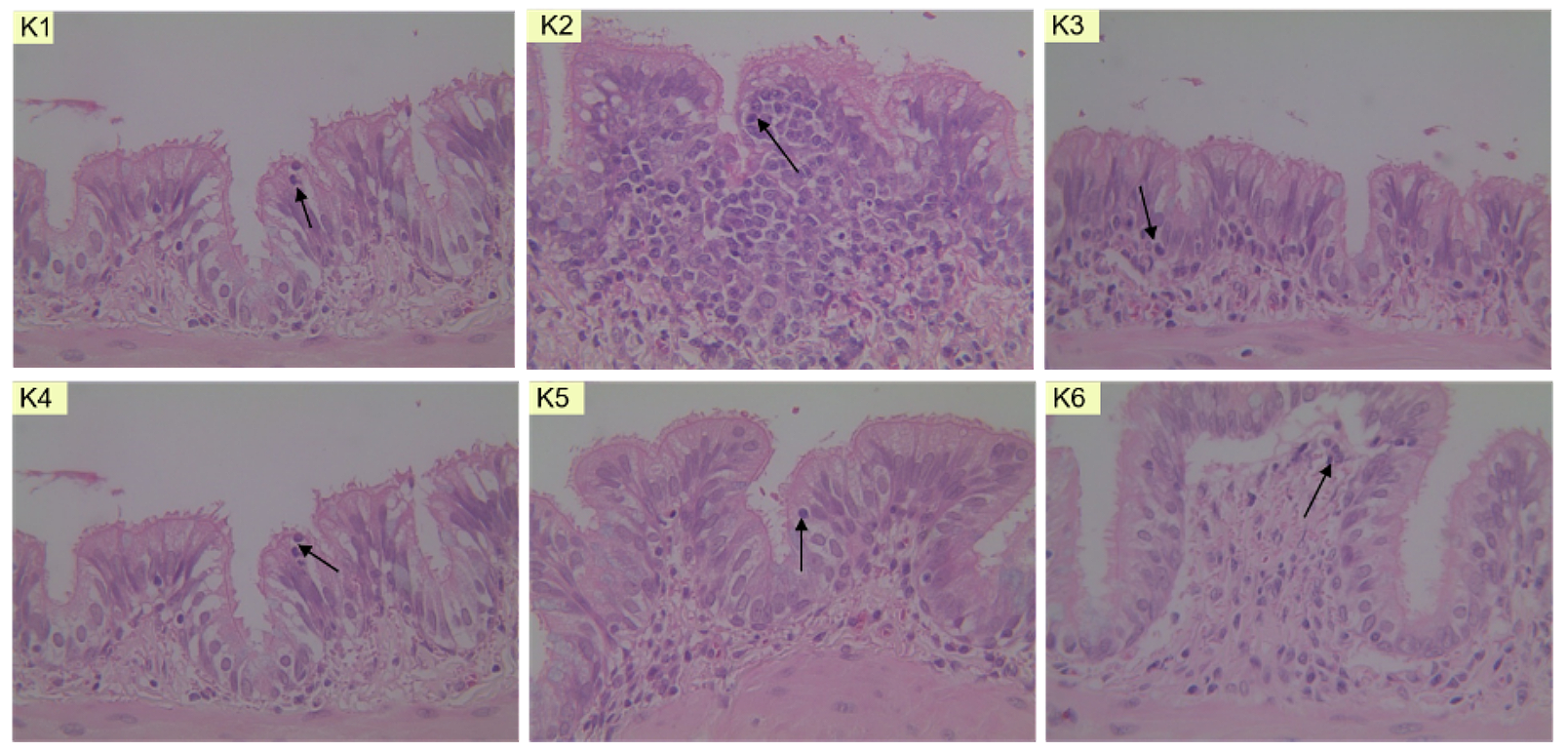
Results of Mast Cell Counts
The parameter observed in this study is mast cells. The analysis of mast cell counts across all groups was conducted by examining lung tissue samples. Histopathological analysis was performed using Hematoxylin Eosin staining, and the samples were microscopically observed at a magnification of 400x. Histopathological images of mast cells are presented in Figure 5.
In Figure 5, mast cells are depicted as having the darkest cellular coloration and exhibiting numerous granules. Following staining, the prepared samples were observed using a light microscope at a magnification of 400x. Subsequently, the eosinophil count was determined across five fields of view. In addition to the microscopic depiction of mast cell preparations within rat lung tissue, the quantification results of mast cell count in this study are presented in Table 5.
| Treatment Groups | Rats No. - | Mast Cell Count per Group | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| K1 | 2 | 3 | 2 | 2 | 9 |
| K2 | 6 | 5 | 8 | 3 | 22 |
| K3 | 2 | 5 | 4 | 2 | 13 |
| K4 | 3 | 4 | 6 | 2 | 15 |
| K5 | 7 | 3 | 1 | 2 | 13 |
| K6 | 0 | 2 | 3 | 2 | 7 |
In the presented table, it is evident that dose groups I, II, and III exhibit a reduction in mast cell counts when compared to both the negative control group and the positive control group. In the negative control group (ovalbumin allergen-induced group), an increase in mast cell count is observed, indicating inflammation in the rat pulmonary tissue. According to the study by Dian A. et al. , TH-2 activation stimulates the release of inflammatory cells, and the high IgE levels on mast cell surfaces lead to mast cell degranulation, consequently releasing inflammatory mediators (14). The negative control group serves as a reference group for the dosed infusion of Ciplukan leaf treatment groups (doses 200 mg/kg BW, 400 mg/kg BW, 800 mg/kg BW) to determine the effectiveness of Ciplukan leaf infusion in reducing mast cell counts.
In the positive control group (ovalbumin-induced group treated with anti-inflammatory drug salbutamol), a decrease in mast cell count is observed compared to the negative control group (ovalbumin-induced group only), indicating the anti-inflammatory effect of salbutamol. Salbutamol, a short-acting beta agonist (SABA), induces the relaxation of smooth muscles in the airway, improving airflow and rapidly relieving acute asthma symptoms (15).
In dose I group (ovalbumin-induced group with subsequent inhalation of Ciplukan leaf infusion at a dose of 200 mg/kgBW) and dose 2 group (ovalbumin-induced group with subsequent inhalation of Ciplukan leaf infusion at a dose of 400 mg/kgBW), a lower mast cell count is observed compared to the negative control group (ovalbumin-induced group only). This suggests that Ciplukan leaf infusion at doses of 200 mg/kgBW and 400 mg/kgBW effectively mitigates asthma symptoms in rats. In the table, a lower mast cell count is demonstrated in dose group 3 (ovalbumin-induced group with subsequent inhalation of Ciplukan leaf infusion at a dose of 800 mg/kgBW) when compared to the negative control group (ovalbumin-induced group only). This reduction in mast cell count indicates the anti-inflammatory effect of administered dose 3 of Ciplukan leaf infusion.
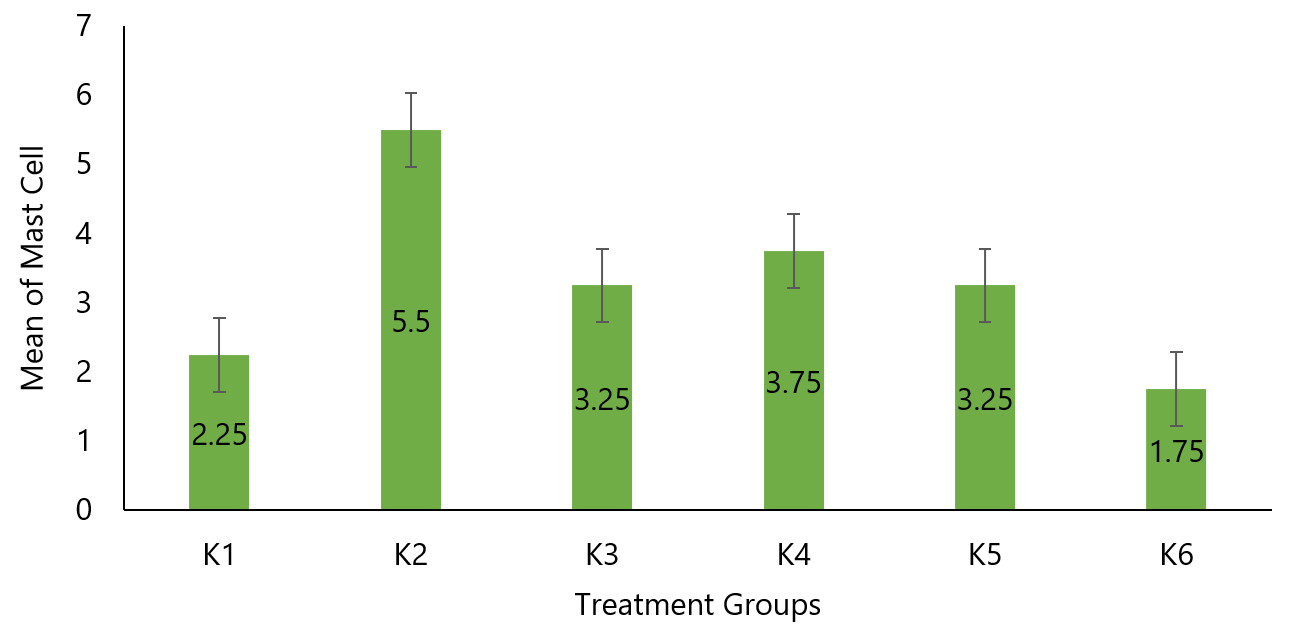
The comparison of mean mast cell counts between the negative control group (K2) with a mean of 5.5± 2.08 and the salbutamol group (K3) with a mean of 3.25±1.5, yielded a p-value of 0.108 (p> 0.05), indicating that there is no significant difference in mast cell counts between the negative control and salbutamol groups. Likewise, the comparison between the negative control group (K2) with a mean of 5.5±2.08 and the group treated with a dose of 200 mg/kgBW (K4) with a mean of 3.75±1.71, resulted in a p-value of 0.243 (p> 0.05), signifying that there is no significant difference in mast cell counts between the negative control and the 200 mg/kgBW Ciplukan leaf infusion group.
Furthermore, the comparison between the negative control group (K2) with a mean of 5.5± 2.08 and the group treated with a dose of 400 mg/kgBW (K5) with a mean of 3.25±2.63, yielded a p-value of 0.191 (p> 0.05), indicating the absence of a significant difference in mast cell counts between the negative control and the 400 mg/kgBW Ciplukan leaf infusion group. Conversely, the comparison between the negative control group (K2) with a mean of 5.5±2.08 and the group treated with a dose of 800 mg/kg BW (K6) with a mean of 1.75±1.26, resulted in a p-value of 0.028 (p< 0.05), demonstrating a statistically significant difference in mast cell counts between the negative control and the 800 mg/kg BW Ciplukan leaf infusion group
Conclusion
In conclusion, the application of Ciplukan leaf infusion exhibits significant anti-asthmatic properties, particularly influencing the bronchiolar epithelium thickness parameter. The dosage of 200 mg/kgBW proves to be the most effective in this regard. Additionally, this infusion also demonstrates effectiveness in reducing eosinophil count, with the dosage of 200 mg/kg BW showing the greatest efficacy. Lastly, for quantifying mast cells in rat specimens, the optimal therapeutic dose appears to be 800 mg/kgBW of Ciplukan leaf infusion.
Declarations
Conflict of Interest
The authors declare no conflicting interest.
Data Availability
The unpublished data is available upon request to the corresponding author.
Ethics Statement
This in vivo test was conducted after obtaining ethical approval from the Ethics Committee of the Faculty of Medicine, Universitas Indonesia - Cipto Mangunkusumo Hospital with ethic number: 134/UN2.F3.11/PDP.02.00/2023. All the actions were taken by minimizing pain and suffering in experimental animals.
Funding Information
Not applicable.
References
- Luliana, S., Susanti, R., & Agustina, E. (2017). Antiinflammatory Activity Test of Aqueous Extracts Herb of Ciplukan (Physalis angulata L.) in Caragenan Inducted Wistar Rat (Rattus norvegicus L.). Traditional Drug Magazine, 22(3), 199. https://doi.org/10.22146/mot.31556
- Pujiasmanto, B., Theresia, M., Budiastuti, S., Rumia, I., & Setyaningrum, D. (2022). "Digitalization of Agriculture Towards Creative Economy Revival": Review [Potential of Ciplukan Planting Media (Physalis angulata)]: A Review. 6(1), 155–162
- Azzikri, et al. Ethnobotany of Medicinal Plants of Solanaceae in the Kerinci Tribe Community in the Lempur Region of Kerinci Regency, Jambi Province. SRIBIOS, Dec 2020; p.12
- Elsa RS, Gabriel VA 2013. Physalis angulata L. (Bolsa Mullaca): A Review of its Traditional Uses, Chemistry and Pharmacology. Boletin Latinoamericano y del Caribe de Plants Medicinales y Aromaticas. 12(5): 431–445
- GAN. The Global Asthma Report 2018. Auckland New Zealand: The Global Asthma Network; 2018. 6–13 p
- Mims, James W. (2015). Asthma: Definitions and Pathophysiology. International Forum of Allergy & Rhinology, 5(S1), S2-S6. doi:10.1002/alr.21609
- Indonesian Society of Respirology. Asthma: Diagnosis and Management Guidelines in Indonesia. PDPI. Jakarta: Indah Offset Citra Grafika; 2019: p.4
- Kam, A., Fauzar, F., Kurniati, R., & Arsyad, Z. (2021). Controller Therapy in Asthma. Journal of Health Sciences Indonesia, 1(3), 438–444. https://doi.org/10.25077/jikesi.v1i3.467
- Lusiastuti, A. M., & Pramudito, H. (2019). In Vivo Method in the Aloe Vera Juice Therapy for Incision Wound of Pseudomonas aeruginosa: Bioethical and Animal Welfare Studies. Proceedings of the National Seminar on Agricultural Bioethics, 60–65.
- Fehrenbach, H., Wagner, C., & Wegmann, M. (2017). Airway remodeling in asthma: What really matters.
- Palupi, D. A. et al. (2020) 'The effect of nigella sativa oil on the number of eosinophils, neutrophils, serum ige levels to asthma mice resistant of corticosteroid', International Journal of Pharmaceutical Research, 12(1), pp. 896–901
- Kim YH, et al. Oleuropein curtails pulmonary inflammation and tissue destruction in models of experimental asthma and emphysema. Journal of Agricultural and Food Chemistry 2018; 66(29): 7643-54. doi: 10.1021/acs.jafc.8b01808
- Diding HP. The Effect of Ovalbumin Aerosol Exposure on Bronchial Eosinophilia in BALB/c Mice. Nexus Medicus, Faculty of Medicine, Sebelas Maret University, Surakarta, 2007: 9-13
- Palupi, D. A., Freistianti, E., & Apriliani, V. E. (2021). Anti-asthma Activity of Red Ginger Extract (Zingiber officinale var rubrum) on Eosinophil Count and Unde-Granulated Mast Cells. Cendekia Journal of Pharmacy, 5(1), 81–91. http://cjp.jurnal.stikescendekiautamakudus.ac.id/index.php/cjp/article/view/134
- Ullmann, N; Caggiano, S; Cutrera, R (2015). Salbutamol and around. Italian Journal of Pediatrics, 41(Suppl 2), A74–. doi:10.1186/1824-7288-41-S2-A74
